Medical Devices
January 30, 2026
Published by admin on January 30, 2026
Regulatory strategy should start the moment you decide to build a medical device—before you lock the intended use, user, setting, claims, or design inputs. Those early […]
October 24, 2025
Published by Harriet Kamendi PhD on October 24, 2025
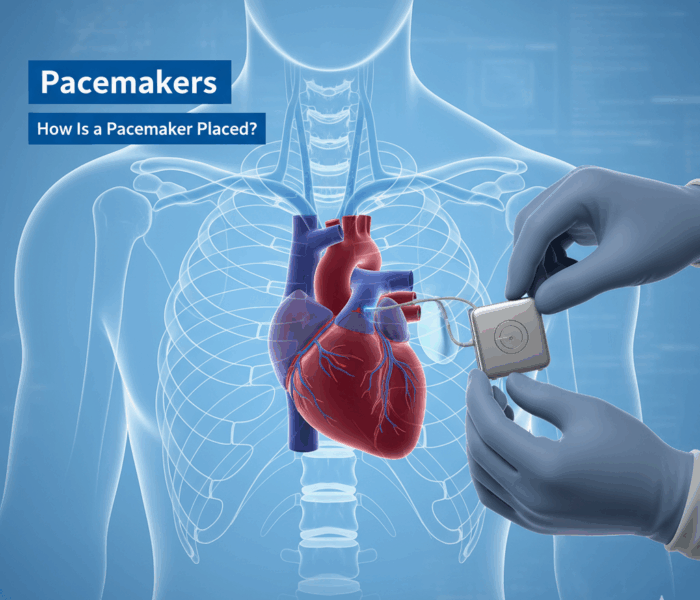
The procedure may be planned ahead of time, or it may be done during an emergency (temporary pacemaker). You will be given medicine to make parts of your body numb or make you sleep during the procedure. You may receive antibiotics to prevent infection and blood thinning medicine to prevent blood clots during the procedure. Different types of pacemakers require different procedures to place them.
August 11, 2025
Published by admin on August 11, 2025
Many medical device developers pass cytotoxicity, irritation, and sensitization testing—only to hit a wall at the implantation stage.
In fact, one of the most common reasons 510(k) submissions face delays is a poorly designed ISO 10993-6 implantation study.
July 3, 2025
Published by admin on July 3, 2025
When it comes to medical device testing, more isn’t always better. One of the biggest mistakes we see? Teams defaulting to comprehensive test panels — even […]