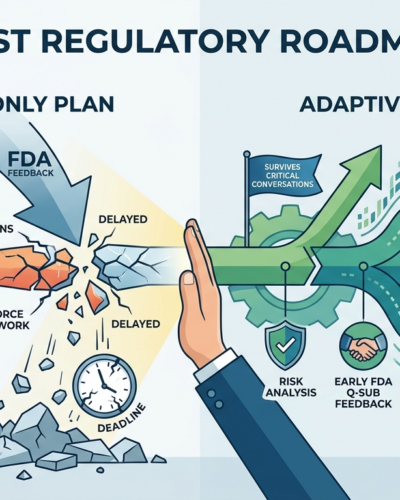
Why Most Regulatory Roadmaps Fail
What Is a Regulatory Roadmap—Really?
April 13, 2026
Why Timelines Alone Are Misleading
April 15, 2026Everything looks fine until FDA responds.
The roadmap is on track.
The milestones are moving.
The team is hitting deadlines.
Then the feedback arrives.
FDA wants clarification on intended use.
The evidence does not fully support the claims.
A design choice creates new questions about risk.
The submission path no longer looks as simple as it did on the kickoff slide.
That is when many companies learn the truth:
Most regulatory roadmaps fail because they are built as
milestone plans, not decision systems.
And milestone-only plans collapse the moment FDA feedback
tests the assumptions underneath them.
Most regulatory roadmaps fail because they focus on
deadlines instead of regulatory decisions. In medical devices, FDA pathway
strategy, intended use, predicate logic, and evidence requirements determine
whether a plan is realistic. When a roadmap is built only around milestones,
FDA feedback can expose weak assumptions too late, forcing rework, delays, and
higher cost. FDA’s Q-Submission Program exists specifically to allow companies
to request feedback and meetings related to potential or planned medical device
submissions before those problems become more expensive.
The Core Problem
A lot of roadmaps are really just project schedules in
disguise.
They say things like:
complete
testing in Q2
meet
FDA in Q3
submit
in Q4
launch
next year
That may help a team track activity, but it does not answer
the harder questions:
Are we
on the right pathway?
Does
the evidence support the claims?
What
assumptions are holding this plan together?
What
happens if FDA disagrees?
That is why these plans break so easily. They are built
around movement, not regulatory logic.
Why Milestone-Only Plans Collapse
1. They assume the pathway is already correct
A milestone plan often starts with a big assumption: the
company already knows the right regulatory path.
That is risky.
For example, a 510(k) is not just a box to tick. FDA says a
510(k) submission must demonstrate that the device is as safe and effective,
meaning substantially equivalent, to a legally marketed device. That requires a
defensible comparison and support for the substantial equivalence claim. If
that logic is weak, the timeline built around it is weak too.
So when a roadmap says “submit 510(k) in 12 months” without
showing why that path is stable, it is not a roadmap. It is a gamble wearing
business casual.
2. They treat FDA feedback like a milestone, not a stress
test
A milestone-only plan usually treats FDA interaction as one
step on the calendar.
But FDA feedback is not just another meeting. It is a test
of the plan.
FDA’s Q-Submission guidance explains that the program
provides mechanisms for submitters to request interactions with FDA related to
medical device submissions, including written feedback and meetings. That means
FDA engagement is meant to shape strategy, not just confirm that the meeting
happened.
When teams treat FDA feedback as a checkbox instead of a
decision point, they miss the whole point of getting feedback early.
3. They separate milestones from evidence quality
A weak roadmap often tracks whether studies are happening,
but not whether those studies answer the right regulatory question.
That is a major problem.
Investors and regulators do not care that testing is “on
schedule” if the evidence does not support the intended claims. FDA’s premarket
framework is evidence-based. The submission must support safety, effectiveness,
or substantial equivalence depending on the pathway. Activity alone does not do
that.
In plain English, wrong data collected on time is still the
wrong data.
4. They hide assumptions until FDA exposes them
Milestone plans often leave the fragile parts unspoken:
the
intended use will stay narrow
the
design will stay stable
the
predicate will hold
bench
testing will be enough
FDA
will see the product the way the company does
Those assumptions sit quietly in the background until FDA
feedback shines a spotlight on them.
Then the team has a painful moment of clarity.
The timeline did not fail because the team was lazy.
It failed because the strategy was built on assumptions nobody tested early
enough.
5. They do not tell teams when to stop and rethink
A strong roadmap should say, “If this changes, we reassess.”
