Uncategorized
March 6, 2026
Published by admin on March 6, 2026
Short answer: FDA failure is not just a regulatory setback. It is a valuation event. When a medical device company receives a major deficiency letter, Refuse-to-Accept decision, or pathway escalation from the U.S. Food and Drug Administration, investors do not see a technical issue. They see risk materializing. And risk directly affects exits.
March 4, 2026
Published by admin on March 4, 2026
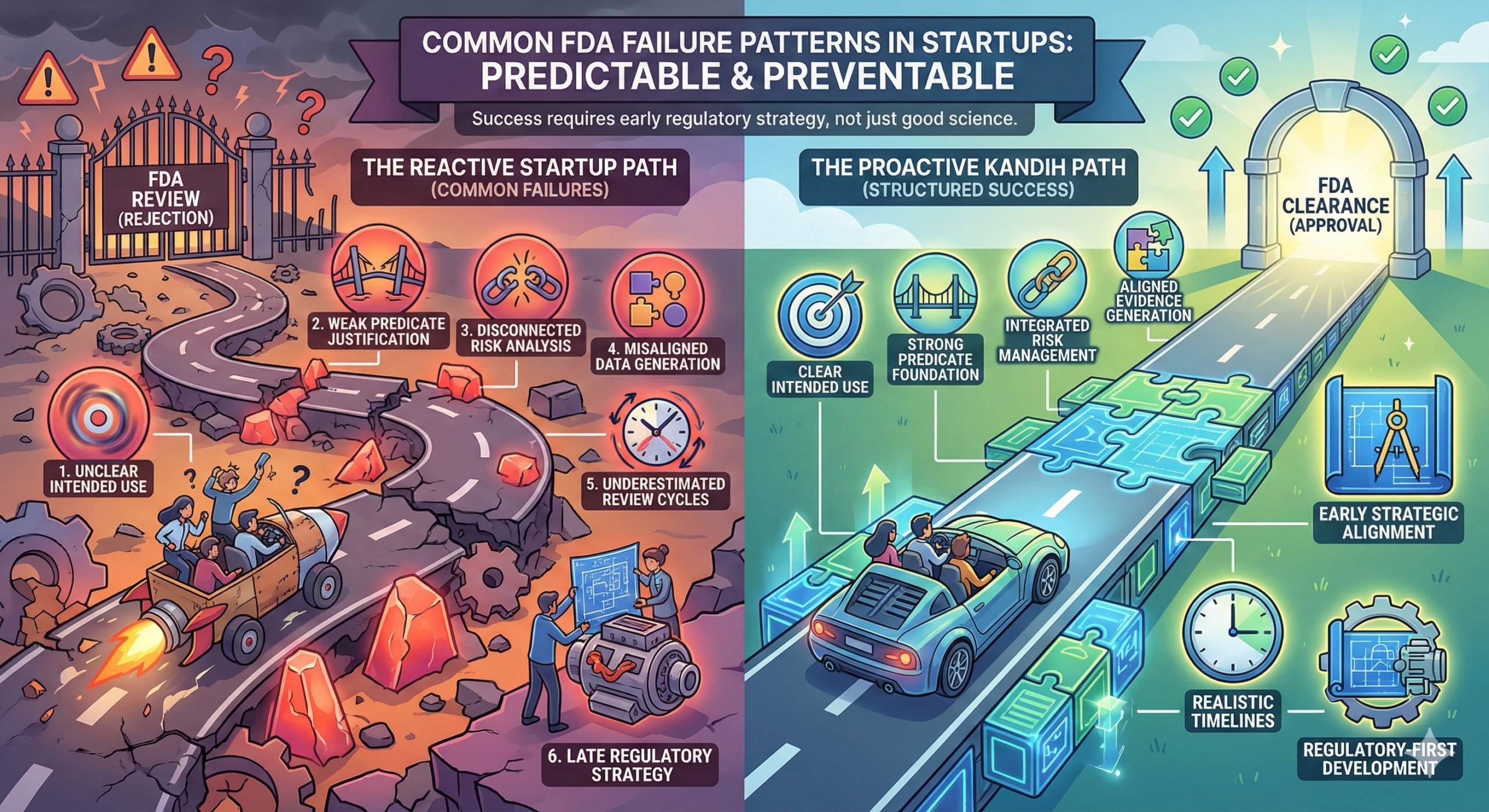
Short answer: most FDA submission failures follow predictable patterns. They are rarely random. They are rarely mysterious. And they are almost always preventable. Startups often assume failure happens because the science was weak. In reality, failure usually happens because the regulatory strategy was fragmented, optimistic, or reactive
February 27, 2026
Published by admin on February 27, 2026
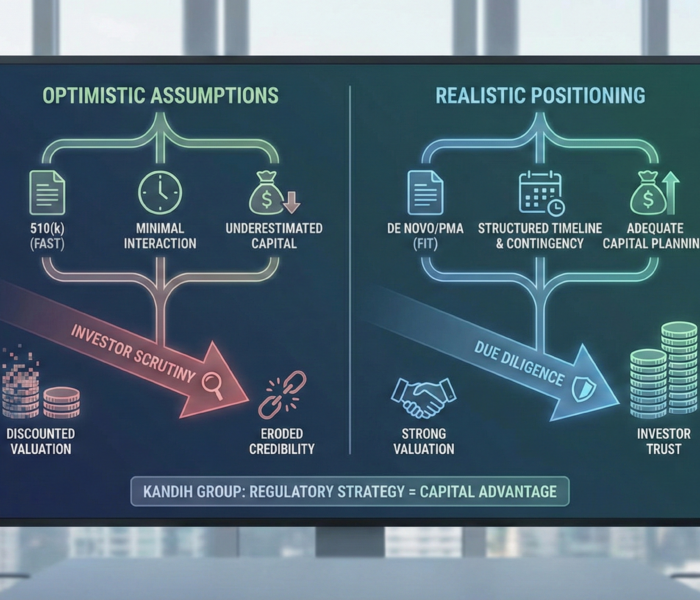
Short answer: investors do not just look at your FDA pathway. They look at how realistic your pathway assumptions are. If your regulatory strategy sounds optimistic, […]
February 25, 2026
Published by Daniel Ndiki on February 25, 2026
We often hear that modern science has "doubled" the human lifespan. We look at the statistics from 1840—where life expectancy was a mere 40 years—and compare them to our 80-year average today, assuming we’ve fundamentally reengineered human biology.
But the data tells a different, more sobering story.