Roadmaps Investors Trust
How Roadmaps Protect Valuation
April 16, 2026Environmental Risk Assessment in Drug Development: What It Is, Why It Matters, and When to Start
April 20, 2026A founder says the company has a regulatory roadmap.
The slide looks polished.
The milestones look neat.
The dates seem aggressive, but still believable.
An investor flips through it and thinks one thing:
Can I trust this?
That is the real test.
Because investors are not looking for a roadmap that sounds smart. They are looking for one that reduces uncertainty, shows clear thinking, and holds up under diligence.
A roadmap becomes credible when it stops being a hopeful timeline and starts becoming an evidence-based decision tool.
Investors trust regulatory roadmaps when they are grounded in real regulatory logic, clear assumptions, defined evidence needs, and visible decision points. In medical devices, pathway choice, evidence burden, and FDA interaction strategy can materially affect cost, timeline, and investment risk. FDA’s premarket framework makes that clear: a 510(k) requires support for substantial equivalence to a legally marketed device, while the Q-Submission Program exists to let companies seek FDA feedback on potential or planned submissions before problems become more expensive.
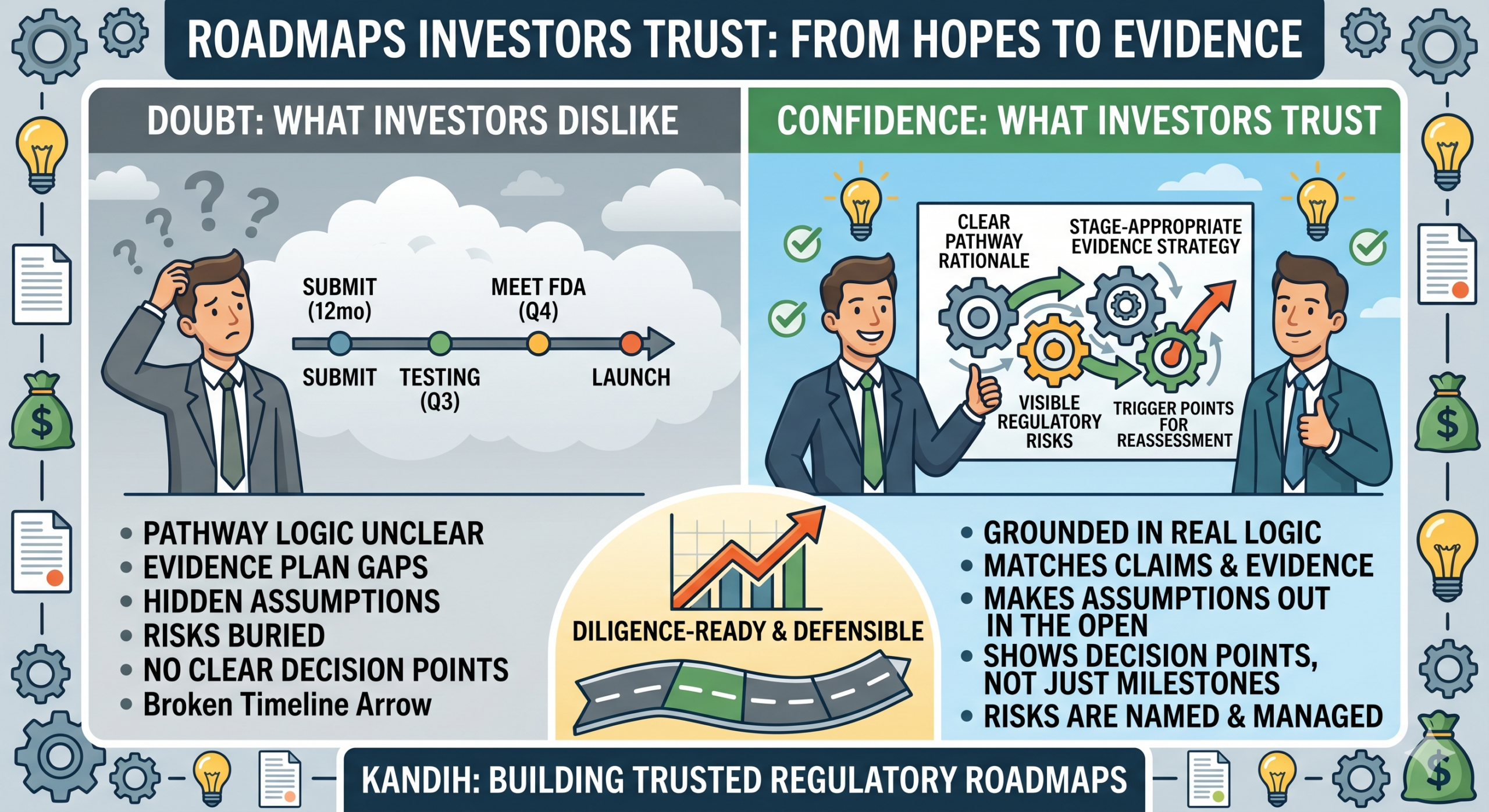
Why Investors Do Not Trust Most Roadmaps
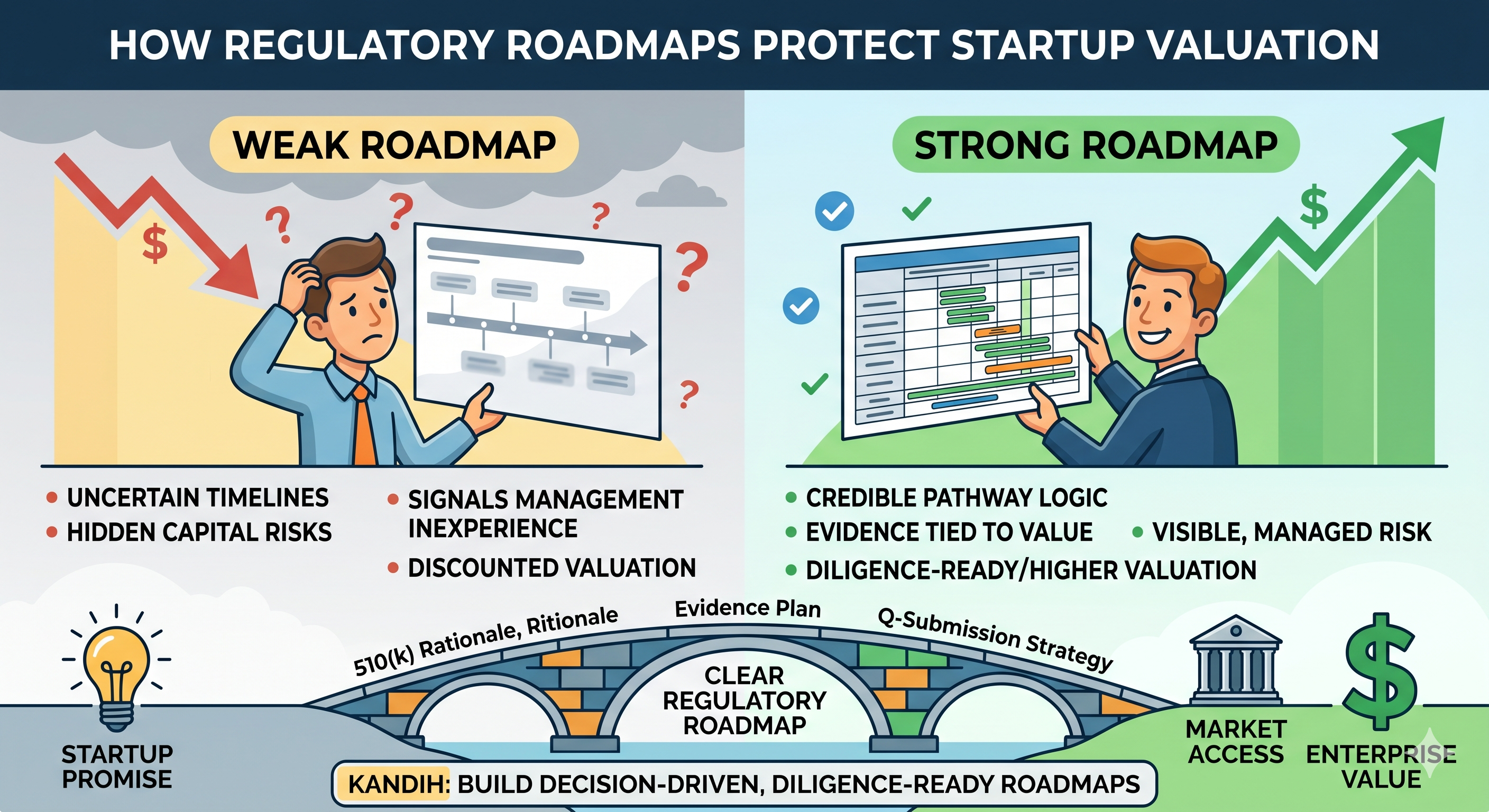
Investors have seen too many plans that look good in a board deck and fall apart in real life.
The problem is usually not that the roadmap is missing dates.
The problem is that it is missing proof.
