May 11, 2026
Published by admin on May 11, 2026
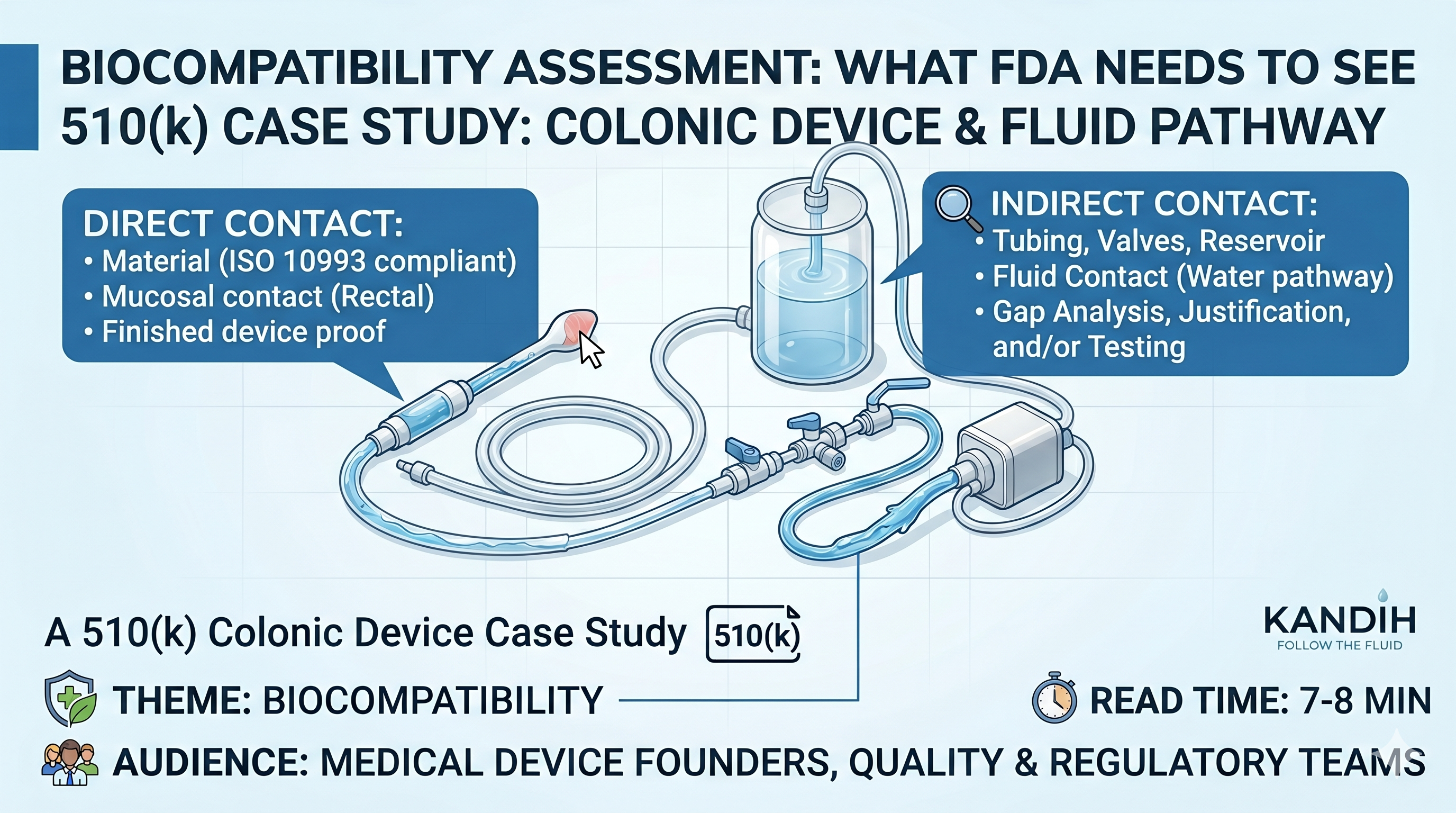
Theme: Biocompatibility Audience: Medical device founders, quality teams, regulatory teams, importers, and anyone trying to understand what FDA may ask in a 510(k) Estimated read time: 7–8 minutes Medical Devices: Irrigation […]
May 4, 2026
Published by admin on May 4, 2026
A device that passes material inspection can still fail ISO 10993 biocompatibility testing — and understanding why the difference between a delay and a disaster is. Kandih Regulatory Insights · The team thought this would be easy. The material was "medical grade." The supplier had used it in healthcare for years. The device seemed simple enough, and the regulatory plan looked clean.
April 27, 2026
Published by admin on April 27, 2026
A founder imports a simple Class I medical device. No electronics.
No software.
No implant.
No dramatic risk profile. The supplier says, “This product has been sold for years.” The customer says, “Send us your biocompatibility testing.” Then the room gets quiet.
April 24, 2026
Published by admin on April 24, 2026
Your 510(k) Timeline Is Decided Months Before You File
Week seven of FDA review.
The email lands.
Subject: Additional Information Request
“Provide clarification regarding exposure assumptions relative to intended use.”
The toxicological risk assessment modeled once-daily adult use on intact skin.
The Instructions for Use allowed multiple daily uses across broader patient populations — including compromised tissue.