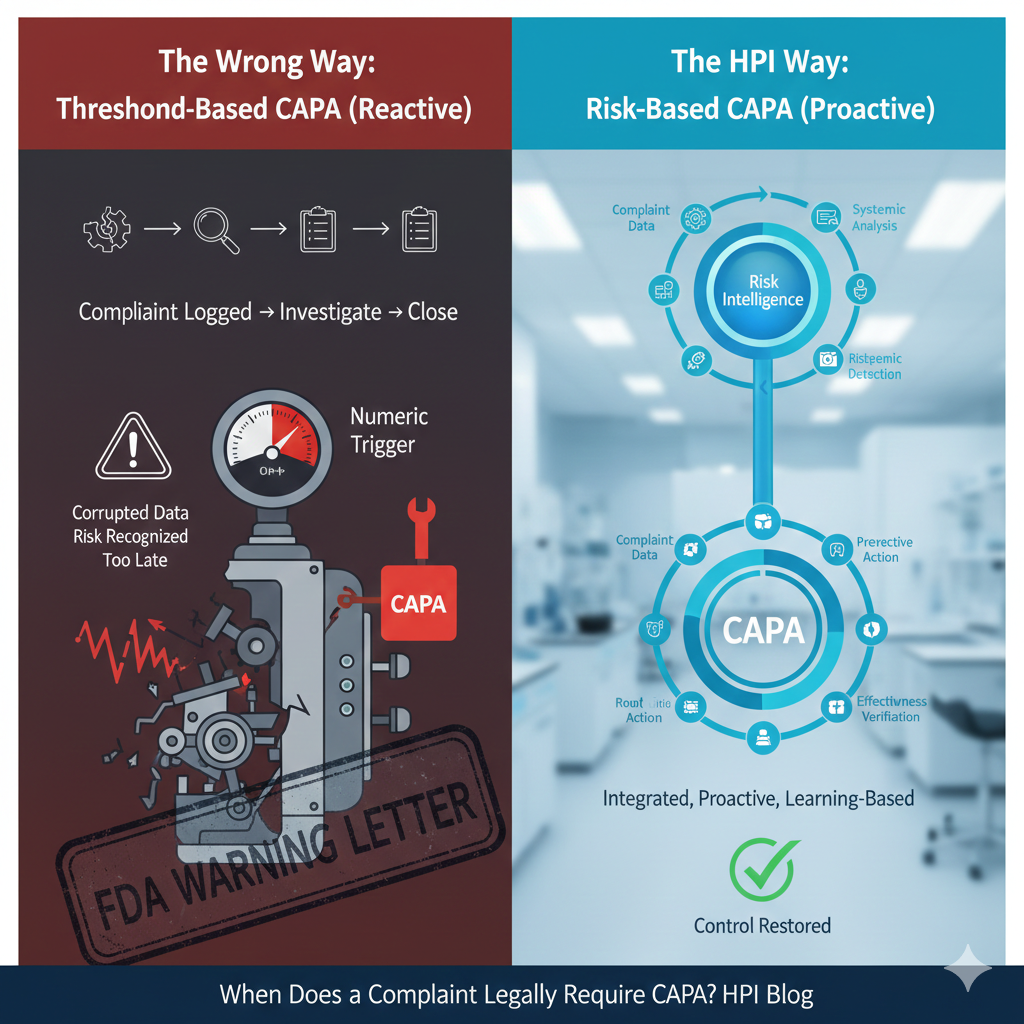
Environmental Risk Assessments (ERAs) for Pharmaceuticals: Global Regulatory Landscape and Implementation
Heart-Healthy Living – Get Your Blood Pressure and Cholesterol Checked
December 5, 2025
High Blood Pressure Medication Voluntarily Recalled — What Toxicologists Saw Coming
December 9, 2025Environmental Risk Assessments (ERAs) for Pharmaceuticals: Global Regulatory Landscape and Implementation
Executive Summary
Pharmaceuticals enter the environment via patient excretion, improper disposal, and manufacturing waste, prompting regulators worldwide to require environmental risk assessments (ERAs) as part of drug approvals. In an ERA, predicted environmental concentrations (PECs) of a drug are compared to ecotoxicity thresholds, often in a tiered fashion (Phase I screening followed by Phase II testing) [1]. This report analyzes ERA regulations and practice globally, from the EU and US to emerging regions in Asia, Latin America, the Middle East, and Africa. It integrates recent developments in China, India, Brazil, the Gulf Cooperation Council (GCC) countries, ASEAN nations, and African markets, alongside established frameworks (EU, US, Canada, Japan, Australia). Key findings include:
- Mandatory ERA frameworks: The EU has required ERAs for all new human medicines since 2006 (Directive 2001/83/EC, Article 8(3)) [2], and the US mandates an environmental review under NEPA (with most drugs eligible for categorical exclusion if PEC<1 μg/L )[3]. Canada’s current system is partial (new APIs trigger review under CEPA) but will soon shift ERA into its Food & Drugs Act [4]. Japan and Australia currently have no formal ERA requirements – Japan offers only basic guidance (2016) with no legal mandate [5], and Australia has no dedicated guideline (beyond general chemical regulations) [6].