A weak roadmap often sounds like this:
- submit in 12 months
- complete testing in Q3
- meet FDA in Q4
- launch after clearance
That may look organized, but it does not answer the questions investors actually care about:
- Why is this the right regulatory path?
- What evidence supports that assumption?
- What could break this plan?
- What happens if FDA disagrees?
- How much time and money sit behind each major risk?
A roadmap investors trust has answers. A roadmap investors doubt has slogans.
What Makes a Roadmap Credible to Investors
1. The pathway logic is clear
A credible roadmap explains why the company believes the product fits a particular regulatory path.
That means showing the reasoning behind the strategy, not just naming the pathway. If the company says it is pursuing a 510(k), the roadmap should show why that makes sense based on the product, intended use, and predicate strategy. FDA defines a 510(k) as a premarket submission used to show a device is as safe and effective, meaning substantially equivalent, to a legally marketed device.
Investors trust roadmaps more when the logic is visible. They trust them less when the path sounds chosen by wishful thinking.
2. The evidence plan matches the claims
A roadmap becomes believable when the evidence plan actually supports what the company wants FDA to accept.
That means the roadmap should show:
- what testing is planned
- why that testing matters
- what claims it is meant to support
- where the evidence gaps still sit
Investors know that “testing is underway” is not the same as “the right evidence is being built.” A roadmap is stronger when it connects evidence to the regulatory question, instead of just listing activities. FDA’s premarket materials make clear that submissions are reviewed based on evidence supporting safety, effectiveness, or substantial equivalence.
3. The assumptions are out in the open
A credible roadmap does not hide its assumptions.
It states them plainly.
For example:
- We assume the intended use stays within this scope.
- We assume the predicate strategy remains defensible.
- We assume bench testing will be sufficient unless FDA signals otherwise.
- We assume no design change will materially alter the risk profile.
Why does this matter to investors? Because hidden assumptions are where timeline surprises breed like rabbits.
A roadmap that makes assumptions visible is easier to diligence, easier to challenge, and easier to trust.
4. The roadmap shows decision points, not just milestones
Investors trust roadmaps that show when the company will stop, review, and decide.
That could include points such as:
- pathway confirmation
- evidence package review
- FDA feedback integration
- submission readiness review
- reassessment after design or claims changes
This is one reason the FDA Q-Submission Program matters so much. It gives companies a formal way to request feedback and meetings related to potential or planned medical device submissions. That feedback can materially change strategy, which means any roadmap pretending FDA input is just a box to tick is not investor-grade.
5. The risks are named, not buried
A roadmap investors trust does not pretend the path is clean.
It shows where the risk lives.
