April 17, 2026
Published by admin on April 17, 2026
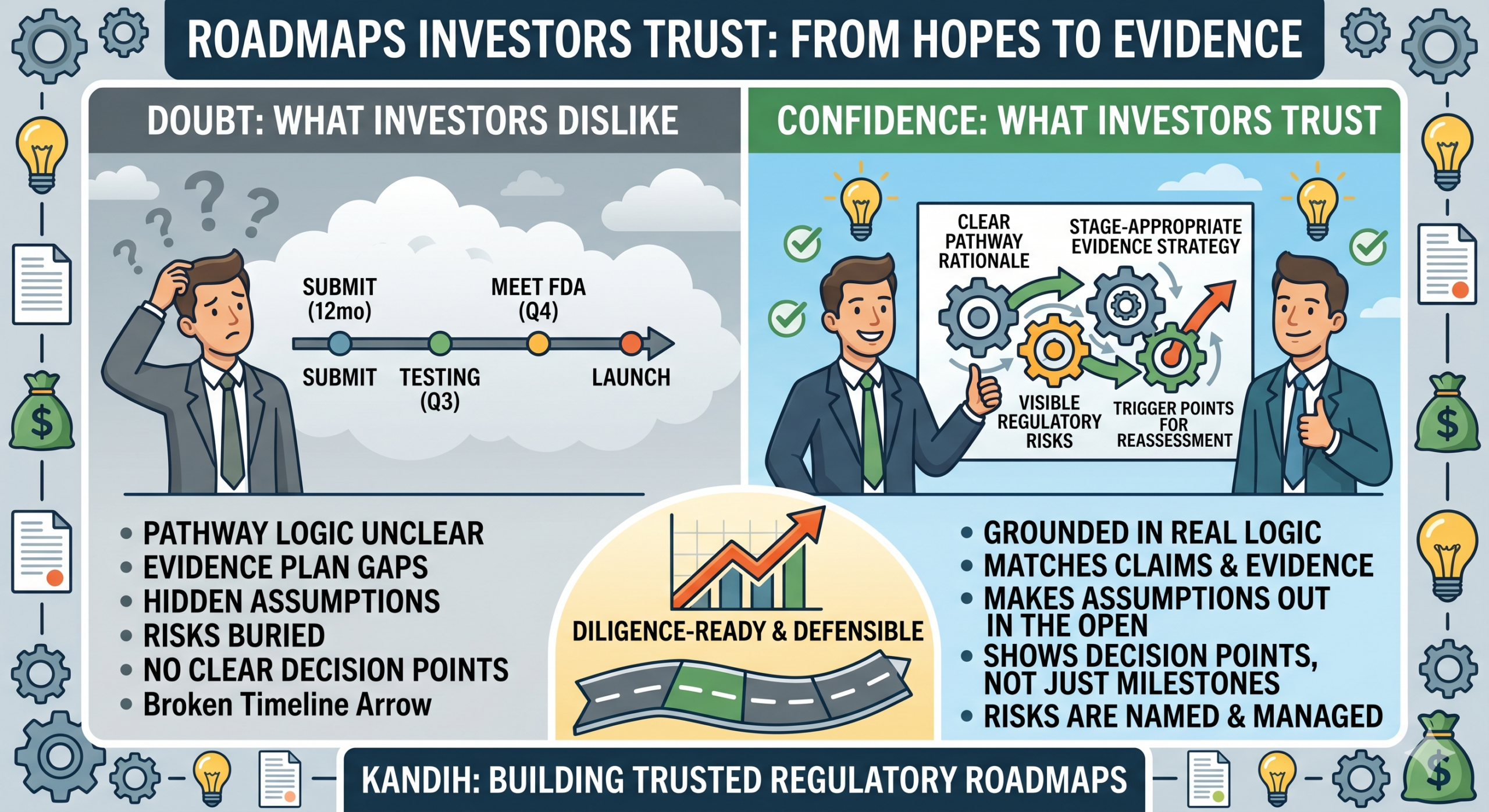
A founder says the company has a regulatory roadmap. The slide looks polished.
The milestones look neat.
The dates seem aggressive, but still believable. An investor flips through it and thinks one thing: Can I trust this? That is the real test.
