When Does a Complaint Legally Require CAPA?

5 Complaint Handling Errors That Kill CAPAs
January 19, 2026
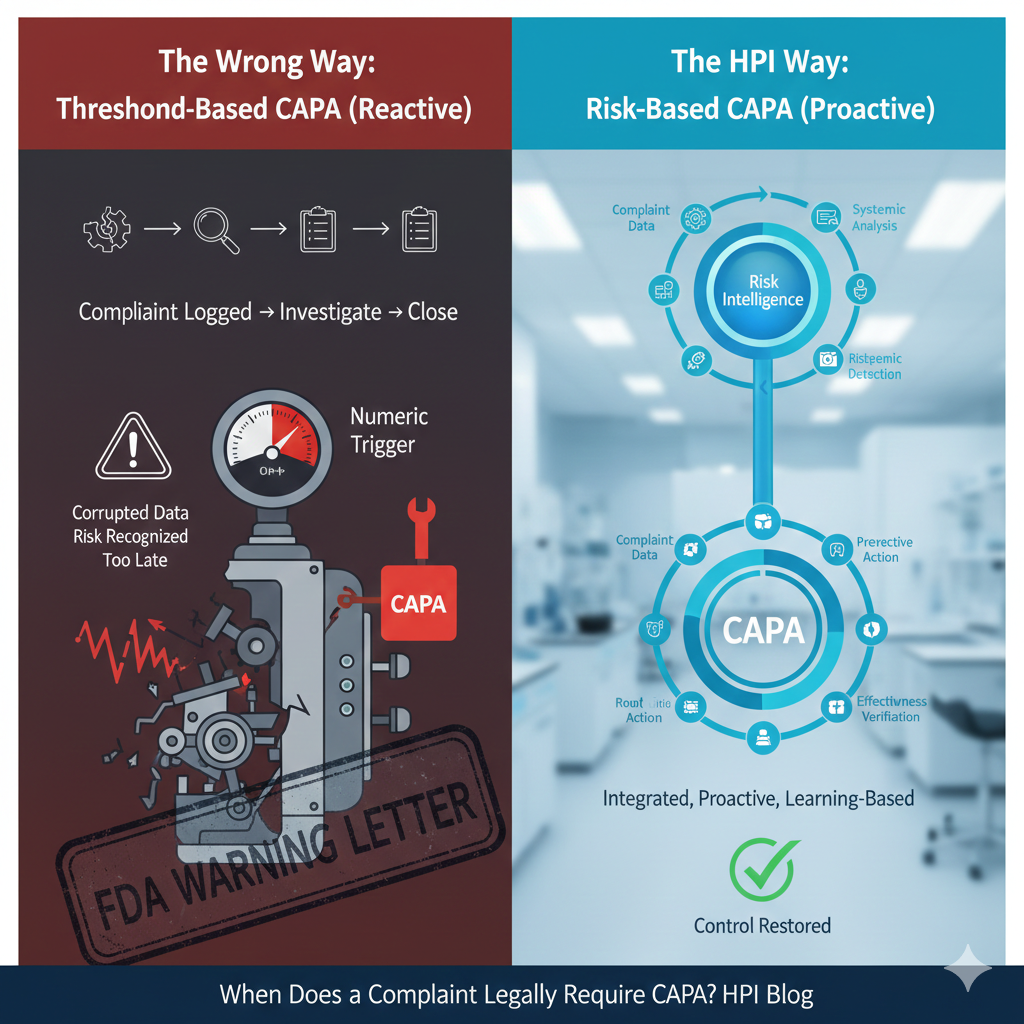
Complaint → Weak Root Cause → Repeat 483
January 21, 2026A complaint legally requires CAPA when it signals loss of control in the quality system—not when it hits a numeric or procedural threshold.
From an FDA enforcement perspective, CAPA is mandatory once complaint data indicates systemic risk, degraded control, or credible recurrence. Waiting for a regulation or SOP trigger means the system recognized risk too late.
Why Waiting for a Regulation Means You’re Already Late
If you’re waiting for a regulation to explicitly tell you when to open CAPA, the system has already failed.
The U.S. Food and Drug Administration does not enforce CAPA as a checkbox exercise. FDA enforces CAPA as a risk-control obligation.
Key distinction:
FDA does not ask, “Did you follow your SOP?”
FDA asks, “Did your system recognize risk and respond proportionately?”
That distinction is where many quality systems quietly break.
The Assumption That Breaks Systems
Common belief:
“A complaint requires CAPA only if it meets a specific regulatory threshold.”
FDA reality:
CAPA is required when a complaint reveals a failure of control, not when a form meets a preset trigger.
CAPA is not a regulatory event.
It is the mechanism by which the quality system restores control.
The FDA Systems Perspective (Not the Procedural One)
Complaints are not records.
They are inputs into a risk-control feedback loop.
From an FDA inspection standpoint, the obligation to initiate CAPA emerges when a complaint—or a pattern of complaints—does one or more of the following:
Signals systemic failure, not an isolated anomaly
Indicates loss of process, design, or detection control
Shows recurrence, severity escalation, or unstable trends
Reveals a gap between intended and actual performance
At that point, CAPA is not optional.
