How Roadmaps Protect Valuation

Why Timelines Alone Are Misleading
April 15, 2026Roadmaps Investors Trust
April 17, 2026A startup can lose value long before anything officially goes wrong.
No recall.
No warning letter.
No failed company announcement.
Just growing doubt.
An investor starts asking harder questions.
The next round takes longer.
The valuation discussion gets a little less confident.
Then a little less generous.
Why?
Because valuation is not driven by promise alone.
It is driven by how much uncertainty still sits between the product and the market.
That is where a regulatory roadmap matters.
Not the fake kind.
Not the pretty timeline in a pitch deck.
A real roadmap protects valuation because it reduces uncertainty investors care about most.
Regulatory clarity supports valuation because it improves investor confidence in timeline credibility, capital efficiency, submission readiness, and overall execution risk. In medical devices, pathway choice and evidence requirements can materially change cost and timing. FDA states that a 510(k) must demonstrate substantial equivalence to a legally marketed device, and the Q-Submission Program exists so companies can request FDA feedback on planned submissions before locking in avoidable mistakes. Those realities make regulatory planning a valuation issue, not just an operations issue.
Why Valuation Is Really About Confidence
Investors do not just price the product.
They price the risk around the product.
That includes questions like:
- Is the regulatory path believable?
- Is the timeline credible?
- Is the evidence plan strong enough?
- Could FDA feedback materially change the burn rate?
- Are there hidden issues that could delay market access?
When those questions do not have strong answers, valuation takes a hit. Even if the science is exciting. Even if the market opportunity is real.
Recent medtech M&A commentary also reflects this broader reality: valuations are shaped not only by growth and market position, but by regulatory considerations and reimbursement clarity.
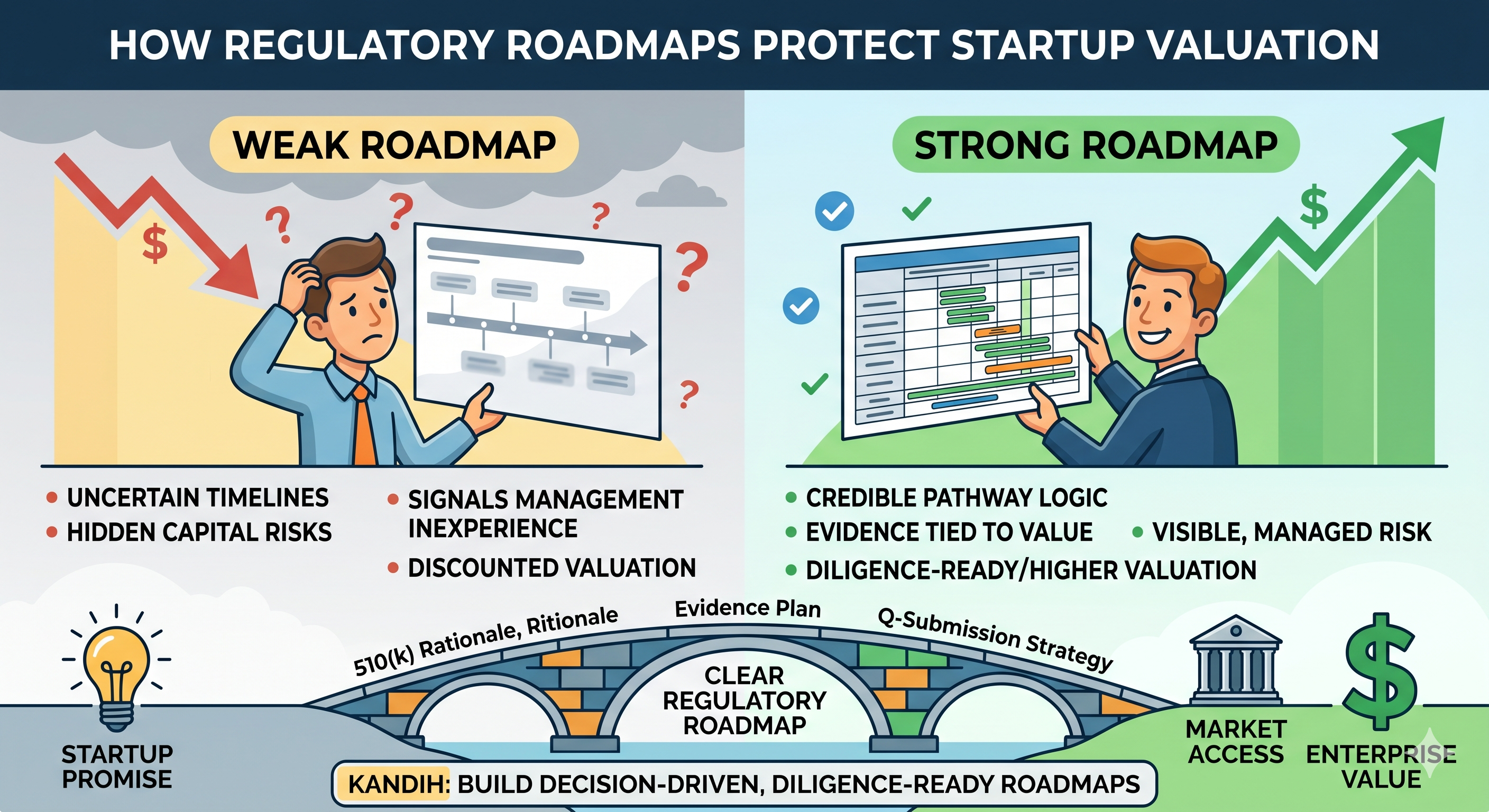
How Weak Roadmaps Hurt Valuation
A weak roadmap creates the kind of uncertainty investors discount immediately.