That may include:
- predicate uncertainty
- evidence burden uncertainty
- claim sensitivity
- design changes with regulatory impact
- possible need for additional FDA interaction
- timing risk tied to unresolved questions
Investors do not expect zero risk. They expect visible risk with a plan to manage it.
That is a big difference.
What Investors Really Want to See
At diligence stage, investors are usually not asking for perfection. They are asking whether management understands what could go wrong and whether the roadmap is strong enough to survive contact with reality.
A roadmap becomes diligence-ready when it shows:
- a defensible pathway rationale
- a stage-appropriate evidence strategy
- clear assumptions
- trigger points for reassessment
- a plan for FDA engagement where needed
- alignment between regulatory, technical, and business milestones
Industry players serving medtech startups frame roadmap work in a similar way. NAMSA, for example, describes a startup roadmap as a cross-functional plan spanning regulatory, clinical, preclinical, and testing areas, with milestones and parallel development opportunities intended to reduce time, cost, and risk.
That is the point. Credibility comes from structure, logic, and traceability.
Not from pretty formatting.
Red Flags That Make Investors Nervous
Investors tend to lose confidence when a roadmap:
- names a pathway without explaining why
- lists studies without showing what regulatory question they answer
- assumes FDA alignment without a plan to verify it
- ignores what happens if claims, design, or risk classification change
- treats submission timing as fixed even though key assumptions are unresolved
That kind of roadmap may impress someone for five minutes.
Then diligence starts.
And five minutes does not survive diligence.
Where Kandih Comes In
Kandih builds diligence-ready, decision-driven regulatory roadmaps.
That means the roadmap is not built to decorate a pitch deck. It is built to stand up to investor scrutiny.
Kandih helps companies turn regulatory planning into something investors can actually evaluate:
- a clear pathway rationale
- defined evidence requirements
- explicit assumptions
- realistic decision points
- visible regulatory risks
- a strategy for using FDA feedback to reduce uncertainty
That gives founders a better planning tool and gives investors a roadmap they can trust without needing to suspend disbelief.
In plain terms, Kandih helps turn “Here is our plan” into “Here is why this plan is credible.”
That is a much stronger sentence in any diligence room.
Key Takeaways
Investors trust roadmaps that are:
- grounded in real regulatory logic
- supported by an evidence-based plan
- honest about assumptions and risk
- structured around decisions, not just dates
- strong enough to hold up under diligence
A roadmap does not become credible because it is detailed.
It becomes credible because it is defensible.
Frequently Asked Questions
What makes a regulatory roadmap credible to investors?
A credible roadmap shows the logic behind the regulatory path, the evidence needed to support it, the assumptions behind the plan, the key decision points, and the risks that could change timing or cost.
Why do investors care about regulatory roadmaps?
Because regulatory strategy affects timeline reliability, capital needs, technical risk, and the overall probability of market access. In medical devices, changes in pathway or evidence burden can materially affect the investment case.
Is a timeline enough for diligence?
No. A timeline shows planned activity. A diligence-ready roadmap must also show pathway rationale, evidence strategy, assumptions, and trigger points for reassessment.
Why does FDA feedback matter so much?
Because FDA feedback can confirm, refine, or challenge the company’s assumptions before a submission is locked in. The Q-Submission Program exists to support those interactions.
Final Thought
Investors do not trust roadmaps because they are confident.
They trust roadmaps because they are clear.
Clear about the path.
Clear about the evidence.
Clear about the risks.
Clear about what still needs to be proven.
That is what makes a roadmap credible.
And in medtech, credible beats optimistic every single time.
References
- FDA: Premarket Notification (510(k))
- FDA: Requests for Feedback and Meetings for Medical Device Submissions: The Q-Submission Program
- FDA: The Q-Submission Program Guidance PDF
- FDA: 510(k) Premarket Notification Database Overview
- FDA: FDA Continues to Strengthen the 510(k) Program
- NAMSA: Solutions for MedTech Startups