Uncategorized
April 23, 2026
Published by admin on April 23, 2026
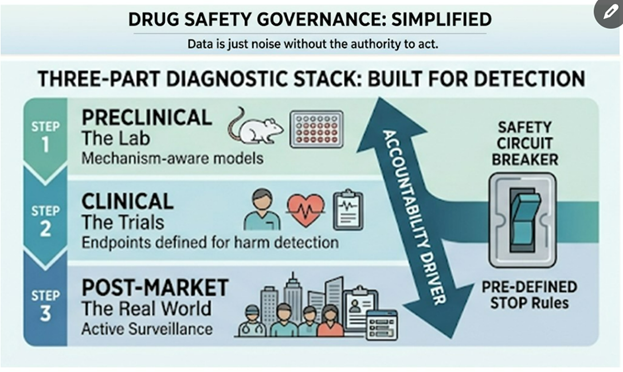
Every major medical product safety disaster has a predictable anatomy. From thalidomide in the 1960s to Vioxx in the 2000s, from Avandia to recent issues with JAK inhibitors, ranitidine, and opioids, the pattern is the same. It’s not that the science was missing – often the safety signals were there – but the system refused to be accountable to the science. In each case, warning signs existed, yet no one owned the obligation to act on them. This uncomfortable truth underpins why simply getting “better data” isn’t enough: safety advice only matters when someone is accountable to enforce it.
April 22, 2026
Published by admin on April 22, 2026
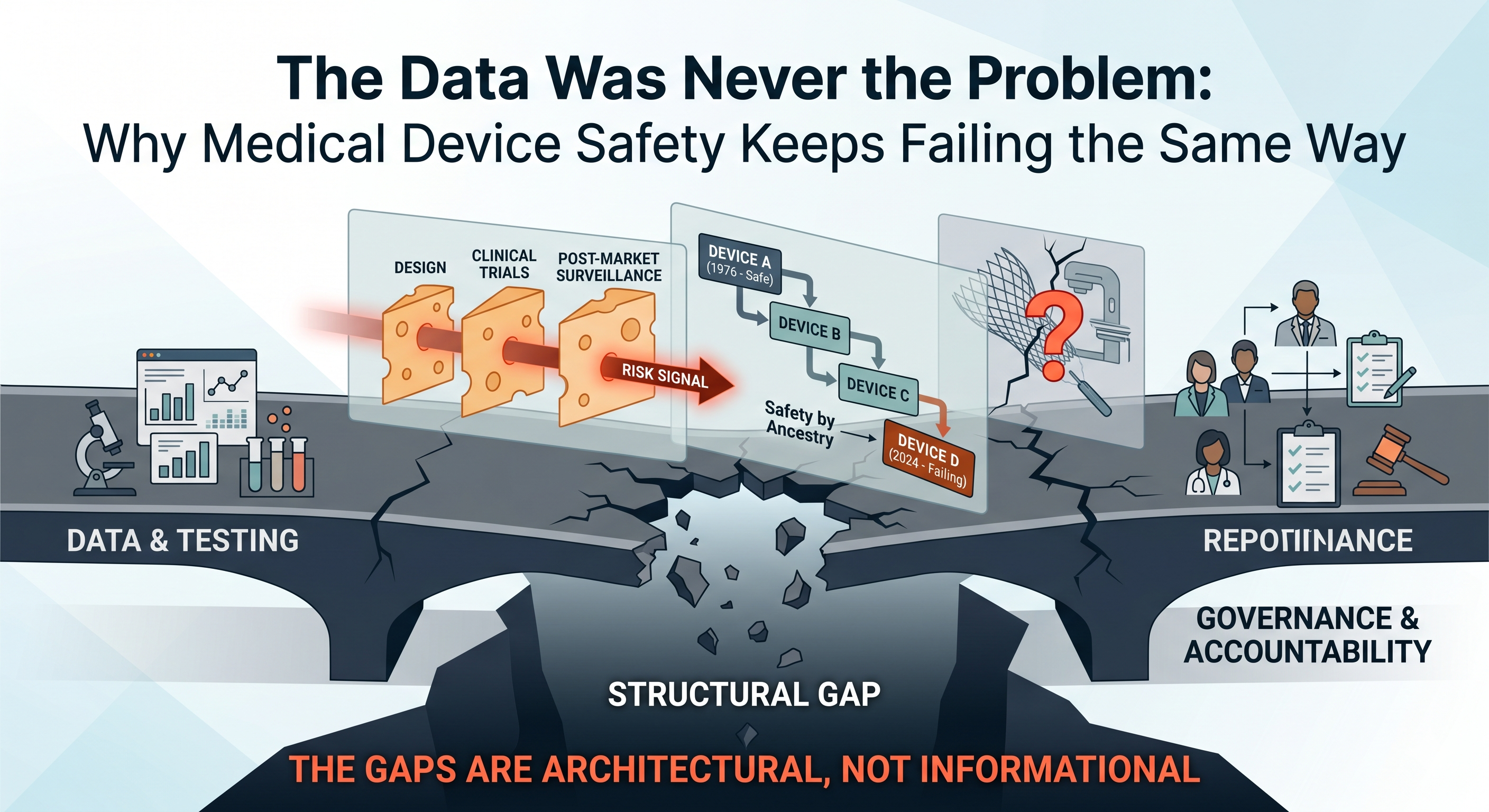
Every major medical device safety disaster has a predictable anatomy — and it's the same anatomy as drug safety disasters. From the Dalkon Shield in the 1970s to metal-on-metal hips in the 2000s, from Therac-25's lethal radiation overdoses to Philips' contaminated CPAP machines, the pattern repeats: warning signs existed, the science was available, and no one with authority was accountable to act on it. The device world just has its own particularly insidious ways of letting danger through.