- New jurisdictions: Many countries outside these regions do not yet mandate ERAs, but trends vary. For example, China’s Ministry of Ecology and Environment (MEE) has strengthened chemical substance reviews (Order No. 12 requires exposure assessments for new uses [7]), but China’s drug regulator (NMPA) currently has no dedicated ERA guideline for medicines. India similarly has no ERA mandate in its drug law, relying on general environmental regulations instead [8]. Brazil’s ANVISA also lacks specific ERA requirements for drugs. In contrast, GCC countries (e.g. Saudi Arabia) now explicitly require an environmental risk evaluation in their drug submission dossiers [9] – reflecting growing regional attention. ASEAN member states largely follow a common technical dossier (ACTD) format with no ERA section, and most African regulators focus on drug safety/efficacy with environment managed by separate laws [8].
- Implementation and harmonization: In practice, EU and US firms routinely include ERA data (hundreds of EU ERAs since 2006) [10], whereas very few ERA studies arise in regions without mandates. Post-market monitoring and older-product reviews are limited globally. Our expanded analysis highlights a patchwork landscape: regions like China, India, and Brazil currently rely on international reviews (EU/FDA) for implicit ERA considerations [8], while the GCC’s new requirement is a notable exception [9]. Strengthening global ERA practice will require international collaboration: harmonizing data requirements, sharing best-practices, and making ERA outcomes transparent can ensure that all new medicines are assessed for ecological impact.
The following sections detail these regulatory landscapes by region and summarize key similarities and differences. Tables (below) compare ERA mandates and triggers across jurisdictions. Updated conclusions consider how this broader global coverage affects policy recommendations for mitigating pharmaceutical pollution.
Global ERA Requirements: Where Are ERAs Mandatory?
Pharmaceutical ERA requirements vary greatly worldwide. Table 1 summarizes which jurisdictions mandate ERAs in drug approvals, their legal basis, and key notes on scope. The EU and US have formal systems (albeit with different approaches), Canada is transitioning to mandatory ERAs, and other regions range from no ERA laws (Japan, Australia) to emerging or partial regimes.
Table 1: ERA mandates for human pharmaceuticals by jurisdiction
| Jurisdiction | ERA Requirement | Legal Basis / Guideline | Notes |
| European Union (EU) | Yes (mandatory for all new marketing authorizations since 2006) | Directive 2001/83/EC Article 8(3); EMA ERA Guideline (2006, rev. 2024) | Every new drug (chemical or biologic, including generics) requires an ERA. Significant manufacturing changes (e.g. new indications) also trigger updated ERAs. Older (pre-2006) products had no prior requirement. |
| United States (US) | Yes (environmental review required; most drugs use categorical exclusions) | National Environmental Policy Act (NEPA) via FDA regulations (21 CFR Part 25); FDA Guidance 1998/2016 | NDAs/ANDAs/INDs must include an Environmental Assessment (EA) or claim a categorical exclusion. The FDA provides exclusions if predicted aquatic exposure <1 µg/L (1 ppb) or no expected use increase. Full EAs are required only in exceptional cases (e.g. very high-volume or environmentally sensitive drugs). |
| Canada | Partial (not yet integrated into drug approvals; new APIs reviewed under chemical laws) | Canadian Environmental Protection Act (CEPA) New Substances Notification Regs; forthcoming Food & Drugs Act ERA provisions (amended 2023) | New APIs not on the Domestic Substances List trigger a CEPA environmental review by ECCC. Health Canada is developing a unified ERA regime (post-2023) to embed ERAs in drug reviews (aligning with EU/US). Currently ~no formal ERA in drug approval. |
| Japan | No (not mandatory – guidance only) | MHLW 2016 ERA notice (basic) | Japan has no legal ERA requirement as of 2025. A 2016 MHLW guideline encouraged considering environmental impact for new drugs (referencing EU/US methods) but imposes no specific studies. Marketing authorizations are not contingent on ERA data, though stricter rules may emerge. |
