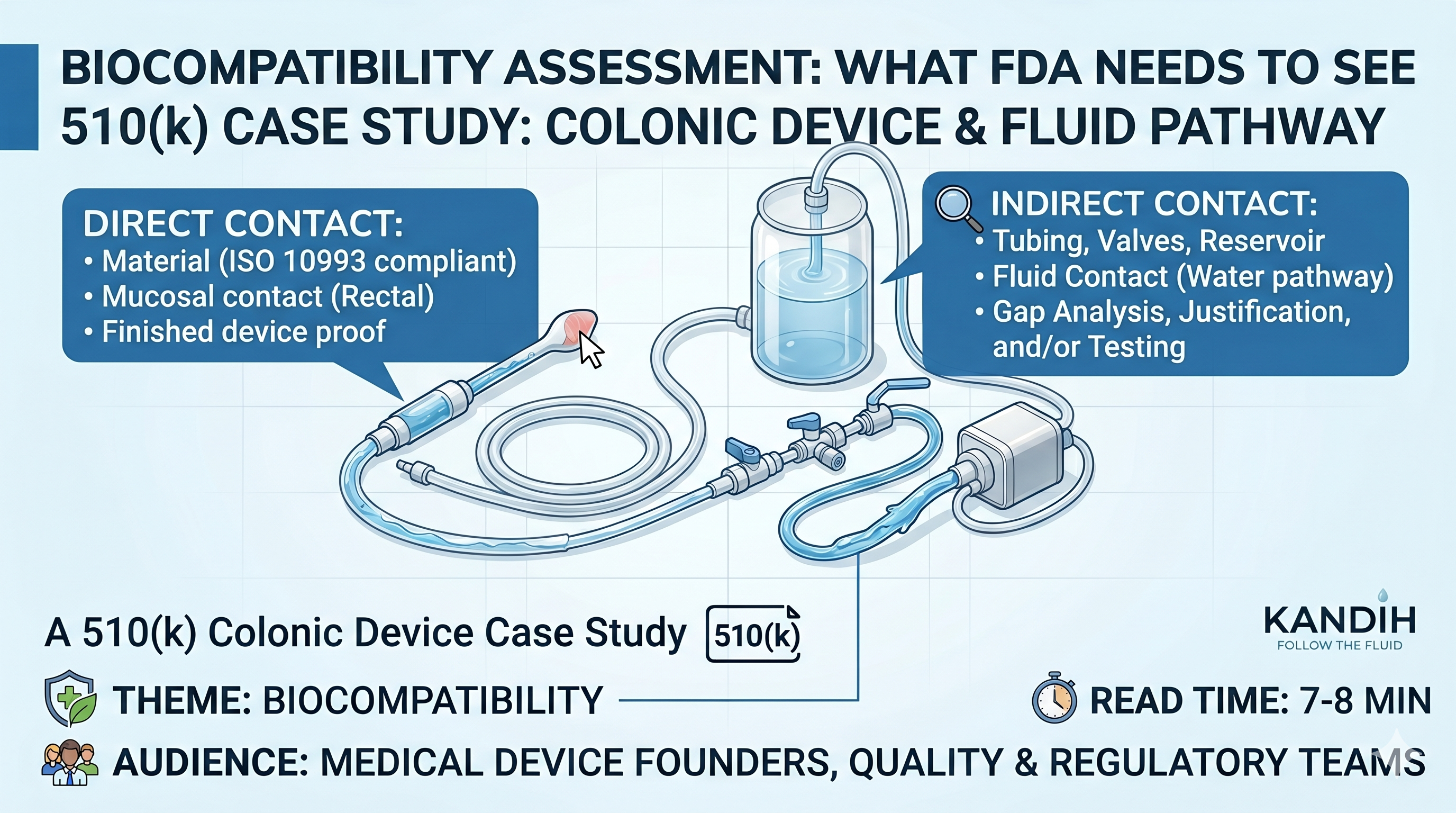
Direct vs. Indirect Contact in ISO 10993: A 510(k) Colonic Device Case

Your material is “medical grade.” So why did it fail?
