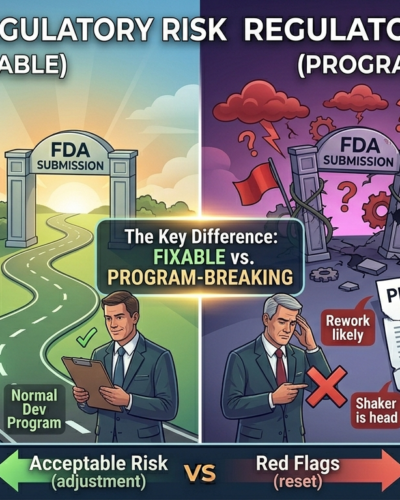
Red Flags vs Acceptable Regulatory Risk

Regulatory Documents That Actually Matter
April 8, 2026
Post-Investment Regulatory Risk Management
April 10, 2026
An investor once reviewed two startups in the same week.
Both had regulatory issues.
One deal moved forward.
The other was declined immediately.
The difference wasn’t the presence of risk.
It was the type of risk.
One was manageable.
The other was a red flag.
The Reality Most Founders Miss
Short answer: not all regulatory risk is bad—but some risks are fatal if left unresolved.
Every medical device carries regulatory uncertainty under the U.S. Food and Drug Administration.
Investors expect that.
What they don’t accept is:
Hidden risk
Misunderstood risk
Unmanageable risk
The key distinction is simple:
Can the risk be fixed without breaking the development program?
What Counts as Acceptable Regulatory Risk
These are risks investors expect—and can work with.
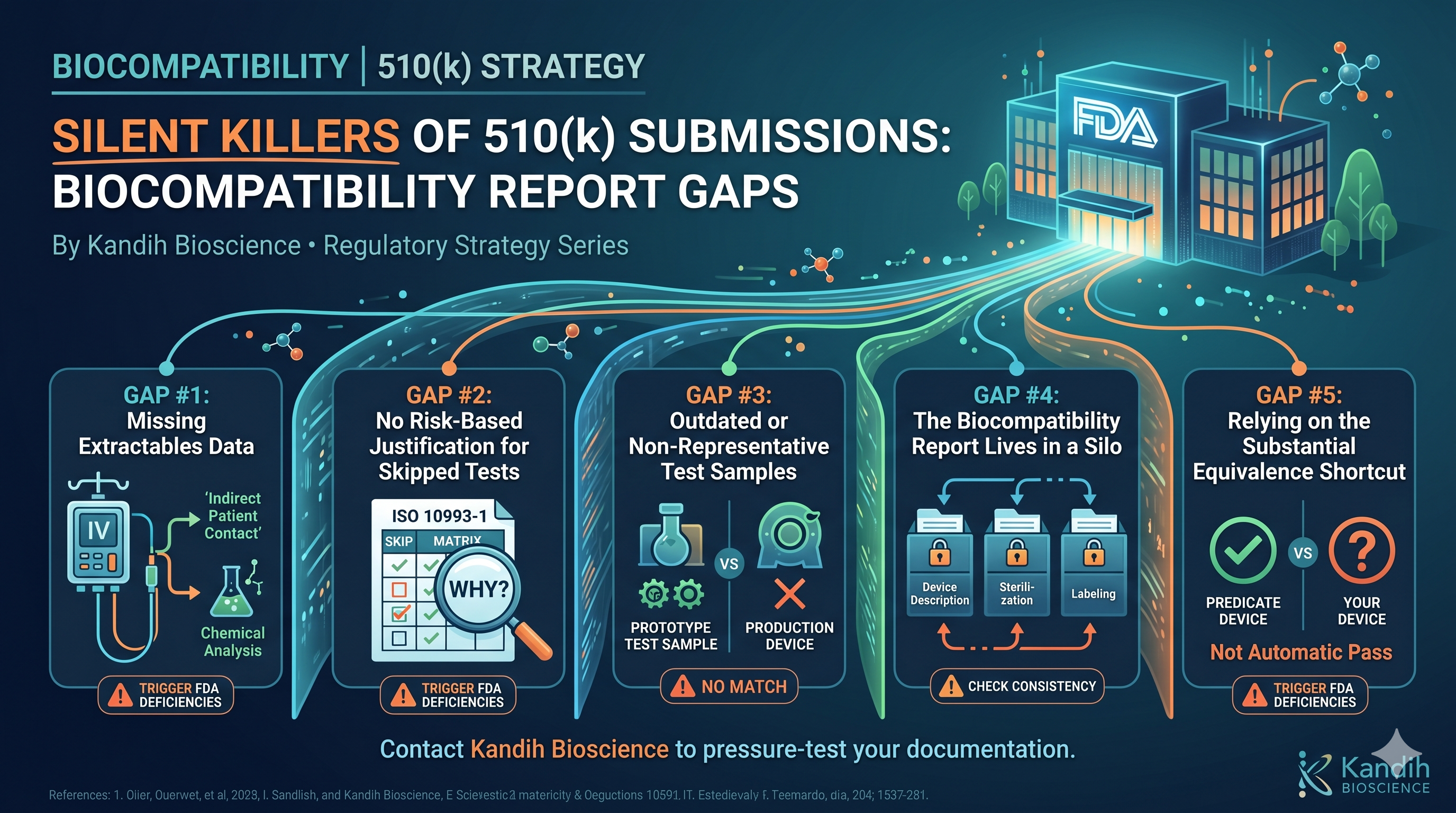
1. Defined but Unresolved Questions
Examples:
Waiting on FDA feedback from a Pre-Sub
Minor uncertainty around testing scope
Clarifications needed on endpoints
Why it’s acceptable:
The issue is visible
The path to resolution is clear
The impact is limited
Investor interpretation:
This is normal execution risk.