| Australia | No (not specific) | None specific for medicines | The TGA has no dedicated ERA guideline for pharmaceuticals. Medicines are generally not subject to mandatory environmental assessment during approval (beyond general industrial/chemical regulations). Recent reports confirm “no specific guidelines” for pharma ERAs. |
| China | No (not specific) | – | China’s drug regulator (NMPA) currently does not mandate ERAs for new drugs. Environmental regulation focuses on industrial chemicals: e.g. MEE’s Order No. 12 (2020) requires exposure assessments for new uses of existing chemicals chemradar.com No dedicated guideline exists for drug ERAs, so pharmaceutical approvals lack a formal ERA process. |
| India | No (not specific) | – | India’s CDSCO has no specific ERA requirement in the Drugs & Cosmetics Act or registration guidelines. Environmental aspects of pharmaceuticals are governed by general laws (e.g. wastewater/waste disposal) and new drug rules do not request ERA data. No formal ERA is currently needed for drug approvals. |
| Brazil | No (not specific) | – | ANVISA’s regulations do not require ERAs as part of new drug submissions. Environmental oversight comes through manufacturing site permits and general environmental legislation. New marketing authorizations in Brazil are not contingent on ERA studies. |
| Gulf Cooperation Council (GCC) | Yes (evaluation required) | GCC Data Requirements (Saudi FDA 2025, Module 1.5) yumpu.com | Gulf countries (e.g. Saudi Arabia) require applicants to include an environmental risk evaluation in the dossier. The Saudi FDA’s 2025 guidelines explicitly state: “The applicant shall include an evaluation for any potential risks of the product to the environment” (use, storage, disposal) yumpu.com . Detailed ERA studies are not explicitly defined, but environmental impact must be addressed. |
| ASEAN (member countries) | No (not specific) | – | ASEAN nations follow a Common Technical Dossier (ACTD) for drug registration. The ACTD (modeled on ICH CTD) has no section for environmental data. Thus, no ASEAN-wide ERA mandate exists. Individual countries may consider environmental factors through other regulations, but ERA is not required under typical drug approval pathways. |
| Africa (various countries) | No (not specific) | – | Most African regulators (e.g. SAHPRA in South Africa, NAFDAC in Nigeria) do not require ERAs for human drugs. Drug approvals focus on quality, safety, and efficacy; environmental considerations fall under general EPA laws. Many African agencies rely on WHO or reference agency approvals (implicitly carrying their ERA requirements) rather than conducting local ERAs. |
Table 1. Which jurisdictions mandate ERAs for human pharmaceutical approvals? As of 2025, ERAs are obligatory in the EU and US (with different mechanisms), partially covered in Canada, and generally not required in other regions. Notably, recent GCC guidance is an exception, introducing an environmental risk section in submissions [9]. Animal drug ERAs are more harmonized globally (VICH), but human drug ERA rules remain a patchwork.
ERA Regulatory Frameworks in Key Regions
European Union (EU)
The EU has the most comprehensive ERA regime. Since October 2005 (centralized products) and 2006 (all new marketing authorizations), every new human medicine must include an ERA [2]. This is mandated by Directive 2001/83/EC, Article 8(3) [2]. The European Medicines Agency (EMA) provides tiered guidance (Phase I screening of PEC in surface water; Phase II tests if needed) to implement this. Any significant change (new indication, higher dosage, expanded patient population) that increases environmental exposure also triggers an ERA update [13]. Even generic drugs must include an ERA, typically by referencing existing data, with no waiver for generics. Older “grandfathered” products (approved pre-2006) had no ERA, though the EU’s pending revision may address such legacy cases. Overall, ERA data from EU submissions are compiled in public assessment reports (EPARs), providing transparency.