May 4, 2026Theme: Biocompatibility
Audience: Medical device founders, quality teams, regulatory teams, importers, and anyone trying to understand what FDA may ask in a 510(k)
Estimated read time: 7–8 minutes

Medical Devices: Irrigation System Confusion
The team thought the device was simple.
Water goes in.
Waste comes out.
Only one part goes into the patient.
So someone said:
“Only the nozzle touches the patient. That’s the only part we need to worry about, right?”
Not quite.
That one sentence is where many medical device teams get into trouble.
For a colonic irrigation device, the nozzle may be the only part that goes into the body. But it may not be the only part that matters for biocompatibility.
Why?
Because the water that enters the patient may pass through tubing, valves, connectors, or a reservoir first. Those parts may never touch the patient directly, but they may touch the water that goes into the patient.
That means they can still matter.
This is the difference between direct contact and indirect contact in ISO 10993.
And if you are preparing a 510(k), this difference can decide whether your biocompatibility file looks solid or full of holes.

Executive Summary
Direct contact means a device part touches the patient’s body.
Indirect contact means a device part does not touch the patient, but it touches something that later goes into or onto the patient.
For a colonic irrigation device, the nozzle may have direct contact with the rectal area. Tubing, water containers, connectors, valves, and other water-path parts may have indirect contact if they touch water before that water enters the colon.
The big lesson is simple:
Do not only ask, “What touches the patient?”
Also ask:
“What touches the water, air, or fluid that later touches the patient?”
That is the part many teams miss.

Why This Gets Confusing
A colonic irrigation system may look simple from the outside.
But inside the system, there may be many parts.
These can include:
- a water container
- tubing
- connectors
- valves
- a pump
- a flow control part
- a rectal nozzle
- a waste tube
- reusable parts
- disposable parts
- cleaning or disinfection steps
Some parts clearly touch the patient.
The nozzle is the obvious example.
Other parts may sit outside the body. They may look harmless. But if they touch the water before it goes into the patient, they may still be important.
Think of it like making tea.
The cup touches your mouth. That is direct contact.
But if the water sat in a dirty kettle first, the kettle still matters.
The kettle did not touch your mouth, but it touched the water you drank.
That is indirect contact.
Medical devices work the same way.
What Is Direct Contact?
Direct contact means the device part physically touches the patient.
For a colonic irrigation system, direct contact may include:
- the rectal nozzle
- the catheter or insertion tip
- any seal or sleeve that touches the patient
- any lubricant supplied with the device
- any part that touches rectal tissue or nearby skin
This matters because different body areas have different levels of sensitivity.
A part that touches normal outside skin is one thing.
A part that touches rectal tissue is different.
Rectal tissue is a mucosal surface. That means it is more sensitive than regular skin. So the biological safety review needs to treat it that way.
A company should not simply write:
“The device has external contact.”
That may be too vague.
A better statement would explain exactly what the part touches, how long it touches, and how often it is used.
FDA does not need fancy language.
FDA needs clear thinking.
What Is Indirect Contact?
Indirect contact means the part does not touch the patient, but it touches something that later touches the patient.
For a colonic irrigation system, indirect-contact parts may include:
- tubing that carries water to the patient
- the water reservoir
- connectors in the water path
- valves in the water path
- pump surfaces that contact water
- fittings that contact water
- flow control parts that contact water
These parts may stay outside the body.
But if water passes through them before entering the colon, those parts may affect the patient.
Why?
Because chemicals from those materials could move into the water.
That water then enters the patient.
So the risk is not from the tube touching the patient.
The risk is from the tube touching the water that touches the patient.
That is indirect contact.
The Common 510(k) Mistake
Many teams preparing a 510(k) look at the device and think:
“Only the nozzle goes into the body, so we only need biocompatibility for the nozzle.”
That may be wrong.
For a colonic irrigation device, FDA may ask about the full water path.
They may want to know:
- What materials touch the irrigation water?
- Can anything from those materials get into the water?
- Is the water path disposable or reusable?
- Are there cleaning chemicals involved?
- Are there manufacturing residues?
- Is the water heated?
- How long does the water contact the tubing or reservoir?
- Is the design similar to the predicate device?
- Is there testing or a good scientific reason to support the approach?