1. It makes the timeline look less believable
If the roadmap says “submit in 12 months” but does not explain why the pathway is stable, investors will treat the date as soft.
And they should.
FDA’s 510(k) framework is not based on wishful timing. It is based on whether the sponsor can support substantial equivalence to a legally marketed device. If that logic is weak, the date attached to it is weak too.
2. It raises concerns about future capital needs
A poor roadmap often hides the possibility of extra studies, added FDA questions, or a pathway shift that changes the evidence burden.
That matters because unexpected regulatory work usually means unexpected cash needs.
And nothing cools investor enthusiasm like the phrase, “We now need more data than originally planned.”
3. It signals management may not fully understand regulatory risk
Investors do not expect zero risk. They expect visible risk.
If the roadmap glosses over assumptions, ignores decision points, or treats FDA interaction like a calendar event instead of a strategic inflection point, it signals that management may not fully understand the exposure. FDA’s Q-Submission Program is specifically designed to support interactions that shape medical device submission strategy, including requests for feedback and meetings.
That is not just a regulatory concern. It is a credibility concern.
How Strong Roadmaps Protect Valuation
A credible roadmap protects valuation because it gives investors reasons to believe the plan is grounded in reality.
1. It shows the pathway logic
A strong roadmap explains why the company believes the device fits a given pathway and what could change that view.
That makes a huge difference in diligence.
If a company says it is pursuing a 510(k), investors want to know the rationale is defensible. FDA’s own description makes clear that the submission must compare the new device to one or more similar legally marketed devices and support the substantial equivalence claim.
That is the kind of logic that strengthens confidence.
2. It connects evidence to value
A strong roadmap does not just say testing is happening. It shows whether the evidence being built actually supports the claims, the intended use, and the submission strategy.
That helps investors judge whether the company is spending money in a way that reduces risk rather than simply generating activity. FDA notes that the 510(k) program involves review of safety and performance data to determine substantial equivalence.
When evidence is clearly tied to the regulatory question, valuation conversations get firmer because the path looks more real.
3. It makes uncertainty visible
This is the part smart investors respect.
A strong roadmap does not pretend there is no uncertainty. It shows where uncertainty lives and how the company plans to reduce it.
That includes:
- pathway assumptions
- evidence gaps
- claim sensitivity
- decision points
- risk triggers for reassessment
Visible uncertainty is easier to diligence than hidden uncertainty. Hidden uncertainty gets punished.
4. It improves diligence quality
When the roadmap is clear, diligence moves faster and with less friction.
Investors can see:
- what the plan depends on
- what has already been validated
- where FDA input matters
- what could still change the cost or timeline
That does not guarantee a higher valuation every time. But it absolutely improves the company’s ability to defend its valuation with logic instead of optimism.
Why Investor Confidence and Regulatory Clarity Are Linked
Investor confidence rises when uncertainty becomes more measurable.
That is why regulatory clarity matters so much.
Not because it removes all risk.
Because it makes risk understandable.
A roadmap that shows the real path, the real assumptions, and the real pressure points gives investors something they can underwrite.
A roadmap built only around milestones does the opposite. It invites suspicion.
Industry-facing medtech firms describe roadmap work in similar business terms. NAMSA frames startup roadmap planning as a cross-functional strategy intended to reduce time, cost, and risk, while medtech valuation commentary points to regulatory considerations as part of valuation frameworks in M&A.
Where Kandih Comes In
Kandih helps protect valuation by building decision-driven, diligence-ready regulatory roadmaps.
That means Kandih does not just map dates and milestones. Kandih helps companies show investors:
- why the current pathway makes sense
- what evidence is needed to support it
- where the assumptions are
- what could change timing or cost
- how FDA engagement can reduce downstream uncertainty
This matters because valuation is strongest when the company can defend its future with more than confidence.
Kandih helps turn regulatory planning into something investors can actually trust.
That is valuation protection in practical terms.
Not hype.
Not theater.
Not “trust us.”
Just better clarity, better logic, and fewer surprises.
Key Takeaways
Roadmaps protect valuation when they:
- reduce perceived regulatory uncertainty
- make timelines more credible
- connect evidence strategy to regulatory outcomes
- surface risks before investors find them in diligence
- improve confidence in management judgment
In medtech, regulatory clarity does not just support execution. It supports enterprise value.
Frequently Asked Questions
How does a regulatory roadmap affect valuation?
A regulatory roadmap affects valuation by influencing investor confidence in timeline credibility, capital needs, and the likelihood of reaching market through the expected pathway. In medical devices, those factors are closely tied to FDA pathway and evidence requirements.
Why do investors care about regulatory clarity?
Because regulatory uncertainty can change cost, delay milestones, and weaken the investment case. Investors are more confident when the regulatory path, evidence plan, and decision points are visible and defensible.
Can a weak roadmap lower valuation even before something goes wrong?
Yes. Investors often discount valuation when the roadmap lacks clear pathway logic, hides assumptions, or fails to show how regulatory risk will be managed. That kind of uncertainty affects diligence and financing discussions early.
What makes a roadmap more investor-friendly?
A roadmap is more investor-friendly when it shows pathway rationale, evidence needs, assumptions, regulatory decision points, and triggers for reassessment instead of just listing milestones.
Final Thought
Valuation is not just about what the company could become.
It is about how believable the path is from here to there.
That is why roadmaps matter.
A weak roadmap adds doubt.
A strong roadmap builds confidence.
And confidence is one of the few things that can protect valuation before the market ever rewards the product.
References
- FDA: Premarket Notification (510(k))
- FDA: Requests for Feedback and Meetings for Medical Device Submissions: The Q-Submission Program
- FDA: FDA Continues to Strengthen the 510(k) Program
MDDI: M&A in the Medical Device Industry: Key Factors Driving Valuation