United States (US)
In the US, all new drug applications (NDAs, ANDAs, BLAs, INDs) require an environmental review under the National Environmental Policy Act (NEPA) via FDA regulations (21 CFR 25) [3]. Practically, however, the FDA introduced categorical exclusions in 1997, recognizing that most human drugs have negligible environmental impact. Sponsors must file either a full Environmental Assessment (EA) or claim a categorical exclusion for each application [14]. Key exclusions include cases where the projected environmental concentration is below 1 µg/L or where no usage increase is expected [14]. Thus annually thousands of drugs invoke an exclusion, and only a handful (∼0.05%) require a detailed EA [15]. When an EA is needed, it follows FDA’s 1998 guidance format (use, PEC via excretion, fate and ecotox data) [16]. If an EA identifies significant impact, an Environmental Impact Statement (EIS) could be required, though in practice this is extremely rare. The US approach aligns with EU goals but uses different triggers (a numerical threshold rather than PEC/PNEC) and philosophy (categorical exclusions) [17][18].
Canada
Canada has historically treated drug ERAs under its chemical safety law (CEPA). Currently, if a new drug’s active ingredient is not on Canada’s Domestic Substances List, Environment and Climate Change Canada reviews submitted ecotoxicity data under CEPA’s New Substances Notification regulations [4]. Meanwhile, Health Canada’s drug office did not itself require ERAs. However, this is changing: amendments to the Food and Drugs Act (2023) will create a dedicated ERA and risk management regime for pharmaceuticals. Once in force, Health Canada aims for “single-window” evaluations where ERAs are integrated into drug approvals (rather than handled separately under CEPA) [12]. Guidance is being developed, aligning Canada with EU/US norms. Until then, Canada’s practice is incomplete: many “new” drugs (using existing chemicals on the DSL) have slipped through with no assessment, prompting regulatory reform.
Other Jurisdictions (Japan, Australia, etc.)
Outside the EU, US, and Canada, most countries lack formal ERA mandates [8]. The report’s analysis highlights the following patterns:
- Japan: No ERA is required. In 2016, Japan’s Ministry of Health issued a very basic notice urging firms to consider environmental risks for new drugs, but it contains no study requirements and defers to US/EU methods [5]. There is no legal obligation to submit ERA data, and current approvals are granted without environmental scrutiny. As awareness grows, Japan may follow the EU’s example, but as of 2025 no change has been implemented.
- Australia: The Therapeutic Goods Administration (TGA) has no specific ERA guideline for human medicines [6]. Pharmaceutical approvals focus on safety and efficacy, with no mandatory environmental assessment. In practice, environmental issues (if any) are handled under broader chemical or environmental laws (e.g. industrial chemicals regulations), but these rarely apply to typical drugs. Literature reviews note that “Australia had no specific guidelines for ERA of pharmaceuticals” as of 2020. Given OECD and UN attention to pharma pollution, Australia may consider adding ERA rules in the future, but none exist yet.
- China: The Chinese NMPA currently does not require ERAs in drug applications. Pharmaceutical environmental protection is indirectly covered by general laws: for example, the Ministry of Ecology’s recent “Order No. 12” requires an environmental exposure assessment when the use of an existing chemical is expanded into new industrial applications [7]. However, this rule targets industrial chemicals and does not extend to routine pharmaceutical registration. In other words, China’s chemical substance management (MEE) is tightening, but there is no parallel guideline for ERA of new medicines. Thus Chinese drug approvals proceed without any ERA, though authorities are increasingly aware of pharmaceutical pollutants in water and could update rules in the future.
- India: The Indian regulator (CDSCO) has no specific ERA requirement. India’s Drugs & Cosmetics Act and regulations do not address environmental risk. Instead, India relies on its environmental ministry’s general pollution rules (e.g. waste-water discharge standards) to control pharma manufacturing emissions. In 2022, CDSCO issued disposal guidelines for unused drugs, but prospective ERA for new drugs is absent. Public and industry attention to water quality is growing in India, yet no drug approval depends on an ERA. [8]
- Brazil: Brazil’s ANVISA likewise has no ERA requirement for human medicines. Brazilian marketing authorizations focus on clinical efficacy, safety, and quality. If anything, Brazil addresses pharmaceutical pollution through environmental licensing of manufacturing facilities, not through drug dossiers. ANVISA has not published guidelines requesting environmental fate/toxicity data for new drugs. Like many emerging markets, Brazil often accepts reference agency approvals (e.g. EU/FDA) to streamline review, indirectly inheriting their ERA standards without conducting its own assessments.