This is where teams get surprised.
They thought the question was about one part.
FDA may see the question as a full pathway.
A Simple Example
Imagine a company is preparing a 510(k) for a colonic irrigation system.
The device has:
- a reusable control unit
- a disposable water tube
- a water reservoir
- a valve
- a rectal nozzle
- a waste tube
The team tests only the nozzle.
Then FDA asks:
“What about the parts that contact the water before the water enters the patient?”
Now the team has to explain the tubing, reservoir, valve, and connectors.
If those parts were already included in the biocompatibility plan, great.
If not, the team may need more testing, more justification, or more documentation.
That can slow down the review.
The frustrating part is that this problem is preventable.
You just need to map the device correctly before submission.
The Best First Question: Follow the Fluid
For irrigation devices, one of the easiest ways to think about biocompatibility is this:
Follow the fluid.
Where does the water start?
What does it pass through?
What touches it?
Where does it enter the patient?
What parts touch waste after it leaves the patient?
Which parts are reused?
Which parts are thrown away?
This simple exercise can reveal risks early.
If a part touches water before the water enters the body, do not ignore it.
It may not need the same testing as the nozzle. But it should at least be considered and explained.
Direct Contact Parts: What Should Be Documented?
For direct-contact parts, the file should clearly explain:
- what the part is
- what body area it touches
- what material it is made from
- whether it has colorants or additives
- whether it is sterile or non-sterile
- whether it is single-use or reusable
- how long it touches the body
- what biocompatibility evidence supports it
- whether testing was done
- why any testing was not done
For the nozzle, do not just say:
“Plastic nozzle.”
That is too thin.
The file should explain the material, body contact, contact time, and evidence showing the finished nozzle is safe for its intended use.
The word finished matters.
A raw material certificate may help, but it does not always prove the final part is safe. The final part may include colorants, processing residues, surface changes, or packaging effects.
The patient touches the finished device, not the supplier’s brochure.
Indirect Contact Parts: What Should Be Documented?
For indirect-contact parts, the file should explain what each part touches.
A simple table can help.
| Part | Touches Patient? | Touches Water Going to Patient? | Include in Biocompatibility Review? |
| Rectal nozzle | Yes | Yes | Yes |
| Water tubing | No | Yes | Yes |
| Water reservoir | No | Yes | Yes |
| Valve | No | Yes | Yes |
| External housing | No | No | Usually no |
| Waste tubing | No | Waste side only | Depends on risk |
This type of table is useful because it shows the logic.
It tells the reviewer:
“We looked at the whole device. We did not forget the fluid path.”
That matters.
A clear table can prevent a lot of back-and-forth later.
Does Every Part Need Testing?
No.
This is important.
Considering a part in the biocompatibility review does not always mean testing it.
Sometimes the answer may be:
- testing is needed
- existing data are enough
- chemical information is enough
- supplier documentation supports the material
- the part has no patient contact or fluid-path contact
- the part can be excluded with a clear reason
The goal is not to test every screw, button, and plastic cover.
The goal is to explain what matters and why.
A part may be excluded if it does not touch the patient and does not touch anything that later touches the patient.
For example, the outer plastic housing of a control unit may not need biocompatibility testing if it does not contact the patient or the patient-bound water.
But do not just leave it out quietly.
Say why it is excluded.
Silence creates questions.
Clear reasoning prevents them.
What About Waste Tubing?
Waste tubing can be tricky.
If the tubing only carries waste away from the patient after use, it may not create the same risk as tubing that carries water into the patient.
But it still should be considered.
Questions to ask include:
- Does waste tubing ever contact patient-bound water?
- Could backflow occur?
- Is there a valve preventing backflow?
- Is the waste path separate from the clean water path?
- Is the tubing disposable?
- Is it reused?
- Does it create user exposure risk?
- Could cleaning residues remain?
If the waste path is clearly separate and does not expose the patient to materials or residues, the risk may be lower.
But the file should explain that.
Again, do not make the reviewer guess.
Contact Time Matters
Biocompatibility also depends on how long the device contacts the body or patient-bound fluid.
A device used for a few minutes is different from a device used for hours.
A device used one time is different from a reusable device used again and again.