Most weak roadmaps never say that.
So when there is:
a
design change
a
shift in claims
an
unexpected test result
new
risk information
FDA
feedback that changes the evidence burden
…the team keeps marching forward as if nothing happened.
That is how small misalignments become expensive delays.
What FDA Feedback Actually Does
FDA feedback often does not “break” a roadmap.
It reveals that the roadmap was already weak.
It exposes issues such as:
unclear
intended use
unstable
predicate strategy
evidence
gaps
claim-evidence
mismatch
underappreciated
risk questions
That is why early feedback matters. FDA created the
Q-Submission pathway so companies can request feedback before locking
themselves into avoidable rework.
Good teams use feedback to adapt.
Weak teams treat feedback like a disruption, when really it
is a warning they should have planned for.
What Strong Roadmaps Do Differently
A real regulatory roadmap is built around decisions, not
just dates.
It should show:
Pathway logic
Why this regulatory path makes sense now, and what could
destabilize it.
Evidence strategy
What evidence is needed, what it is meant to prove, and
where the gaps still are.
Assumption mapping
What the current plan depends on, stated clearly instead of
buried in a slide deck.
Reassessment triggers
What events force the team to pause, review, and update
strategy.
That kind of roadmap is harder to make.
It is also far less likely to explode on contact with FDA.
Why This Matters for Founders and Investors
For founders, weak roadmaps waste time, money, and
credibility.
For investors, weak roadmaps create hidden execution risk. A
company can look on plan right up until FDA feedback turns the next milestone
into a moving target.
That is why roadmap quality matters so much in medtech.
Industry guidance and competitor content also frame development planning as
broader than scheduling, emphasizing planning, research, risk analysis,
documentation, and the need to update plans as development evolves.
A roadmap that cannot adapt is not efficient. It is brittle.
Where Kandih Comes In
Kandih uses an adaptive, decision-driven roadmap approach.
That means the roadmap is not built as a fixed sequence of
milestones that assumes everything will go according to plan. It is built to
help companies respond intelligently when the plan is challenged.
Kandih helps teams define:
the
current pathway logic
the
evidence needed to support it
the
assumptions carrying the strategy
the
trigger points that require reassessment
the
moments where FDA feedback should change the next decision
That makes the roadmap more useful because it reflects the
real world of device development, where evidence evolves, risk shifts, and FDA
feedback can materially change the path forward.
A milestone-only plan asks, “Are we still on schedule?”
An adaptive roadmap asks, “Are we still on the right path?”
That is the better question.
Key Takeaways
Most regulatory roadmaps fail because they:
focus
on milestones instead of decisions
assume
the pathway is stable without proving it
track
activity instead of evidence quality
hide
assumptions until FDA exposes them
lack
triggers for reassessment
FDA feedback does not usually create these weaknesses. It
reveals them.
Frequently Asked Questions
Why do regulatory roadmaps fail?
They usually fail because they are built as timelines
instead of decision frameworks. Once FDA feedback tests pathway logic, evidence
quality, or assumptions, the plan no longer holds.
Why is FDA feedback so disruptive?
Because it can expose weak assumptions about intended use,
evidence burden, predicate strategy, or risk that were never tested properly in
the original plan. The Q-Submission Program exists so companies can seek that
feedback earlier.
Is a milestone plan enough for a medical device roadmap?
No. Milestones help manage activity, but they do not replace
regulatory decision-making. A strong roadmap also needs pathway logic, evidence
strategy, visible assumptions, and reassessment triggers.
What makes a roadmap more resilient?
A resilient roadmap adapts when data changes, design
changes, or FDA feedback changes the strategy. That is why adaptive roadmaps
are stronger than milestone-only plans.
Final Thought
Most regulatory roadmaps do not fail because teams forgot to
plan.
They fail because teams planned the easy part.
Dates are easy.
Milestones are easy.
Regulatory uncertainty is the hard part.
That is why the better roadmap is not the prettier one.
It is the one that can survive the first serious
conversation with FDA.
References
FDA: Requests for Feedback and Meetings for Medical Device
Submissions: The Q-Submission Program
FDA: Premarket Notification (510(k))
FDA: The Q-Submission Program Guidance PDF
FDA: FDA Continues to Strengthen the 510(k) Program
Greenlight Guru: Medical Device Development Process
Greenlight Guru: Creating a Regulatory Roadmap for MedTech
Market Success