- GCC (Gulf States): In a notable departure, Gulf regulators have begun to embed environmental considerations. The Saudi FDA’s 2025 “Data Requirements” guideline explicitly includes Module 1.5: Environmental Risk Assessment [9]. It states: “The applicant shall include an evaluation for any potential risks of the product to the environment… risks arising from use, storage and disposal.” In practice, this means drug dossiers must discuss environmental impact in Module 1.6 (similar to the EU format). However, the guidance does not specify detailed study protocols or strict triggers; it appears to require qualitative risk discussion rather than mandatory testing. Other GCC countries (UAE, Qatar, etc.) follow harmonized guidances and likely have similar provisions. This GCC requirement (which is not a law but a regulatory guideline) means that, for the first time in the Middle East, environmental impact is a formal part of drug evaluation.
- ASEAN countries: ASEAN members (Brunei, Cambodia, Indonesia, Laos, Malaysia, Myanmar, Philippines, Singapore, Thailand, Vietnam) use an ASEAN Common Technical Dossier for drug registration. The ACTD parallels the ICH/CTD format (Quality, Preclinical, Clinical, and Administrative modules) and contains no separate module for environmental risk. Therefore, no ASEAN-wide mandate exists for ERAs, and individual ASEAN regulators (for example, Thai FDA or Indonesia’s BPOM) do not request environmental fate data as part of their review. Drugs approved via ASEAN reliance mechanisms may carry EU/EU ERA data from reference countries, but that is incidental rather than required by ASEAN law.
- Africa (various countries): No regional African body mandates ERAs for pharmaceuticals. National regulators (e.g. South Africa’s SAHPRA, Kenya’s Pharmacy and Poisons Board, etc.) focus on human safety and efficacy. Environmental oversight is addressed at the country level by environmental agencies (not health ministries), so a new drug is generally not subjected to an ERA. Some countries may encourage manufacturers to follow WHO recommendations (which do not include ERAs for drugs). In practice, many African countries register products based on WHO prequalification or approvals by EMA/FDA, effectively outsourcing environmental review to those agencies [8].
Together, these regional narratives paint a fragmented picture. Only the EU and US fully integrate ERAs into the drug approval process; Canada is joining them; Japan and Australia are “watching” with minimal guidance; and the rest of the world is mostly “off-stage” on ERAs[8][9]. The GCC is an exception by including environment in the dossier for all new drugs. Importantly, in regions without ERA mandates, pharmaceutical developers often still gather environmental data to support EU/US submissions, but local regulators typically do not enforce it. This creates a gap: a medicine could be rigorously assessed for environmental risk in one country yet escape scrutiny in another [8].
Conclusions and Implications
Our expanded review shows that environmental risk assessment for human drugs remains unevenly implemented worldwide. The EU’s pioneer mandate and the US’s NEPA-based system ensure most new pharmaceuticals in those markets are evaluated for ecological impact. Canada is poised to do the same. Elsewhere, formal ERA processes are largely absent, although awareness is growing: Chinese and Indian authorities have strengthened chemical safety laws [7] but have not yet imposed ERA requirements on medicines. Brazil and other Latin American regulators have not integrated ERAs into drug approval. In contrast, the Gulf region’s regulatory overhaul is noteworthy: Saudi Arabia (and by extension GCC) now requires environmental risk evaluations in every submission [9]. This trend suggests that middle-income and emerging economies are beginning to recognize pharmaceutical pollution as an issue, even if concrete regulations lag.
These findings reinforce that today’s global ERA landscape is a patchwork. Many countries without local mandates effectively rely on international reference approvals (EMA/FDA) as proxies for environmental review [8]. As a result, harmonized ERA standards could protect the environment not only in wealthy markets but also indirectly through global regulatory reliance. For example, transparency measures like EPAR-style public reporting could inspire other regulators to demand similar data.
- Implications: To truly mitigate pharmaceutical environmental risks, international cooperation is needed. Regulatory agencies and pharmaceutical companies should work together to harmonize ERA requirements across regions. This could involve:
- Adopting common guidance: Aligning on data formats and assessment triggers (e.g. ICH-style ERA guidelines for human drugs) would help countries implement ERAs consistently. (The veterinary ICH/VICH process shows this is feasible.)
