March 27, 2026
Published by admin on March 27, 2026
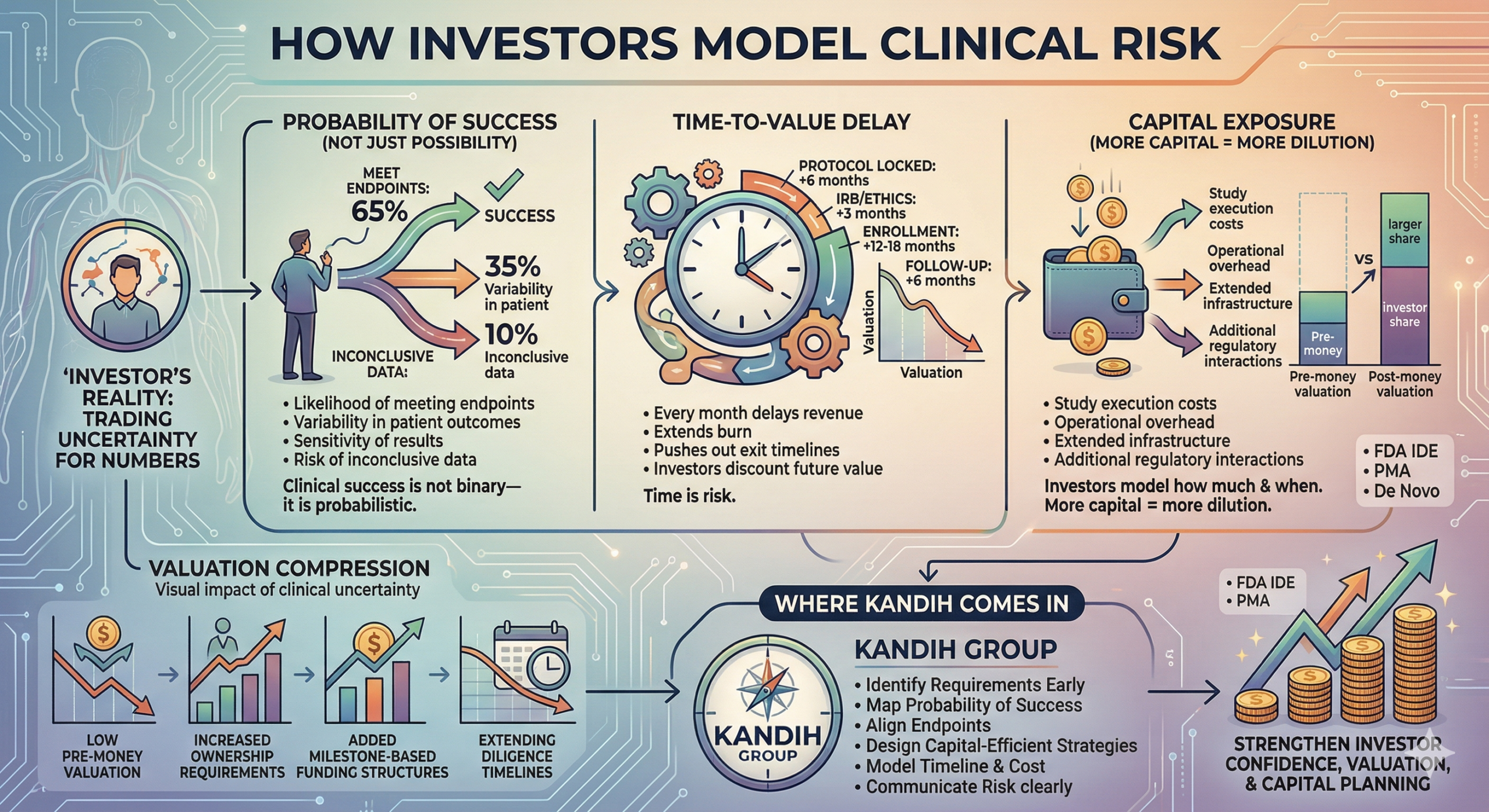
startup had everything investors usually want—strong prototype, clear market, experienced team. Then one question changed the entire conversation: “What’s your clinical risk?” The room shifted. The valuation dropped.
The timeline stretched.
The deal slowed.
