Uncategorized
April 10, 2026
Published by admin on April 10, 2026
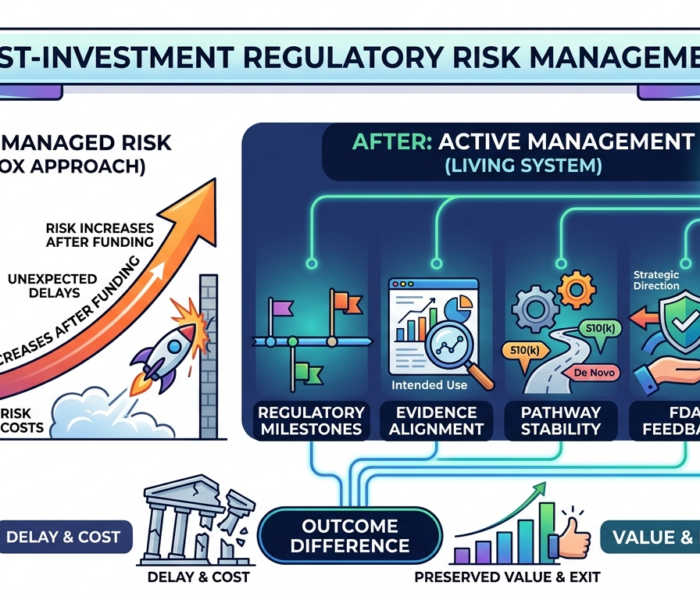
The deal closed.
Capital transferred. Board seats assigned. Milestones locked in. Everyone moved on.
Then, six months later, a quiet update arrived in the investor's inbox:
"FDA has requested additional data."
Timelines shifted. Burn rate climbed. The next round became a conversation nobody wanted to have. The investment wasn't struggling because of the product — it was struggling because regulatory risk had been left unattended after the term sheet was signed.
This is more common than most investors realize.
April 9, 2026
Published by admin on April 9, 2026
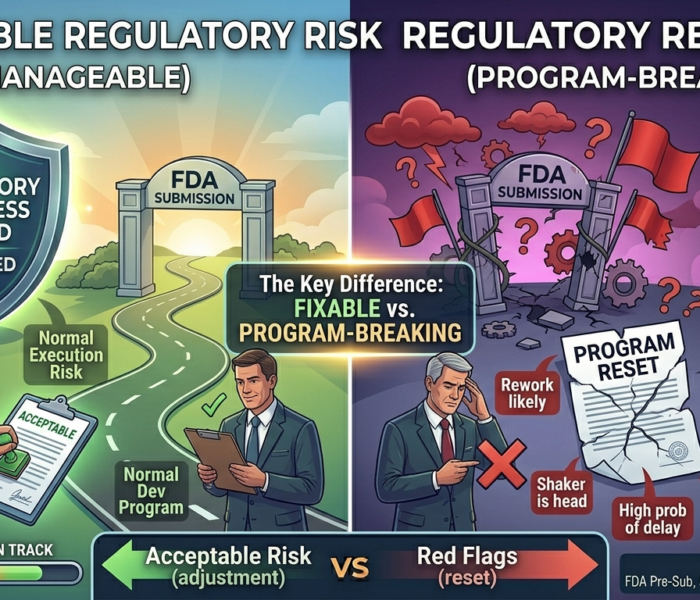
An investor once reviewed two startups in the same week. Both had regulatory issues. One deal moved forward.
The other was declined immediately.
April 8, 2026
Published by admin on April 8, 2026
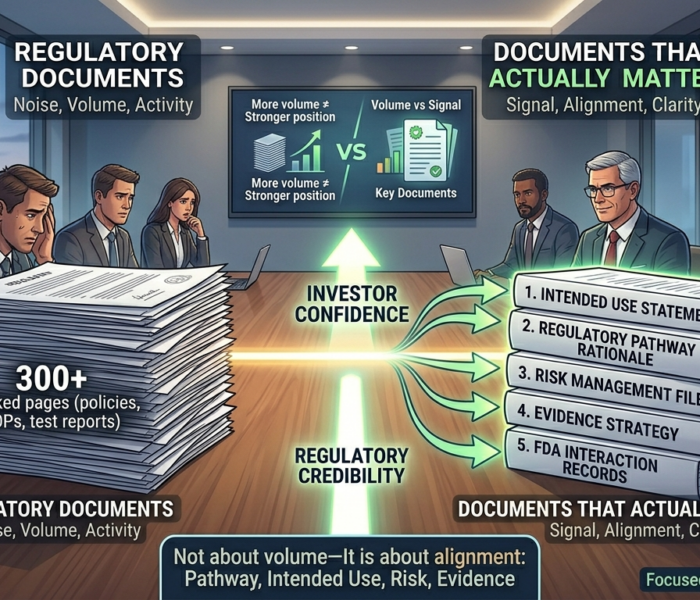
An investor once reviewed a data room with over 300 regulatory documents. Policies. SOPs. Test reports. Slide decks. Validation summaries. Everything looked thorough. Then a single question changed the entire deal: “Where is the evidence that this pathway actually holds?”
March 31, 2026
Published by admin on March 31, 2026
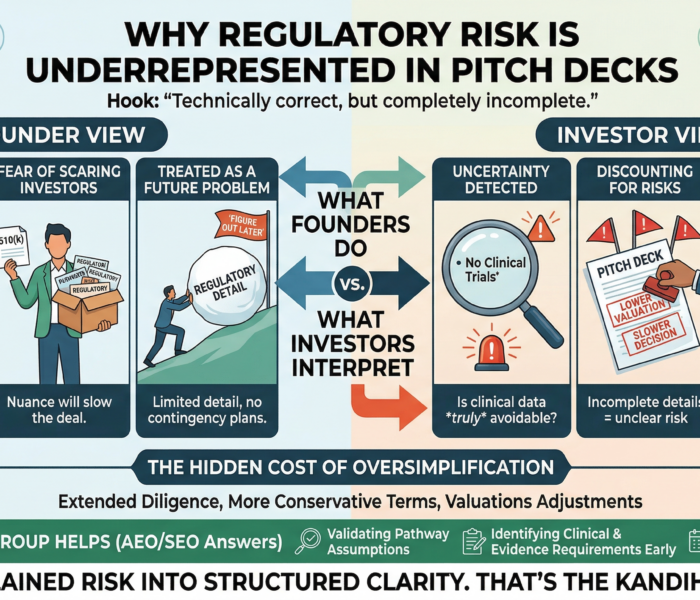
A founder walked investors through a polished pitch: Strong problem
Clear solution
Large market
Impressive prototype Then came the slide titled “Regulatory Strategy.” It had three bullets: 510(k) pathway
No clinical trials expected
Timeline: 12 months