- Technical capacity building: Many low-/middle-income regulators may need scientific support to interpret ERAs. Training and global databases (like EMA’s or OECD’s) can help.
- Regulatory reliance: Expanding formal reliance on ERA portions of foreign approvals, or requiring applicants to append EU/FDA ERA reports, can fill gaps where local rules are weak.
- Transparency and monitoring: Publishing ERA outcomes and implementing post-market monitoring (e.g. measuring drug residues) would “close the loop” as envisioned in OECD guidance.
A harmonized global framework – in which every new medicine’s environmental fate is as rigorously assessed as its clinical safety – remains the goal [19][20]. As more regions (e.g. GCC, China, others) engage with environmental risk, the pieces of this framework can come together. Ensuring that China, India, Brazil, ASEAN, Africa, and other regions align with ERA best practices will prevent “unseen” pollution. In the next decade, we expect regulators worldwide to increasingly treat environmental protection as part of public health. By integrating ERAs into international policy and sharing the burden of study generation, the pharmaceutical industry and regulators can reduce duplicative work and fill regulatory blind spots – striving to safeguard both human health and ecosystems globally [21][7][9].
- European Parliament and Council of the EU. (2001). Directive 2001/83/EC on the Community code relating to medicinal products for human use. Official Journal of the European Communities, L311, 67–128. Available at EUR-Lex: https://eur-lex.europa.eu/legal-content/
- European Medicines Agency. (2006, rev. 2024). Guideline on the environmental risk assessment of medicinal products for human use (EMEA/CHMP/SWP/4447/00). London: EMA. (Original guideline effective Dec 1, 2006; revised guideline in preparation 2024.) Retrieved from EMA: https://www.ema.europa.eu/en/environmental-risk-assessment-medicinal-products-human-use-scientific-guideline
- Food and Drug Administration. (2023). Title 21, Code of Federal Regulations, Part 25: Environmental Impact Considerations. Washington, DC: U.S. Government Publishing Office. (Original regulation issued 1977; current as of 2023). Available at eCFR: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-A/part-25
- Government of Canada. (1999). Canadian Environmental Protection Act, 1999 (S.C. 1999, c.33). Ottawa: Justice Canada. (Act assented September 14, 1999). Available at Justice Laws: https://laws-lois.justice.gc.ca/eng/acts/C-15.31
- Ministry of Health, Labour and Welfare (Japan). (2016). Guidance on the Environmental Risk Assessment in new pharmaceutical development. Tokyo: MHLW. (MHLW notification No. 0331-6, issued March 2016 – basic guideline encouraging consideration of environmental impact for new drugs.)
- Saudi Food and Drug Authority. (2011). Data Requirements for Human Drugs Submission (Version 1.0 – Content of the Dossier). Riyadh: SFDA Drug Sector. (Issued June 22, 2011; includes Section 1.5 “Environmental Risk Assessment” for non-GMO products.) Retrieved from SFDA: https://www.sfda.gov.sa/En/Drug/Topics/Regulations-–-Guidelines.htm (Archived document)
- Ministry of Ecology and Environment (China). (2020). Order No. 12 – Measures for Environmental Management Registration of New Chemical Substances. Beijing: MEE. (Promulgated April 29, 2020; effective Jan 1, 2021, replacing former MEP Order 7/2010.) [English summary available via Beveridge & Diamond bdlaw.com]
- ChemRadar News. (2022). Saudi FDA implements environmental risk assessment requirement in drug submissions. ChemRadar Regulatory News. Retrieved from ChemRadar: https://www.chemradar.com/en/news/detail/etd7zm2n5vy8
- Jose, J., Pinto, J. S., Kotian, B., Thomas, A. M., & Charyulu, R. N. (2020). Comparison of the regulatory outline of ecopharmacovigilance of pharmaceuticals in Europe, USA, Japan and Australia. Science of the Total Environment, 709, 134815. https://doi.org/10.1016/j.scitotenv.2019.134815
- Oldenkamp, R., Hamers, T., Wilkinson, J., Slootweg, J., & Posthuma, L. (2024). Regulatory risk assessment of pharmaceuticals in the environment: Current practice and future priorities. Environmental Toxicology and Chemistry, 43(3), 611–622. https://doi.org/10.1002/etc.5535