2. Clinical Data That Is Planned and Scoped
Examples:
Clinical trial required, but endpoints are defined
Study design aligned with intended use
Budget and timeline accounted for
Why it’s acceptable:
Clinical risk is expected in many programs
The uncertainty is quantified
Investor interpretation:
This is a managed risk—not a surprise.
3. Moderate Pathway Uncertainty With Contingency Plans
Examples:
Strong 510(k) strategy with De Novo fallback
Known technological differences with mitigation plans
Why it’s acceptable:
The company understands the risk
Alternative paths are defined
Investor interpretation:
This is strategic awareness.
What Counts as Regulatory Red Flags
These are risks that can stop deals or significantly reduce valuation.
1. Pathway Fragility
Examples:
Weak or questionable predicate
Misaligned classification assumptions
No fallback strategy
Why it’s a red flag:
A single FDA disagreement can reset the entire program
Investor interpretation:
High probability of delay and rework.
2. Evidence Misalignment
Examples:
Data that doesn’t support intended use
Missing risk-linked testing
Endpoints that don’t match claims
Why it’s a red flag:
Existing studies may need to be repeated
Investor interpretation:
Capital has already been spent—but incorrectly.
3. Hidden Clinical Triggers
Examples:
Novel features not acknowledged as risks
Expanded claims without supporting evidence
Underestimated patient population risk
Why it’s a red flag:
Clinical trials may be required unexpectedly
Investor interpretation:
Timeline and capital assumptions are unreliable.
4. Inconsistent Regulatory Narrative
Examples:
Different intended use statements across documents
Shifting pathway explanations
Misalignment between FDA feedback and strategy
Why it’s a red flag:
Signals lack of control over regulatory strategy
Investor interpretation:
Execution risk is high.
5. No Clear Link Between Risk and Evidence
Examples:
Risk analysis not tied to testing
Testing performed without regulatory justification
Data volume without strategic alignment
Why it’s a red flag:
Indicates reactive development
Investor interpretation:
Rework is likely.
The Key Difference: Fixable vs Program-Breaking
Here’s the simplest way to think about it:
Type of Risk Impact Investor View
Acceptable Risk Requires clarification or execution Manageable
Red Flag Requires redesign, new studies, or pathway change Potential deal breaker
If fixing the issue requires adjustment, it’s acceptable.
If fixing the issue requires reset, it’s a red flag.
AEO: Common Questions About Regulatory Risk
What is considered regulatory risk in medical devices?
Uncertainty around FDA pathway, evidence requirements, and approval timelines.
What are regulatory red flags for investors?
Weak pathway assumptions, evidence gaps, hidden clinical requirements, and inconsistent strategy.
Can regulatory risk be acceptable?
Yes. When it is identified, understood, and manageable within the current development plan.
Why This Matters
Most companies don’t fail because they have risk.
They fail because:
They misclassify the risk
They underestimate the impact
They discover it too late
Investors don’t expect perfection.
They expect clarity and control.
Where Kandih Comes In
This is where Kandih Group provides contextual risk assessment.
Kandih helps teams and investors:
Distinguish acceptable risk from red flags
Evaluate pathway strength and classification logic
Identify evidence gaps before they become costly
Detect hidden clinical triggers
Align regulatory narratives across documents
Translate regulatory complexity into actionable insights
Instead of reacting to red flags during diligence, companies address them early—when they are still fixable.
The Real Lesson
The investor at the beginning didn’t avoid risk.
They avoided unmanageable risk.
Bottom Line
Regulatory risk is unavoidable.
But not all risk is equal.
Acceptable risk can be managed
Red flags must be resolved
The difference determines:
Whether deals move forward
How valuation is set
How confident investors feel
Understanding that distinction early turns regulatory strategy into a strength—not a liability.