For a colonic irrigation system, the file should explain:
- how long the nozzle is inserted
- how long water contacts the tubing
- whether the same parts are reused
- whether disposable parts are replaced after each use
- whether cleaning or disinfection is required
- whether repeated use changes exposure
Do not choose the shortest time just because it looks better.
Choose the time that matches real use.
Regulators are not allergic to risk.
They are allergic to weak explanations.
Cleaning and Reuse Can Add New Questions
Reusable devices can create extra biocompatibility concerns.
Why?
Because cleaning and disinfection can leave residues.
If a reusable part contacts water that later enters the patient, then cleaning chemicals may become part of the biological safety question.
The file may need to address:
- cleaning instructions
- disinfection chemicals
- rinse steps
- residue risk
- material compatibility
- reuse limits
- validation evidence
A device may be safe when new but create concerns after repeated cleaning.
That is why reuse should not be treated as a small detail.
For reusable devices, the cleaning process is part of the safety story.
Common Mistakes Teams Make
Mistake 1: Only testing the nozzle
The nozzle matters, but the water path may matter too.
Mistake 2: Ignoring parts outside the body
A part can stay outside the body and still affect the patient if it touches patient-bound water.
Mistake 3: Forgetting cleaning residues
Reusable parts may carry cleaning or disinfection residue risk.
Mistake 4: Depending only on raw material certificates
Raw material data can help, but the finished device still needs to be assessed.
Mistake 5: Copying a predicate device too closely
A predicate helps, but it does not replace your own biological evaluation.
Mistake 6: Not explaining exclusions
If a part is excluded, explain why.
Quick Answer: What Is Direct Contact in ISO 10993?
Direct contact means a device part physically touches the patient’s body.
In a colonic irrigation device, the rectal nozzle or catheter may be a direct-contact part because it touches rectal tissue during use.
Quick Answer: What Is Indirect Contact in ISO 10993?
Indirect contact means a device part does not touch the patient directly, but it touches something that later touches the patient.
In a colonic irrigation system, tubing, reservoirs, connectors, valves, or pump parts may be indirect-contact parts if they touch irrigation water before that water enters the colon.
Quick Answer: Does a 510(k) Need Biocompatibility for Indirect Contact Parts?
Sometimes, yes.
A 510(k) may need biocompatibility evidence for indirect-contact parts if those parts touch water, gas, or fluid that later contacts the patient.
The company should explain which parts are included, which parts are excluded, and why.
The answer should be based on body contact, fluid path, contact time, materials, and risk.
Where Kandih Comes In
Kandih helps medical device companies make sense of direct and indirect contact before it becomes a 510(k) problem.
For colonic irrigation systems and other fluid-path devices, Kandih helps teams answer a basic but important question:
Which parts actually matter for biocompatibility?
Kandih helps with:
- direct vs. indirect contact mapping
- 510(k) biocompatibility gap reviews
- patient-contact and fluid-path assessments
- ISO 10993 biological evaluation strategy
- Biological Evaluation Plans
- Biological Evaluation Reports
- supplier documentation review
- material risk review
- chemical characterization strategy
- toxicological risk assessment coordination
- FDA response support
- customer-facing documentation
Kandih also helps teams avoid two expensive mistakes:
Testing too much because no one has a plan.
Or testing too little because no one followed the fluid path.
The goal is simple:
Build a biocompatibility file that makes sense, answers the obvious questions, and does not collapse when FDA or a customer asks, “What about this part?”
Final Takeaway
Direct contact is easy to see.
Indirect contact is easy to miss.
For a colonic irrigation device, the nozzle may be the obvious patient-contacting part. But the tubing, reservoir, valves, and connectors may also matter if they touch water before it enters the patient.
That does not mean every part needs testing.
It means every part needs to be sorted properly.
A strong biocompatibility file explains:
- what touches the patient
- what touches water going to the patient
- what does not touch the patient or fluid path
- what evidence supports each decision
- why the finished device is safe for its intended use
In plain language:
Follow the patient. Follow the fluid. Explain your choices.
That simple approach can save a company from delays, extra testing, and painful FDA questions.
Because in medical devices, the part you forgot is often the part someone else will notice.
References