- European Federation of Pharmaceutical Industries and Associations (EFPIA), Association of the European Self-Care Industry (AESGP), & Medicines for Europe. (2023). Proactively managing the environmental risks associated with the patient use of human medicinal products: An extended Environmental Risk Assessment (eERA) proposal. Brussels: EFPIA/AESGP/Medicines for Europe. (White paper published Jan 20, 2023.) aesgp.eu
- DLRC Group. (2024, July 31). EU requirements for Environmental Risk Assessment (ERA): an overview. DLRC Regulatory Blog. (Summary of EMA’s 2024 revised ERA guideline and legal basis in Article 8(3) of Directive 2001/83/EC.) Retrieved from DLRC: https://www.dlrcgroup.com/eu-requirements-for-environmental-risk-assessment-era/ dlrcgroup.com
- U.S. Food and Drug Administration. (2025, January 7). Environmental Impact Review at CDER. FDA/CDER Regulatory Guidance. (Overview of NEPA requirements and categorical exclusions for drug applications fda.gov content current as of Jan 7, 2025.) Retrieved from FDA: https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cder/environmental-impact-review-cder
- Health Canada. (2025, July 2). Environmental regulatory initiative (Environmental risk management amendments to the Food and Drugs Act). Government of Canada – Drugs and Health Products Legislation/Guidelines. (Details Canada’s plan to integrate ERA into drug approvals, enabled by 2023 FDA amendments canada.ca.) Retrieved from Canada.ca: https://www.canada.ca/en/health-canada/services/drugs-health-products/legislation-guidelines/environmental-risk-management-amendments-food-drugs-act/environmental-regulatory-initiative.html
- Smithers Group. (2024). Pharmaceutical Environmental Risk Assessment – Updated requirements under revised EMA guideline. Smithers Regulatory Services. (Corporate webpage outlining the new 2024 EMA ERA guideline changes and Smithers’ ERA services smithers.com.) Retrieved from Smithers: https://www.smithers.com/industries/pharmaceutical/environmental-risk-assessment-eu-ema
- Food and Drug Administration. (2022, Feb 7). Agency Information Collection Activities; Submission for OMB Review; Comment Request – Environmental Impact Considerations. Federal Register, 87(25), 6872-6875 govinfo.gov. Washington, DC: Office of the Federal Register. (Docket No. FDA-2021-N-0405; FR Doc. 2022-02475.) Retrieved from GovInfo: https://www.govinfo.gov/content/pkg/FR-2022-02-07/pdf/2022-02475.pdf
- Bubela, T., Boch, R., & Viswanathan, S. (2019). Recommendations for regulating the environmental risk of shedding for gene therapy and oncolytic viruses in Canada. Frontiers in Medicine, 6, 58. https://doi.org/10.3389/fmed.2019.00058 (PubMed Central ID: PMC6447701)
- Norton Rose Fulbright. (2021, April 21). Proposed modernization of Canada’s environmental protection laws to have far-reaching effects for chemicals, drugs, and consumer products. Norton Rose Fulbright Publications – Thought Leadership. (Summary of Bill C-28 amendments to CEPA and Food & Drugs Act introducing a new ERA regime for drugs nortonrosefulbright.com. Retrieved from NRF: https://www.nortonrosefulbright.com/en-ca/knowledge/publications/a6d7fa9f/proposed-modernization-of-canadas-environmental-protection-laws
- Lee, D., & Choi, K. (2019). Comparison of regulatory frameworks of environmental risk assessments for human pharmaceuticals in EU, USA, and Canada. Science of the Total Environment, 681, 452–462. https://doi.org/10.1016/j.scitotenv.2019.03.372
- Zinken, J., Pasmooij, A. M. G., Ederveen, A. G. H., Hoekman, J., & Broekmans, Y. (2024). Environmental risk assessment in the EU regulation of medicines for human use: an analysis of stakeholder perspectives on its current and future role. Drug Discovery Today, 29(12), 104213. https://doi.org/10.1016/j.drudis.2024.104213
- Hochmuth, D., et al. (2024). Prioritising non-animal methods to improve environmental risk assessment for pharmaceuticals and promote innovation in the EU. Poster presentation at the EUSAAT 2024 Congress (European Society for Alternatives to Animal Testing), Linz, Austria, Aug 2024. Retrieved from PETA Science Consortium Int.: https://www.thepsci.eu/wp-content/uploads/2024/09/2024-EUSAAT_Hochmuth-et-al_EMA-ERA-guidance.pdf