Failure to initiate CAPA is interpreted as failure to recognize or act on risk, not as a documentation oversight.
What FDA Inspectors Actually Evaluate
Inspectors do not look for a sentence that says “this complaint equals CAPA.”
They evaluate whether your decision logic is coherent, consistent, and risk-based.
Specifically, FDA looks for evidence that:
Complaint data is actively trended, not passively logged
CAPA triggers are tied to risk signals, not convenience or volume
Similar complaints lead to similar escalation decisions
The absence of CAPA can be defended with documented risk rationale, not opinion
If two comparable complaints lead to different outcomes without a clear, risk-based explanation, FDA reads that as loss of control.
Where Organizations Get This Wrong (Systemically)
1. Treating CAPA as a Regulatory Event
CAPA is opened only after a threshold is crossed.
FDA view:
CAPA is a control response, not a reaction to regulatory pressure.
2. Confusing Administrative Closure With Regulatory Effectiveness
Closing a complaint investigation is treated as “problem solved.”
FDA cares whether:
The failure mode was addressed
Recurrence likelihood was reduced
The system demonstrated learning over time
If complaints recur and CAPA was never initiated, FDA assumes the system failed to interpret the signal.
3. Using Subjective or Inconsistent CAPA Triggers
CAPA initiation depends on who reviewed the complaint, workload, or discomfort with escalation.
FDA interpretation:
This is a governance failure, not flexibility.
The Regulatory Insight (Say This Out Loud)
A complaint legally requires CAPA when it reveals that the quality system is no longer reliably controlling risk.
Not when:
A number is reached
A box is checked
An auditor suggests it
But when:
Control has degraded
Risk has become systemic
Detection has failed
Recurrence is plausible
That is the enforcement logic FDA applies—whether or not it appears verbatim in your SOP.
The Design Principle (Your One Takeaway)
Design CAPA initiation as a risk-interpretation decision, not a complaint-count rule.
Your system must be able to explain, consistently and with evidence:
Why this complaint did not require CAPA, and
How you know control was still intact
If it cannot, CAPA was likely required—whether you opened it or not.
FAQ
Q: Is CAPA legally required for every complaint?
A: No. CAPA is required when complaint data indicates systemic risk or loss of control, not for every individual complaint.
Q: Does FDA require a specific trigger for CAPA?
A: No. FDA evaluates whether CAPA initiation decisions are risk-based, consistent, and defensible.
Q: Can a closed complaint still require CAPA?
A: Yes. Complaint closure does not equal restored control. Trends and recurrence determine CAPA necessity.
Q: What happens if similar complaints don’t trigger CAPA?
A: FDA interprets inconsistent escalation as loss of quality system control.
If your team cannot consistently articulate why CAPA was or was not initiated—using the same risk logic across complaints—your system is relying on discretion, not design.
That is exactly where CAPA remediation, diagnostic audits, and system-level redesign create regulatory predictability.
If your CAPA system can’t explain itself to an inspector, it’s not designed yet.
References:
21 CFR §820.100 – Corrective and Preventive Action (CAPA)
Establishes CAPA as a system for analyzing quality data, identifying causes, implementing corrective/preventive actions, and verifying effectiveness—not a checklist trigger.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-820/subpart-J/section-820.100
21 CFR §820.198 – Complaint Files
Requires complaint evaluation to determine the need for investigation and corrective action, explicitly linking complaints to systemic quality response.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-820/subpart-M/section-820.198
21 CFR §211.198 – Complaint Files (CGMP)
Requires investigation of complaints and mandates follow-up when complaints indicate potential quality failures—often enforced through CAPA expectations even when not named explicitly.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-C/part-211/subpart-J/section-211.198
21 CFR §211.192 – Production Record Review / Investigations
Forms the legal backbone for FDA’s expectation that recurring complaints or deviations trigger systemic investigation and corrective action.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-C/part-211/subpart-J/section-211.192
FDA Compliance Program Guidance Manual (CPGM) 7346.832 – QSIT
Describes how FDA inspectors trace complaints, deviations, and failures through CAPA to assess system effectiveness and management control.
https://www.fda.gov/media/75506/download
FDA Guidance: Quality Systems Approach to Pharmaceutical CGMP Regulations
Explicitly frames CAPA as part of an integrated quality system tied to management responsibility and risk control.
ICH Q10 – Pharmaceutical Quality System
Anchors CAPA initiation to detection of systemic risk, management oversight, and maintenance of a state of control.
https://database.ich.org/sites/default/files/Q10%20Guideline.pdf
ICH Q9(R1) – Quality Risk Management
Defines risk-based decision-making principles FDA uses implicitly when evaluating CAPA initiation and complaint escalation logic.
https://www.fda.gov/media/177720/download
ISO 13485:2016 – Medical Devices QMS
Links complaint handling, risk management, and CAPA effectiveness across the product lifecycle (frequently referenced during inspections).