June 1, 2026
Published by admin on June 1, 2026
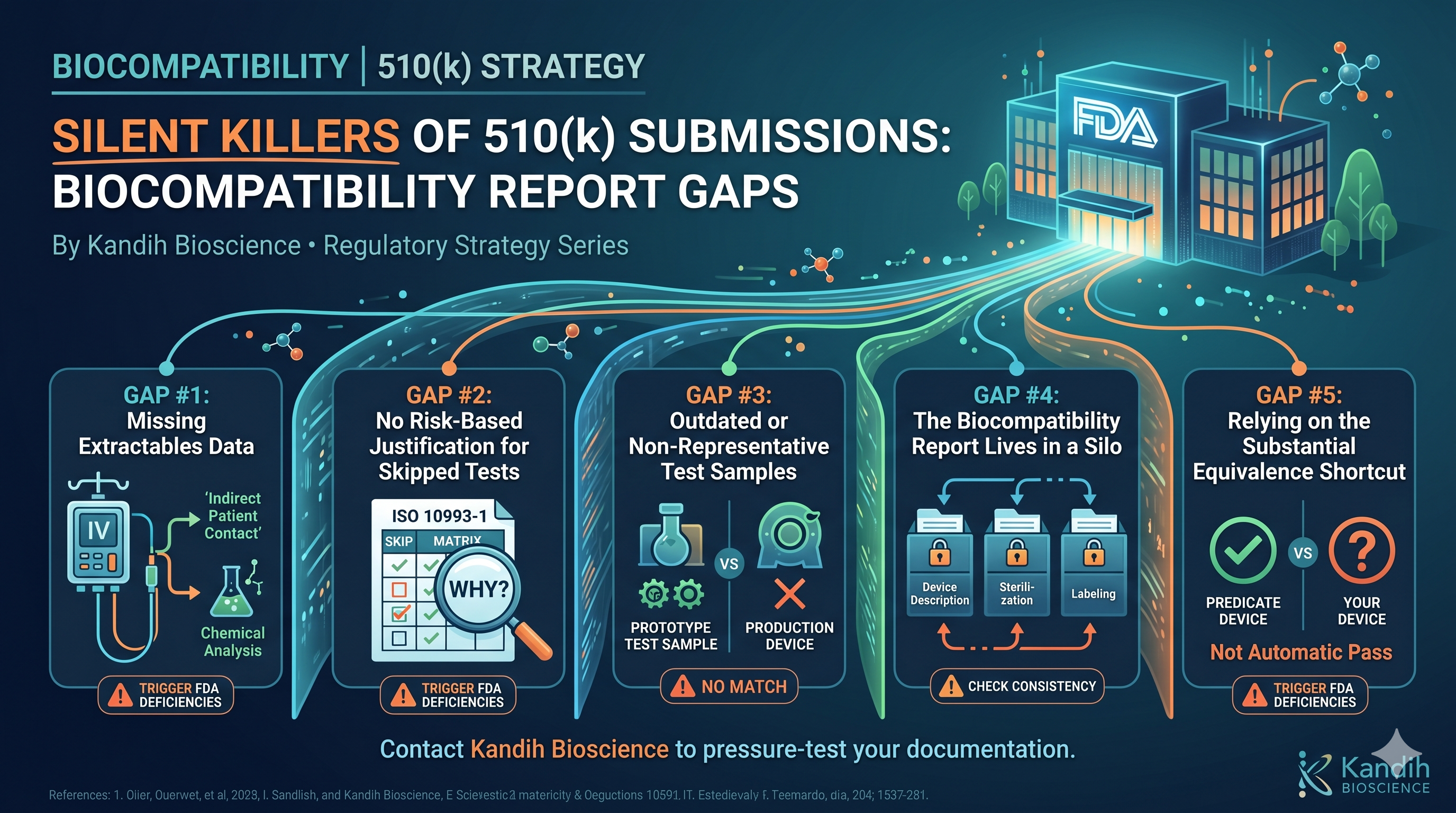
Silent Killers of 510(k) Submissions: Biocompatibility Report Gaps By Kandih Bioscience • Regulatory Strategy Series The truth is, it rarely comes out of nowhere. There are specific, well-documented gaps in […]
May 25, 2026
Published by admin on May 25, 2026
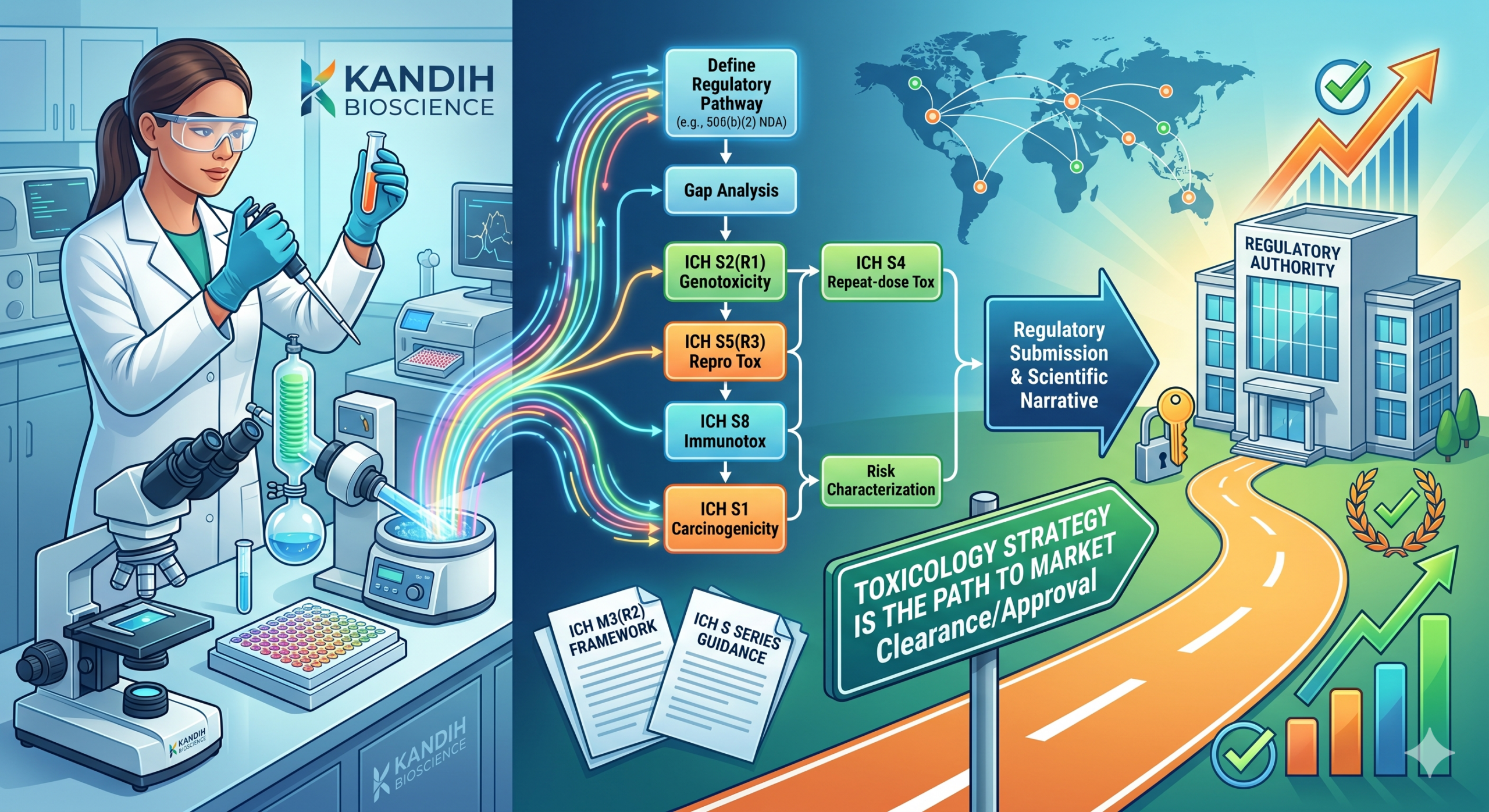
A toxicology strategy is a structured plan that connects your safety data to your regulatory pathway. It does more than confirm that a product won't harm patients — it tells reviewers how you evaluated risk, why your evidence is sufficient, and what that means for market clearance or approval. Without it, even a clean set of tox studies can stall a submission.
May 18, 2026
Published by admin on May 18, 2026
The team had a problem. Their medical device needed a biocompatibility explanation. Not necessarily a huge testing program. Not necessarily a stack of lab reports. But they needed a clear reason why the device was biologically safe. So someone wrote: “The device is made from medical-grade materials and is therefore biocompatible.” That sentence looked harmless.