April 20, 2026
Published by admin on April 20, 2026
A drug can be clinically successful, commercially promising, and still carry an environmental problem nobody planned for. That usually does not show up in the first investor deck.
It does not get much airtime in early product meetings.
And it definitely does not sound exciting at kickoff. Until it becomes a filing issue.
April 16, 2026
Published by admin on April 16, 2026
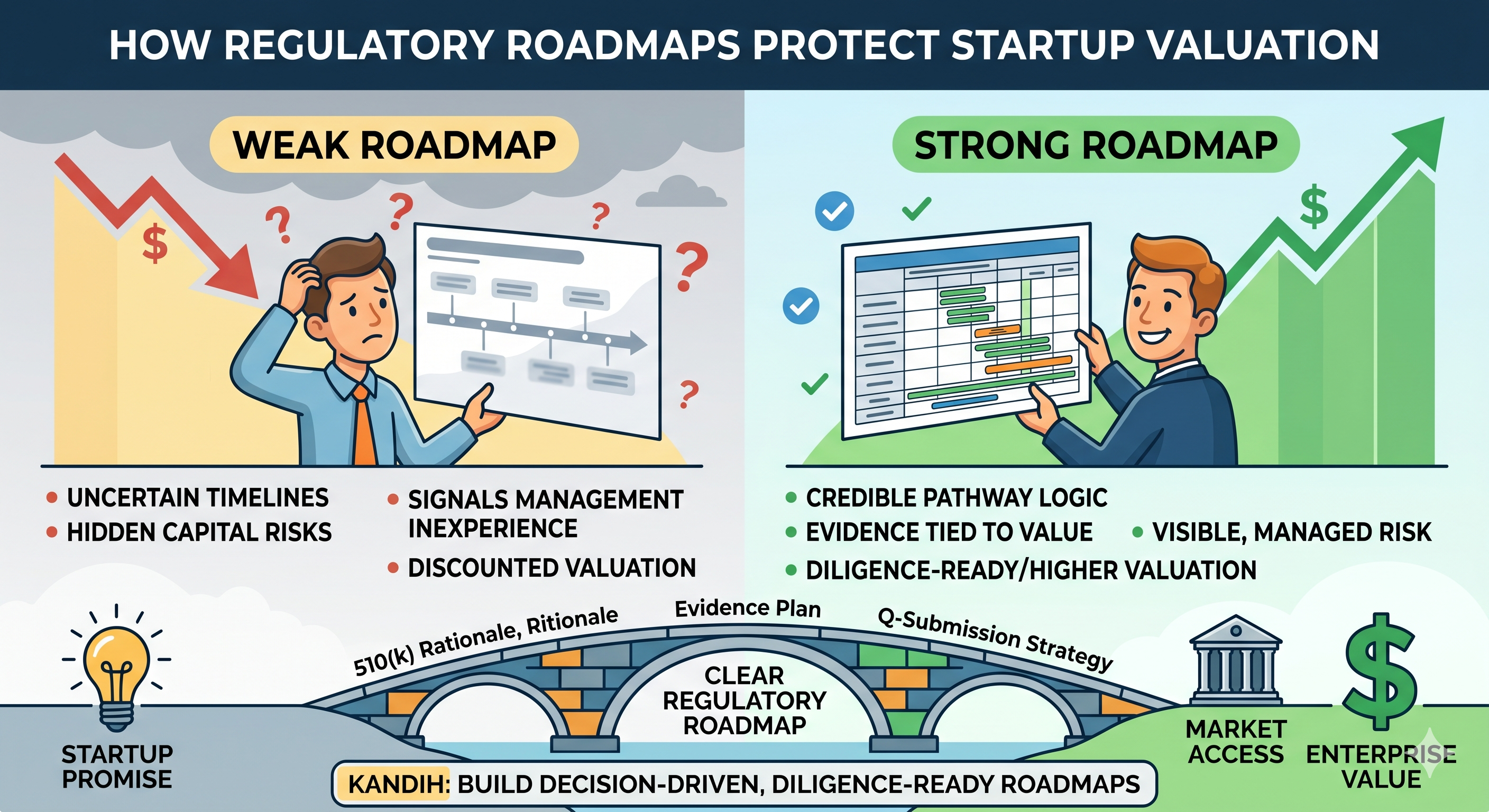
A startup can lose value long before anything officially goes wrong. No recall.No warning letter.No failed company announcement. Just growing doubt. An investor starts asking harder […]