March 26, 2026
Published by admin on March 26, 2026
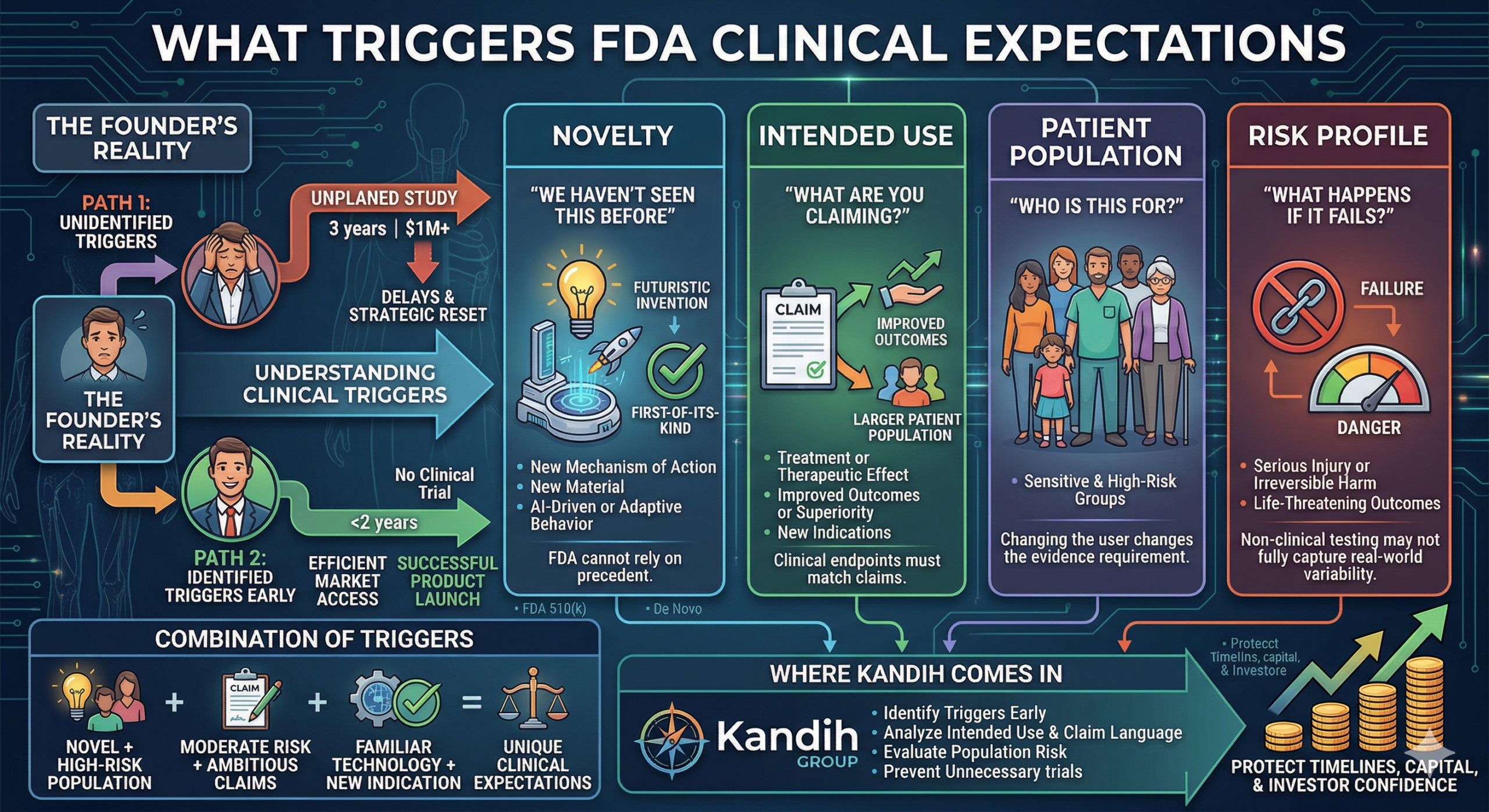
A founder once celebrated getting FDA feedback:
“Clinical data will be required.” It sounded like progress. Six months later, they were back in the market raising emergency capital. Same device. Same team.
But now their burn rate had doubled—and their valuation had dropped.
March 25, 2026
Published by admin on March 25, 2026
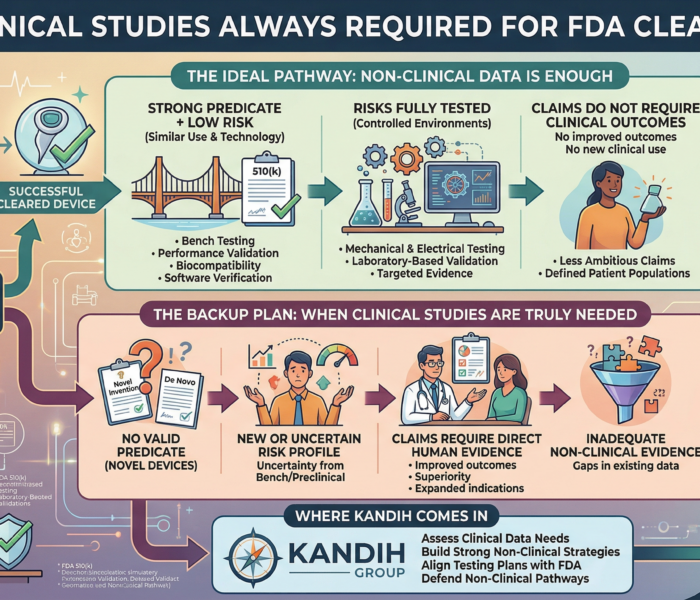
Hook:Two companies built nearly identical devices. One reached market in under two years with no clinical trial.The other spent three years running a costly study they […]
March 24, 2026
Published by admin on March 24, 2026
A founder once delayed their entire product launch by 18 months—planning a clinical trial they never actually needed. The device worked. The science was solid. The team had funding. What they didn’t have was clarity. So they defaulted to the safest assumption:
“FDA will probably require a clinical study.”