May 11, 2026
Published by admin on May 11, 2026
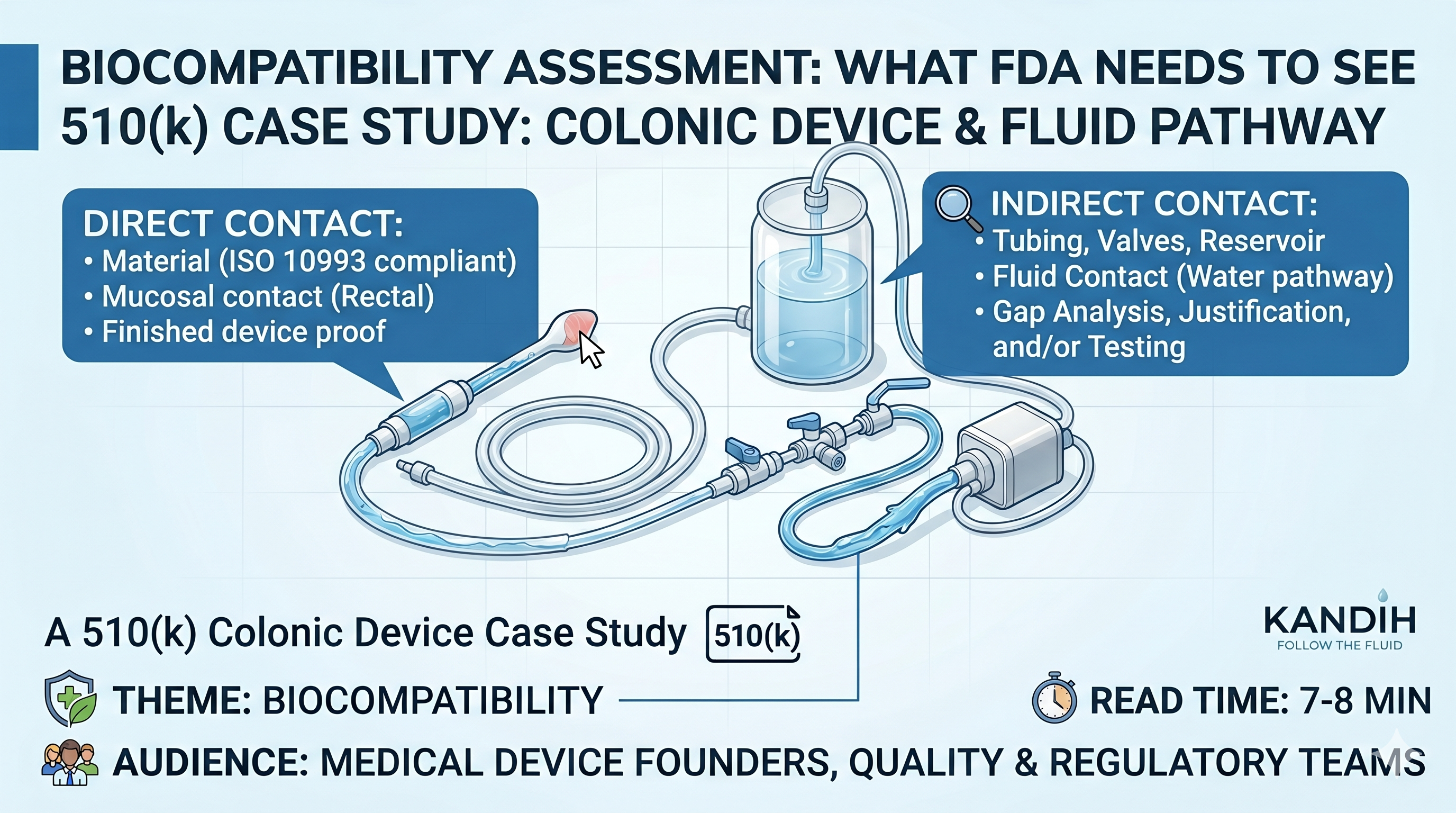
Theme: Biocompatibility Audience: Medical device founders, quality teams, regulatory teams, importers, and anyone trying to understand what FDA may ask in a 510(k) Estimated read time: 7–8 minutes Medical Devices: Irrigation […]