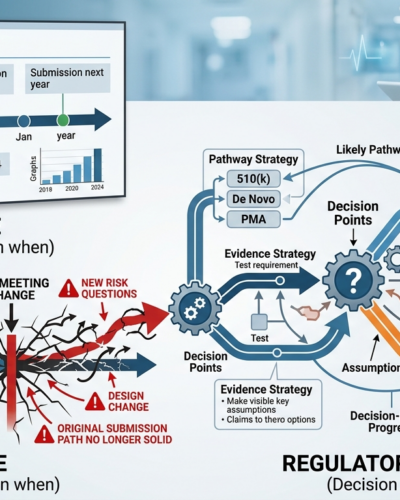
What Is a Regulatory Roadmap—Really?
Post-Investment Regulatory Risk Management
April 10, 2026
Why Most Regulatory Roadmaps Fail
April 14, 2026A medical device company can look perfectly organized on paper and still be heading toward a regulatory mess.
The timeline may look polished.
Prototype by Q2. Verification by Q3. Submission next year.
Investors feel reassured.
The team feels productive.
The board sees movement.
Then one FDA meeting changes everything.
The claims are too broad. The evidence plan is not strong enough. A design change raises new risk questions. The original submission path no longer looks as solid as it did in the kickoff meeting.
That is when many companies realize they were not working from a true regulatory roadmap at all.
They were working from a timeline.
And those are not the same thing.
A Regulatory Roadmap Is Not Just a Timeline
A timeline shows when tasks are supposed to happen.
A regulatory roadmap should show what decisions need to be made, what evidence is needed to support those decisions, and what risks could force the plan to change.
That distinction matters because market access for medical devices depends on regulatory judgment, not just project management. FDA pathways are shaped by intended use, risk, device type, available predicates, and the level of evidence needed to support safety and effectiveness or substantial equivalence.
So when a company says it has a roadmap, the real question is this:
Is it a list of dates?
Or is it a framework for making the right decisions under uncertainty?
Only one of those is a true regulatory roadmap.
What a Regulatory Roadmap Really Does
A real roadmap helps a company answer the questions that decide whether development moves forward smoothly or breaks down later.
It should help the team define:
- the likely regulatory pathway
- the evidence needed to support that pathway
- the assumptions behind the current plan
- the major risks that could change timing, cost, or submission strategy
- the points where FDA feedback should be used to reduce uncertainty
That is why a roadmap should be treated as a decision framework, not a decorative planning tool.
Strong medtech content from leading industry publishers consistently frames roadmap and strategy work around classification, evidence planning, market access, and cross-functional alignment rather than dates alone.
Why Timeline-Only Planning Fails
Here is the trap.
A timeline assumes the important decisions have already been made correctly.
But in early and mid-stage development, that is often not true.
A company may assume:
- the 510(k) pathway is available
- bench testing will be enough
- the intended use is clear
- the product claims will hold
- FDA will view the risk profile the same way the team does
That is a lot of faith packed into one schedule.
Once those assumptions are challenged, the timeline starts to crack. The company is not delayed because it moved too slowly. It is delayed because it moved forward on unstable assumptions.
That is where cost, rework, and investor anxiety usually enter the room.
What Should Be in a Real Regulatory Roadmap?
A professional roadmap should be built around decisions, not just milestones.
1. Pathway strategy
The roadmap should explain the likely regulatory path and why it fits the product.
That means pressure-testing whether the device is truly aligned with a 510(k), De Novo, or PMA pathway, depending on risk, novelty, and available comparators. FDA uses these pathways differently, and the choice has major implications for cost, timing, and evidence burden.
2. Evidence strategy
The roadmap should connect each key regulatory decision to the evidence needed to support it.
That includes questions such as:
- What testing is required?
- Will non-clinical evidence be enough?
- Could clinical evidence be needed?
- Do the studies actually support the intended claims?
Collecting data without a clear regulatory purpose is not progress. It is expensive motion.
3. Assumption mapping
Every roadmap sits on assumptions.
A strong roadmap makes them visible.
Maybe the team assumes the design will stay stable. Maybe it assumes the claims will remain narrow. Maybe it assumes FDA feedback will be favorable.
Those assumptions should not be hidden in a project chart. They should be called out early so the team knows what needs to be tested.
4. Reassessment triggers
This is where weak roadmaps usually fall apart.
A real roadmap should identify the events that trigger a strategic review, such as:
- a design change
- a shift in intended use
- unexpected test results
- new risk information
- FDA feedback that changes the evidence burden
Without those trigger points, the roadmap is static. And static plans rarely survive real product development.
Why This Matters for Founders and Investors
Founders need clarity because every wrong decision burns time and capital.
Investors need clarity because regulatory mistakes rarely stay contained. They affect timelines, milestone credibility, follow-on financing, and sometimes the entire investment thesis.
That is why a regulatory roadmap should not be treated as a one-time planning exercise. It is a business-critical tool for reducing uncertainty as the product evolves.
Competitor content in the sector also tends to position roadmap work as cross-functional and commercially important, not just technical. NAMSA, for example, describes startup roadmap planning in terms of parallel development opportunities, reduced time, reduced cost, and reduced risk.
Where Kandih Comes In
Kandih builds decision-driven regulatory roadmaps.
That means the work goes beyond plotting milestones on a slide.
Kandih helps companies define the decisions that actually shape outcome:
- which pathway makes sense now
- what evidence is truly needed
- where the assumptions are weak
- what changes could destabilize the plan
- when FDA engagement can reduce future rework
This is the difference between having a roadmap that looks professional and having one that actually protects the business.
A good roadmap should not just tell you what comes next.
It should tell you what must be true before you move.
That is the value of a decision-driven approach.
Final Thought
So, what is a regulatory roadmap—really?
It is not a timeline.
It is not a checklist.
It is not a document built to make a startup look organized.
It is a decision framework that helps a company make smarter regulatory choices before those choices become expensive corrections.
In medtech, that difference is not cosmetic.
It is operational. Financial. Strategic.
And sometimes, it is the difference between steady progress and a very expensive lesson.
References
- FDA: Premarket Notification (510(k))
- FDA: De Novo Classification Request
- FDA: Premarket Approval (PMA)
- FDA: Requests for Feedback and Meetings for Medical Device Submissions: The Q-Submission Program
- Greenlight Guru: Creating a Regulatory Roadmap for MedTech Market Success
- NAMSA: Solutions for MedTech Startups