Pharmaceuticals
January 20, 2026
Published by admin on January 20, 2026
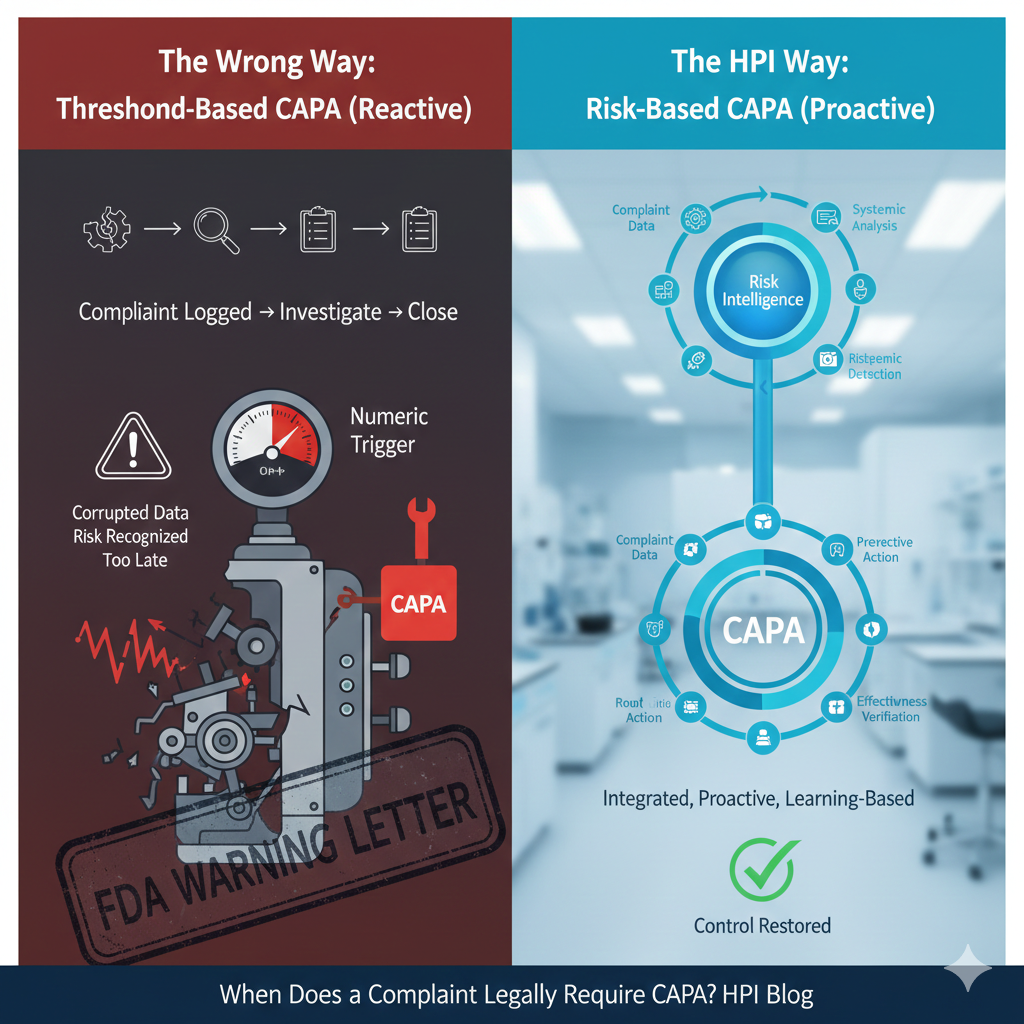
A complaint legally requires CAPA when it signals loss of control in the quality system—not when it hits a numeric or procedural threshold.From an FDA enforcement […]
December 8, 2025
Published by admin on December 8, 2025
Pharmaceuticals enter the environment via patient excretion, improper disposal, and manufacturing waste, prompting regulators worldwide to require environmental risk assessments (ERAs) as part of drug approvals. In an ERA, predicted environmental concentrations (PECs) of a drug are compared to ecotoxicity thresholds, often in a tiered fashion (Phase I screening followed by Phase II testing) [1]. This report analyzes ERA regulations and practice globally, from the EU and US to emerging regions in Asia, Latin America, the Middle East, and Africa. It integrates recent developments in China, India, Brazil, the Gulf Cooperation Council (GCC) countries, ASEAN nations, and African markets, alongside established frameworks (EU, US, Canada, Japan, Australia). Key findings include: