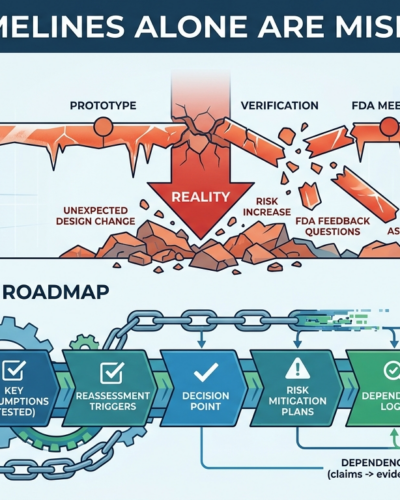
Why Timelines Alone Are Misleading
Why Most Regulatory Roadmaps Fail
April 14, 2026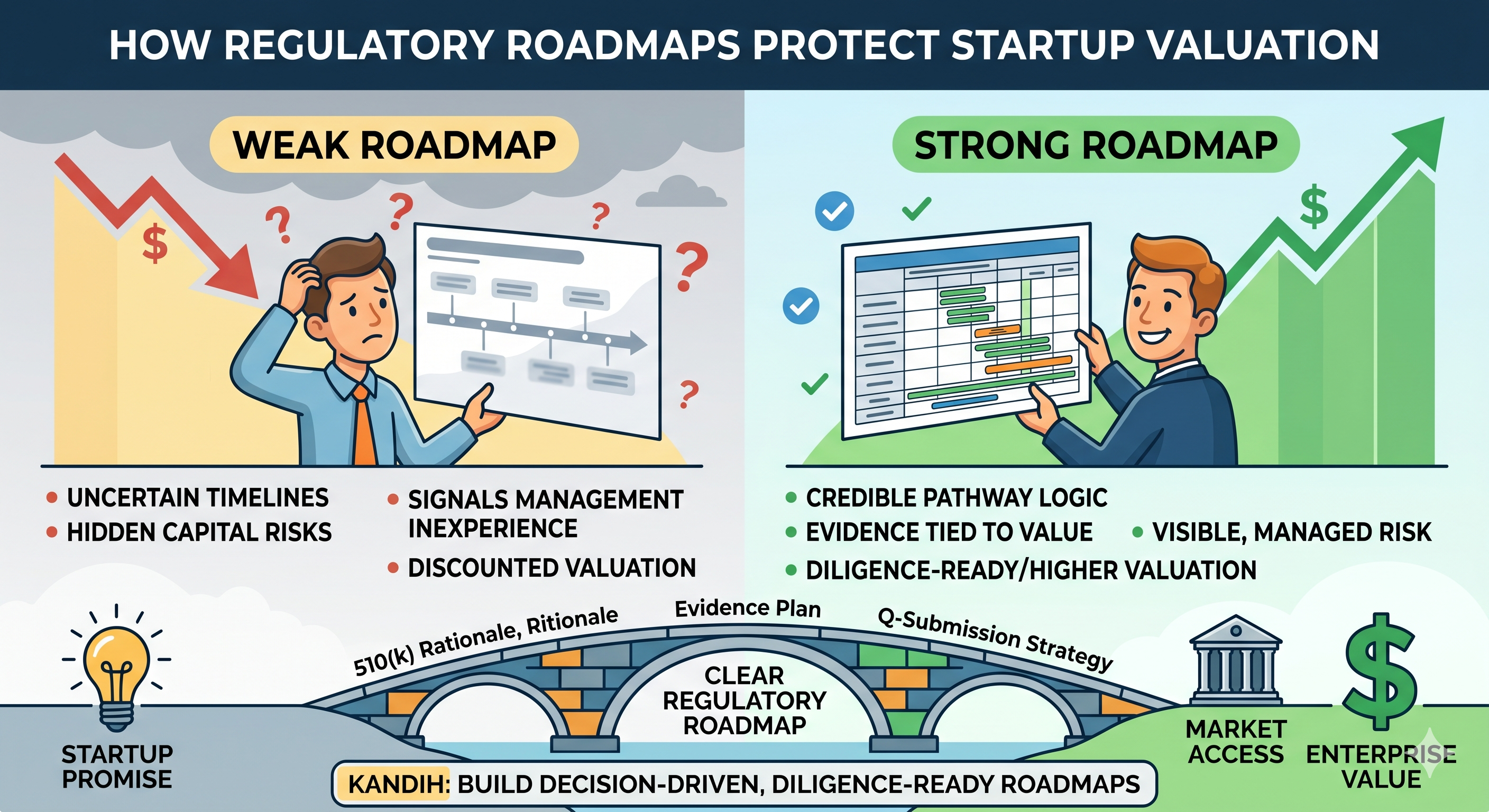
How Roadmaps Protect Valuation
April 16, 2026The timeline looked brilliant.
Prototype by June.
Verification by September.
FDA meeting by November.
Submission next year.
Clean. Sharp. Reassuring.
Then reality showed up.
One design change delayed testing.
A risk issue changed the evidence plan.
FDA feedback raised questions the team had not built for.
Suddenly, the timeline that looked so certain started behaving like fiction.
That is the problem with timelines alone.
They make uncertainty look smaller than it is.
And in medical device development, uncertainty is not a side issue. It is part of the work.
Timelines are useful for tracking activity, but they are misleading when they ignore uncertainty and dependency logic. In medical device development, regulatory strategy depends on pathway choice, evidence requirements, design stability, intended use, and FDA feedback. A 510(k), for example, depends on showing substantial equivalence to a legally marketed device, while the Q-Submission Program exists so companies can seek FDA feedback on planned submissions before locking in the wrong plan. That means a roadmap built only around dates can hide major strategic risk. (fda.gov)
Why Timelines Feel Safer Than They Really Are
People like timelines because timelines are simple.
They create the feeling that work is under control.
They show progress.
They give investors and teams something easy to follow.
But timelines only show one thing well:
when you hope something happens.
They do not show:
- how confident you are in that plan
- what assumptions the plan depends on
- what could change the path
- which steps rely on other steps being right first
That is where the danger starts.
A timeline can look precise while hiding a pile of uncertainty underneath it.
The Real Issue: Uncertainty
In regulatory planning, uncertainty is everywhere.
A company may not yet know:
- whether the pathway is fully stable
- whether the intended use will stay narrow
- whether the chosen predicate will hold
- whether bench testing will be enough
- whether FDA will agree with the proposed strategy
Those are not minor details. Those are the decisions that shape cost, time, and submission success.
FDA’s 510(k) framework makes this clear. A 510(k) is not just a filing event on a calendar. It is a submission intended to show that a device is as safe and effective as, meaning substantially equivalent to, a legally marketed device, and that claim must be supported. (fda.gov)
So when a timeline says “510(k) submission in Q4,” it may sound solid. But unless the underlying assumptions are tested, that date is more wish than plan.
The Other Issue: Dependency Logic
This is the part many teams skip.
A good roadmap does not just ask, “What comes next?”
It asks, “What must be true before the next step makes sense?”
That is dependency logic.
For example:
- You cannot finalize a submission strategy if the intended use is still moving.
- You cannot rely on an evidence plan if the claims are not stable.
- You cannot trust a submission date if a design change may affect risk classification.
- You cannot assume testing is enough if FDA has not yet weighed in on the evidence approach.
That is why timelines alone mislead people. They show sequence, but they often fail to show dependence.
And when the hidden dependencies start shifting, the whole schedule starts wobbling.
Why This Becomes Obvious Under FDA Feedback
FDA feedback often reveals the exact parts of the plan that were treated too casually.
The Q-Submission Program exists to give submitters a way to request FDA feedback and meetings related to medical device submissions. In practice, that means companies have a mechanism to pressure-test their assumptions before they lock in costly work. (fda.gov)
And that is where timeline-only planning runs into trouble.
The team may have scheduled the FDA interaction as a milestone.
But FDA sees it as a strategic review point.
That difference matters.
Because if FDA feedback changes:
- the evidence burden
- the pathway logic
- the risk discussion
- the scope of claims
…the original timeline was never as solid as it looked.
What Timeline-Only Planning Gets Wrong
It treats dates like certainty
A date on a roadmap often looks more certain than it deserves to look.
But if the date depends on unresolved questions, it is not really a commitment. It is a placeholder with nice formatting.
It hides assumptions
Many timelines quietly assume that the product, evidence plan, and regulatory path will all stay stable.
That is a dangerous habit.
It ignores linked risks
Regulatory planning is not linear. One change can affect several other parts of the strategy at once.
A design change may affect risk.
Risk may affect testing.
Testing may affect claims.
Claims may affect pathway logic.
Pathway logic may affect timing and cost.
That is dependency logic in the real world.
It makes movement look like progress
A team can be busy and still be heading in the wrong direction.
That is why activity alone is a poor measure of regulatory readiness.
Industry guidance and competitor content also emphasize that device development starts with planning, research, documentation, and risk analysis, not just scheduling tasks. (greenlight.guru)
What Better Planning Looks Like
A strong roadmap keeps the timeline, but adds the logic the timeline is missing.
That means showing:
1. Uncertainty
What is still unknown?
What assumptions need to be tested?
2. Dependencies
What decisions depend on other decisions?
What needs to be true before moving ahead?
3. Decision points
Where does the team stop, review, and confirm the plan still holds?
4. Risk triggers
What kinds of changes force reassessment?
That turns a roadmap from a static schedule into a useful management tool.
Why This Matters for Founders and Investors
Founders need realistic planning because wrong assumptions burn cash fast.
Investors need realistic planning because timeline risk is rarely just timing risk. It is often a signal of deeper regulatory uncertainty.
That is why roadmaps that only show milestones can be misleading in diligence. They may look organized while hiding the exact risks that matter most.
A roadmap becomes more credible when it shows not only where the company plans to go, but also what could change the route.
Where Kandih Comes In
Kandih incorporates risk into planning.
That means Kandih does not build roadmaps as simple sequences of dates and milestones. Kandih builds planning frameworks that reflect uncertainty, dependencies, and the reality that regulatory strategy evolves as the product evolves.
Kandih helps companies define:
- the assumptions behind the current path
- the dependencies between design, evidence, claims, and submission strategy
- the risk points that could change timing or cost
- the moments where FDA feedback should inform the next move
That approach leads to roadmaps that are more honest, more useful, and far more resilient.
Because the goal is not to create a timeline that looks confident.
The goal is to create a plan that stays credible when reality starts pushing back.
Key Takeaways
Timelines alone are misleading because they:
- make uncertainty look smaller than it is
- hide dependencies between key decisions
- treat dates as fixed when the logic is still moving
- confuse activity with progress
- break down quickly when FDA feedback exposes weak assumptions
A better roadmap includes time, but it also includes risk, dependency logic, and decision points. (fda.gov)
Frequently Asked Questions
Why are timelines alone misleading in regulatory planning?
Because they show expected dates but often hide uncertainty, assumptions, and dependencies that determine whether those dates are realistic. (fda.gov)
What is dependency logic in a regulatory roadmap?
Dependency logic means identifying what must be true before the next step makes sense. For example, pathway timing may depend on evidence strategy, which may depend on stable claims and design inputs. (greenlight.guru)
Why does FDA feedback affect timelines so much?
Because FDA feedback can change the assumptions behind the plan, including evidence expectations, pathway logic, and risk interpretation. The Q-Submission Program exists to help companies seek that feedback earlier. (fda.gov)
What makes a roadmap more realistic?
A realistic roadmap includes uncertainty, dependencies, decision points, and risk triggers, not just dates and milestones. (greenlight.guru)
Final Thought
A timeline can tell you when you want to arrive.
It cannot tell you whether the road is stable.
That is why timelines alone are misleading.
They give comfort without context.
Precision without confidence.
Motion without enough logic.
In medical device development, that is a dangerous bargain.
References
- FDA: Premarket Notification (510(k))
- FDA: Requests for Feedback and Meetings for Medical Device Submissions: The Q-Submission Program
- FDA: FDA Continues to Strengthen the 510(k) Program
- Greenlight Guru: Medical Device Development Process
- Greenlight Guru: Creating a Regulatory Roadmap for MedTech Market Success