Uncategorized
March 24, 2026
Published by admin on March 24, 2026
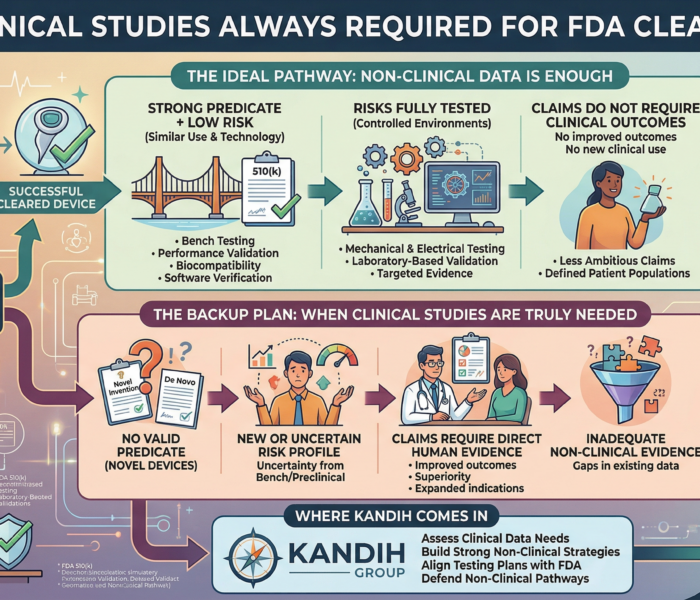
A founder once delayed their entire product launch by 18 months—planning a clinical trial they never actually needed. The device worked. The science was solid. The team had funding. What they didn’t have was clarity. So they defaulted to the safest assumption:
“FDA will probably require a clinical study.”
March 23, 2026
Published by admin on March 23, 2026
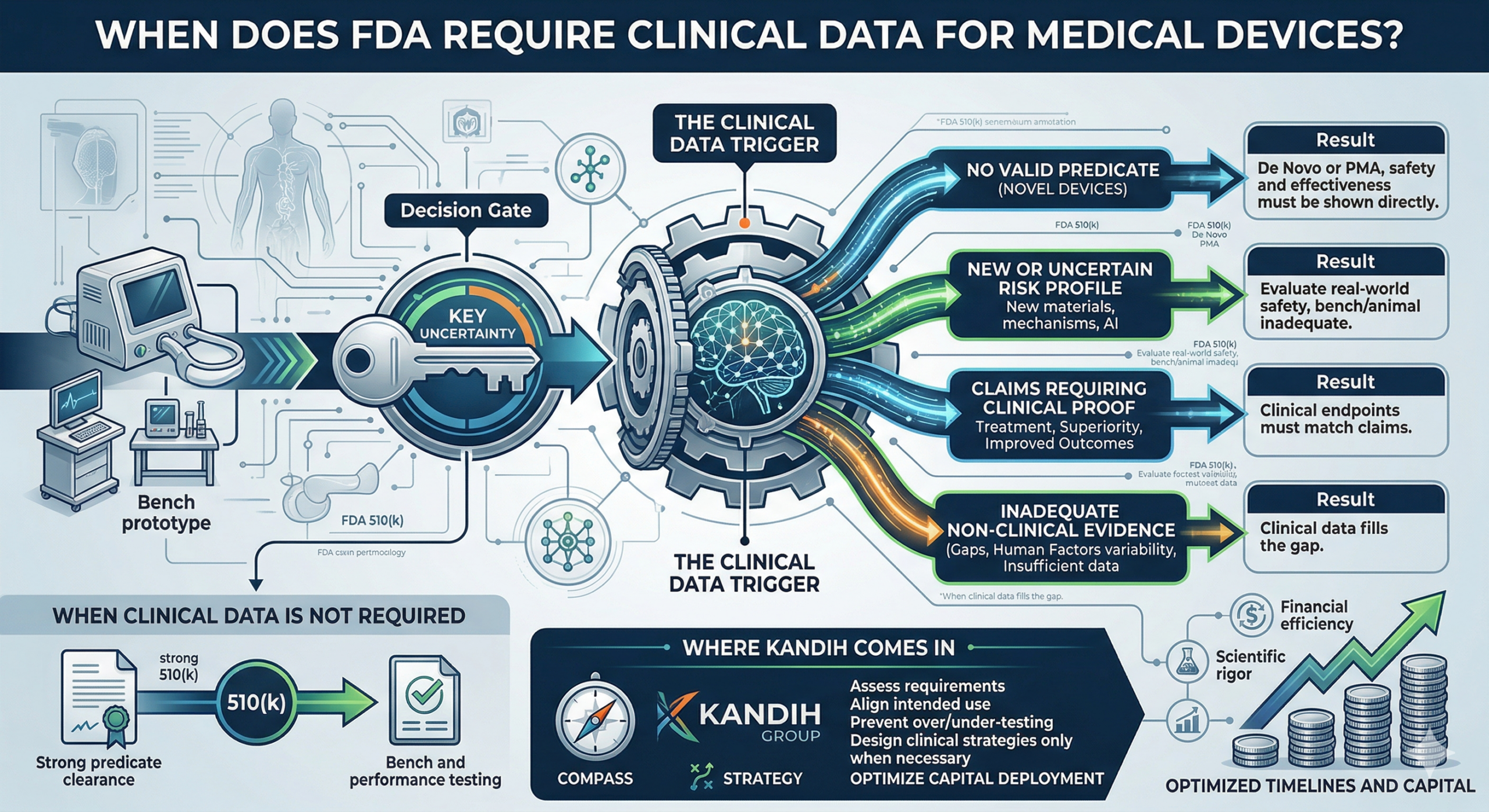
Short answer: the FDA requires clinical data when risk, uncertainty, or lack of precedent cannot be addressed through non-clinical testing. Clinical trials are not automatic.
