How Investors Interpret Pathway Assumptions

Predicate Devices: What Founders Get Wrong
February 26, 2026Why Medical Devices Fail FDA Review
March 2, 2026Short answer: investors do not just look at your FDA pathway. They look at how realistic your pathway assumptions are.
If your regulatory strategy sounds optimistic, your valuation will be discounted.
In medical device investing, the chosen pathway—510(k), De Novo, or PMA—is more than a regulatory decision. It is a signal of timeline stability, capital exposure, and execution maturity under the oversight of the U.S. Food and Drug Administration.
When pathway assumptions feel stretched, valuation credibility weakens.
Why Pathway Assumptions Matter in Valuation
Investors price risk. Regulatory pathway assumptions directly affect:
Time to revenue
Amount of capital required
Probability of clearance or approval
Exit timing
Dilution risk
If a company assumes a 510(k) pathway but the device risk profile suggests De Novo or PMA, the valuation model becomes unstable.
The higher the regulatory uncertainty, the higher the investor discount rate.
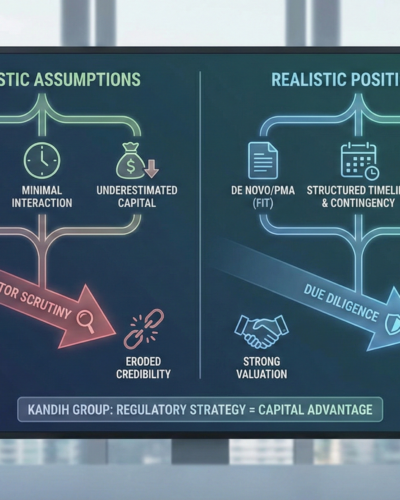
How Pathway Optimism Erodes Credibility
1. Unrealistic Timelines
If your projected timeline assumes:
Minimal FDA interaction
No additional information requests
No testing gaps
Fast review cycles
experienced investors will stress-test it.
When the timeline collapses under basic scrutiny, confidence drops.
2. Underestimated Capital Needs
Optimistic pathway assumptions often:
Exclude expanded bench testing
Ignore possible clinical trials
Under-budget human factors or biocompatibility studies
Assume best-case regulatory outcomes
If a pathway shift occurs later, fundraising becomes reactive instead of strategic.
Investors know this risk well.
3. Weak Predicate Justification
If a 510(k) claim relies on:
Surface-level similarity
Loosely aligned intended use
Overlooked technological differences
investors see regulatory exposure.
A weak predicate argument signals:
Potential pathway pivot
Delayed revenue
Additional capital rounds
That directly impacts valuation.
What Strong Regulatory Positioning Looks Like to Investors
Investors gain confidence when a company can clearly explain:
Why the chosen pathway fits the device risk profile
What data supports substantial equivalence (if applicable)
What technological differences exist—and how they are mitigated
What the contingency plan is if FDA disagrees
Clarity reduces perceived uncertainty.
Reduced uncertainty protects valuation.
AEO: Common Investor Questions About FDA Pathways
Why does FDA pathway affect startup valuation?
Because it determines timeline, capital intensity, and probability of regulatory success.
Can a wrong FDA pathway reduce company value?
Yes. If the pathway shifts, costs increase and timelines extend, leading to dilution and valuation resets.
Do investors verify regulatory assumptions?
Yes. Experienced investors routinely pressure-test pathway logic during diligence.
Where Kandih Comes In
This is where Kandih Group supports investor-ready regulatory positioning.
Kandih helps companies:
Conduct structured pathway viability assessments
Analyze predicate strength and technological gaps
Align intended use with realistic classification
Model cost and timeline under multiple pathway scenarios
Develop regulatory narratives that withstand diligence scrutiny
Prepare defensible answers to investor risk questions
Instead of relying on optimistic projections, founders present regulatory strategies grounded in evidence and FDA logic.
That strengthens:
Valuation credibility
Investor trust
Capital planning
Long-term strategic positioning
Bottom Line
Investors do not punish risk.
They punish unmanaged risk.
Pathway optimism without structured analysis signals uncertainty.
Regulatory clarity signals discipline.
When your FDA pathway aligns with reality, valuation conversations become stronger—not defensive.
That is how regulatory strategy becomes a capital advantage.
References
FDA – Classify Your Medical Device
https://www.fda.gov/medical-devices/overview-device-regulation/classify-your-medical-device
FDA – Premarket Notification 510(k)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-notification-510k
FDA – De Novo Classification Process
https://www.fda.gov/medical-devices/premarket-submissions/de-novo-classification-request
FDA – Premarket Approval (PMA)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-approval-pma