April 16, 2026
Published by admin on April 16, 2026
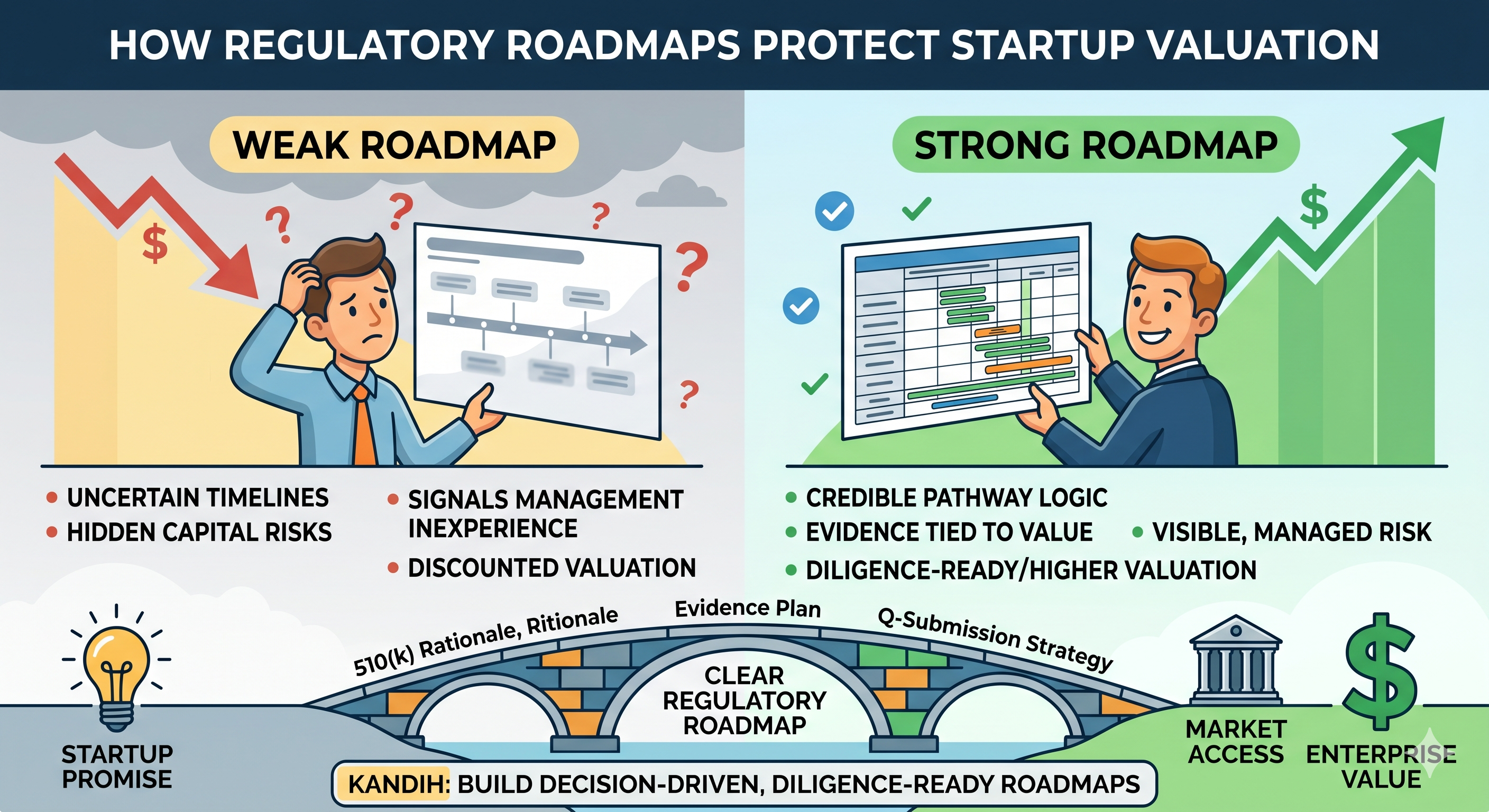
A startup can lose value long before anything officially goes wrong. No recall.No warning letter.No failed company announcement. Just growing doubt. An investor starts asking harder […]
April 15, 2026
Published by admin on April 15, 2026
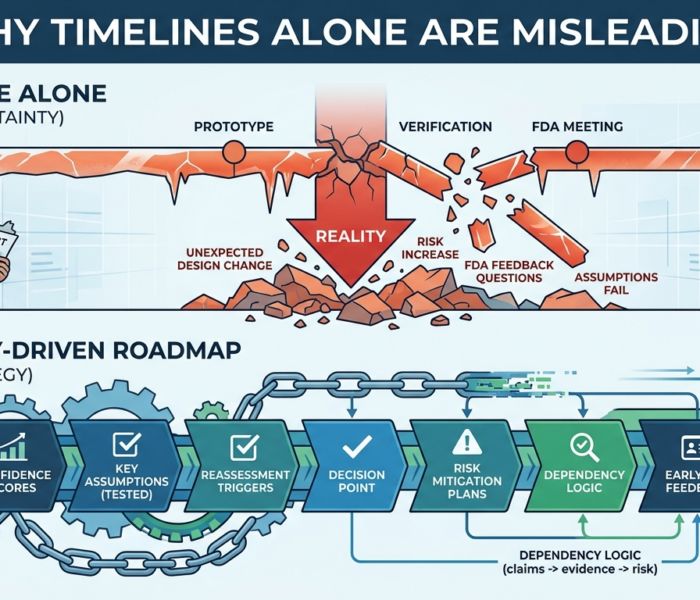
The timeline looked brilliant. Prototype by June.Verification by September.FDA meeting by November.Submission next year. Clean. Sharp. Reassuring. Then reality showed up. One design change delayed testing.A […]
April 14, 2026
Published by admin on April 14, 2026
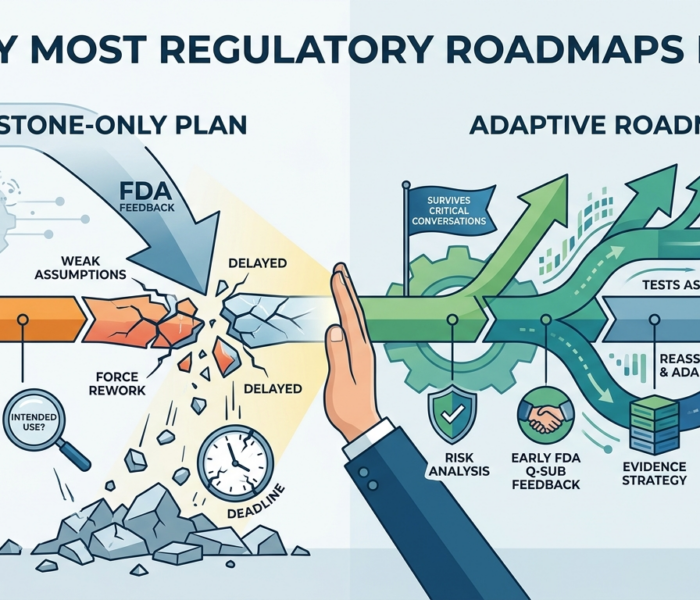
Everything looks fine until FDA responds. The roadmap is on track. The milestones are moving. The team is hitting deadlines. Then the feedback arrives. FDA wants […]