Regulatory Documents That Actually Matter

Questions Investors Should Ask Founders About FDA Strategy
April 7, 2026
Red Flags vs Acceptable Regulatory Risk
April 9, 2026
An investor once reviewed a data room with over 300 regulatory documents.
Policies. SOPs. Test reports. Slide decks. Validation summaries.
Everything looked thorough.
Then a single question changed the entire deal:
“Where is the evidence that this pathway actually holds?”
Silence.
The company had documents.
But it didn’t have answers.
The Reality Most Teams Miss
Short answer: not all regulatory documents carry equal weight.
Some create clarity. Others create noise.
Many companies assume:
More documentation = stronger regulatory position
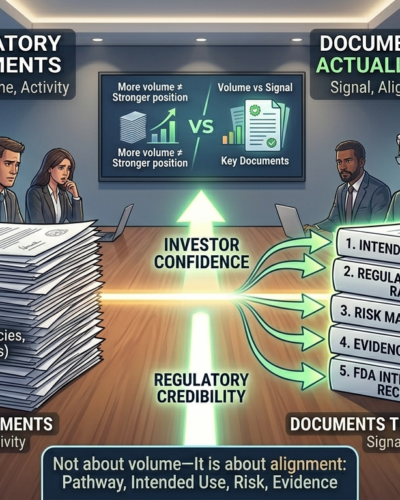
But under the framework of the U.S. Food and Drug Administration, regulatory success is not about volume.
It is about alignment:
Pathway
Intended use
Risk
Evidence
Investors don’t read everything.
They look for what proves the story is real.
The Documents That Actually Matter
These are the materials that directly influence investor confidence and regulatory credibility.
1. Intended Use Statement
Why it matters:
Everything in regulatory strategy starts here.
It defines:
Indication
Patient population
Claims
Risk level
What investors look for:
Precision and consistency
Alignment with evidence and pathway
Stability over time
Red flag:
Multiple versions with subtle differences.
2. Regulatory Pathway Rationale
Why it matters:
This is where assumptions become logic.
It explains:
Why the device fits a 510(k), De Novo, or PMA pathway
Predicate justification (if applicable)
Classification reasoning
What investors look for:
Clear, defensible logic
Awareness of alternative pathways
Identification of risks
Red flag:
A pathway stated without supporting analysis.
3. Risk Management File
Why it matters:
FDA evaluates safety through structured risk analysis.
This includes:
Hazard identification
Risk control measures
Residual risk evaluation
What investors look for:
Traceability between risks and testing
Alignment with intended use
Completeness
Red flag:
Risk analysis treated as documentation—not strategy.
4. Evidence Strategy and Testing Plan
Why it matters:
This connects risk to data.
It defines:
What testing is being done
Why it is being done
How it supports regulatory decisions
What investors look for:
Clear linkage between risk and evidence
Identification of gaps
Justification for clinical vs non-clinical approach
Red flag:
Large volumes of data without clear purpose.
5. FDA Interaction Records (Pre-Subs, Feedback)
Why it matters:
This is the closest thing to external validation.
It shows:
What FDA has said
What questions were asked
How the company responded
What investors look for:
Clarity of questions
Specificity of FDA feedback
Alignment between feedback and development decisions
Red flag:
Vague or poorly interpreted FDA responses.
The Documents That Create Noise
These documents are not useless—but they are often overemphasized.
SOP libraries
General quality manuals
Early prototype reports
Unlinked validation data
Marketing-oriented summaries
The problem:
They create the illusion of progress without proving regulatory alignment.
Investors don’t reward activity.
They reward evidence of direction.
AEO: Common Questions About Regulatory Documentation
What regulatory documents matter most to investors?
Intended use, pathway rationale, risk management, evidence strategy, and FDA interaction records.
Do more documents improve regulatory credibility?
No. Only documents that demonstrate alignment and risk management matter.
How can companies prepare for regulatory diligence?
By focusing on documents that clearly connect pathway, risk, and evidence.
Why This Matters in Diligence
During diligence, investors are not overwhelmed by volume.
They filter quickly:
What defines the product?
What supports the pathway?
What proves the evidence is sufficient?
If those answers are not clear, everything else becomes irrelevant.
Where Kandih Comes In
This is where Kandih Group focuses diligence on what actually matters.
Kandih supports investors and companies by:
Identifying high-signal regulatory documents
Evaluating pathway and classification logic
Aligning risk management with evidence strategy
Interpreting FDA feedback in context
Filtering out noise to highlight material risks
Structuring regulatory narratives that hold under scrutiny
Instead of reviewing everything, we focus on what changes decisions.
That improves:
Diligence efficiency
Risk clarity
Investment confidence
The Real Lesson
The company at the beginning didn’t fail because they lacked documentation.
They failed because their documentation didn’t answer the right questions.
Bottom Line
Regulatory documentation is not about volume.
It is about signal:
Does the pathway hold?
Is the risk understood?
Does the evidence support the claim?
Everything else is secondary.
When documentation is focused and aligned, it builds confidence.
When it is scattered and excessive, it raises questions.
That’s the difference between looking prepared—and actually being ready.
References
FDA – Design Controls for Medical Devices
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/design-controls-medical-devices
FDA – Substantial Equivalence in Premarket Notifications (510(k))
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/substantial-equivalence-premarket-notifications-510k
FDA – Requests for Feedback and Meetings for Medical Device Submissions (Q-Submission Program)
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/requests-feedback-and-meetings-medical-device-submissions-q-submission-program
FDA – Refuse to Accept Policy for 510(k)s
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/refuse-accept-policy-510ks