They are triggered. And most founders misunderstand when that trigger actually happens. The Story Most Founders Live Through A founder builds a promising device.
The prototype works. Bench testing looks strong. Early feedback is positive
March 17, 2026
Published by admin on March 17, 2026
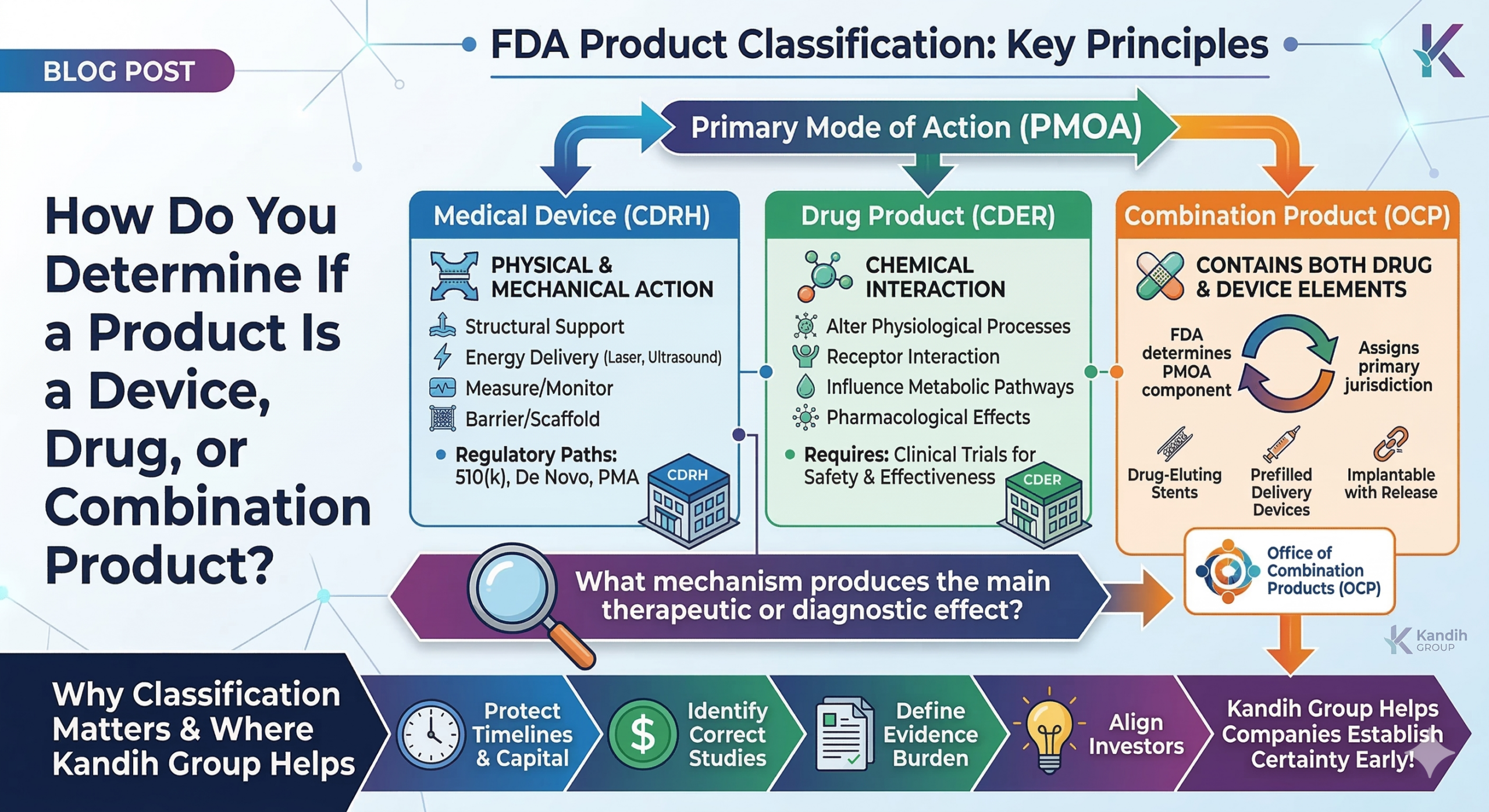
Short answer: the classification depends on the product’s primary mode of action (PMOA)—how the product achieves its main intended effect in the body. This determination matters because it decides which center of the U.S. Food and Drug Administration will regulate the product and what type of evidence will be required.
March 11, 2026
Published by admin on March 11, 2026
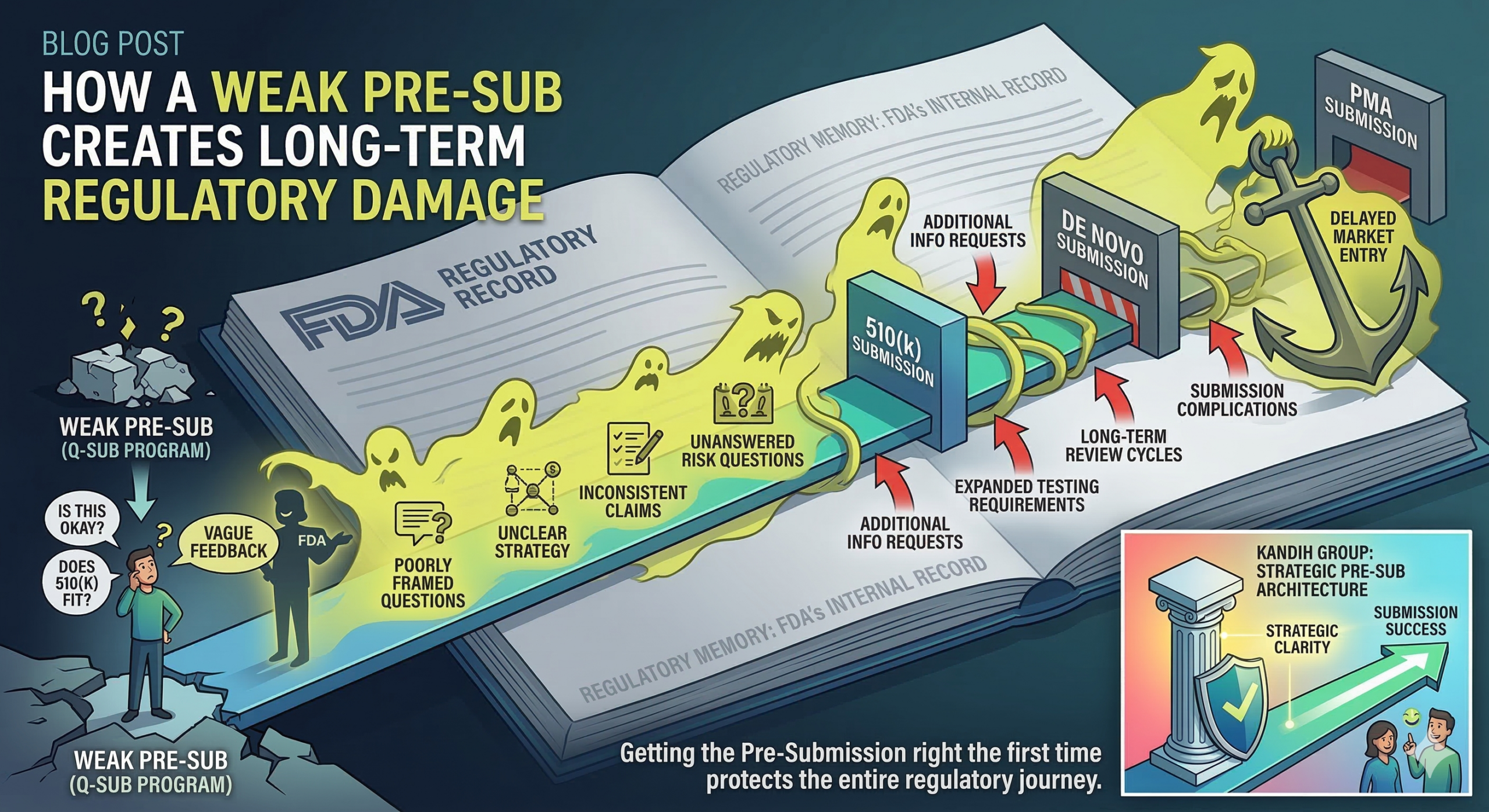
Short answer: a weak Pre-Submission (Pre-Sub) does not just waste a meeting with the FDA. It creates a regulatory record that can follow your device through the rest of development. Many founders treat a Pre-Sub as a casual conversation with the U.S. Food and Drug Administration. In reality, it is a formal interaction within the FDA’s Q-Submission program. The discussion becomes part of the agency’s internal regulatory history for your device.