March 24, 2026
Published by admin on March 24, 2026
A founder once delayed their entire product launch by 18 months—planning a clinical trial they never actually needed. The device worked. The science was solid. The team had funding. What they didn’t have was clarity. So they defaulted to the safest assumption:
“FDA will probably require a clinical study.”
March 23, 2026
Published by admin on March 23, 2026
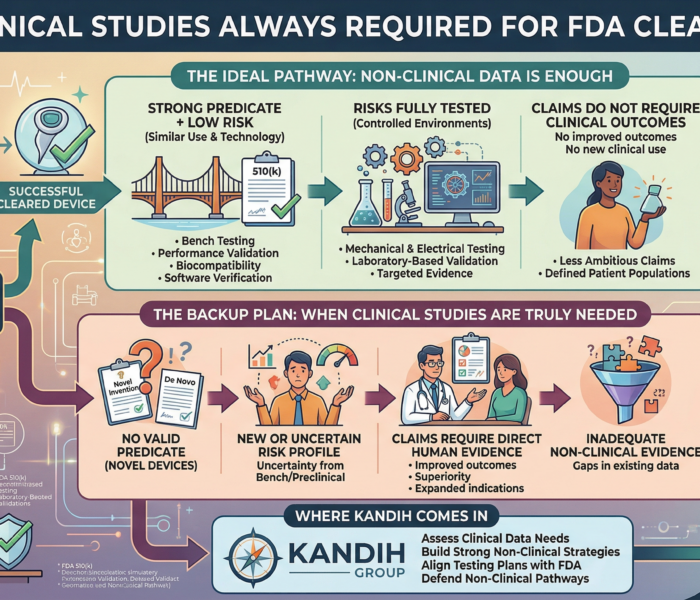
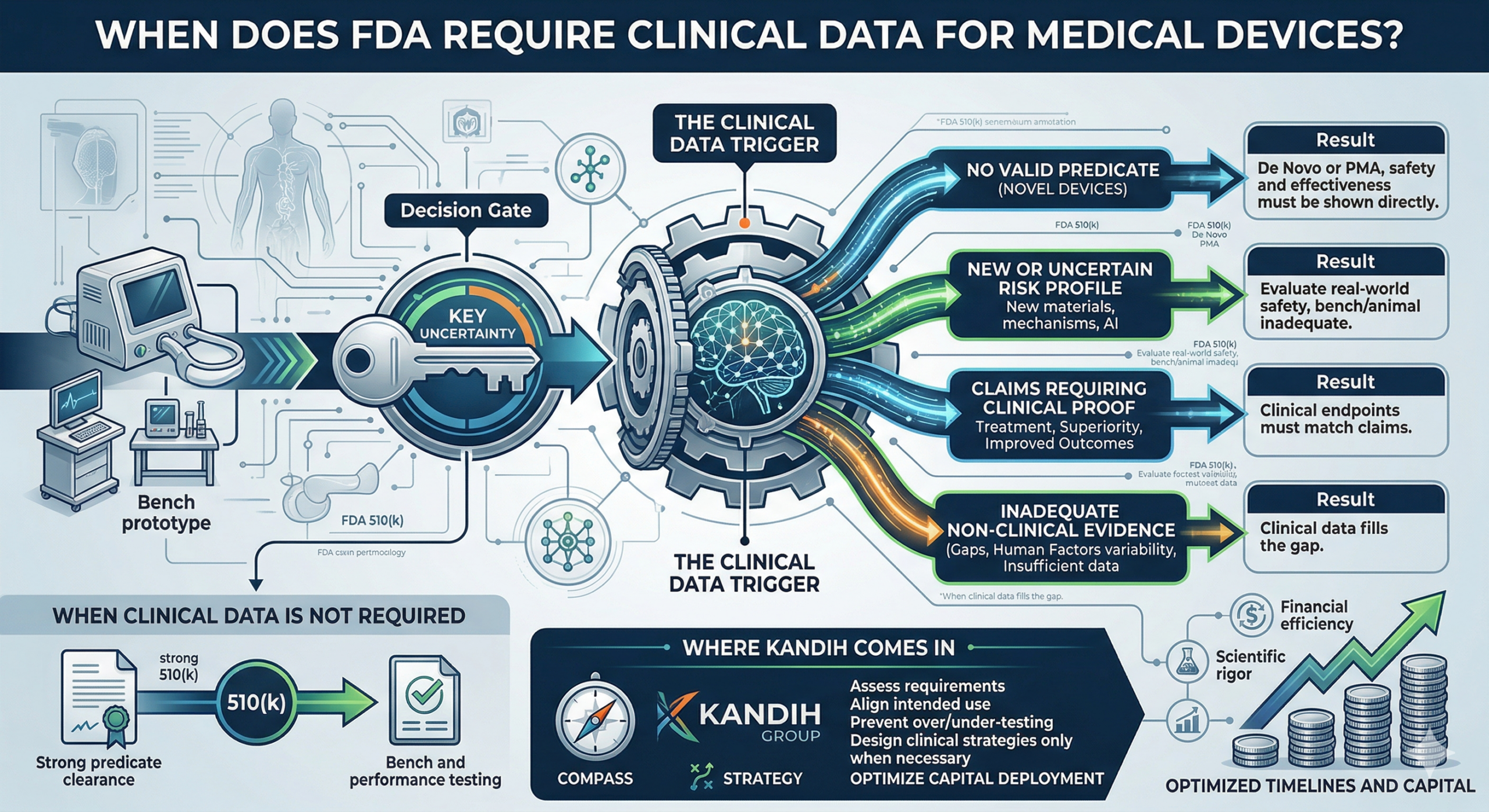
Short answer: the FDA requires clinical data when risk, uncertainty, or lack of precedent cannot be addressed through non-clinical testing. Clinical trials are not automatic.
They are triggered. And most founders misunderstand when that trigger actually happens. The Story Most Founders Live Through A founder builds a promising device.
The prototype works. Bench testing looks strong. Early feedback is positive
March 20, 2026
Published by admin on March 20, 2026
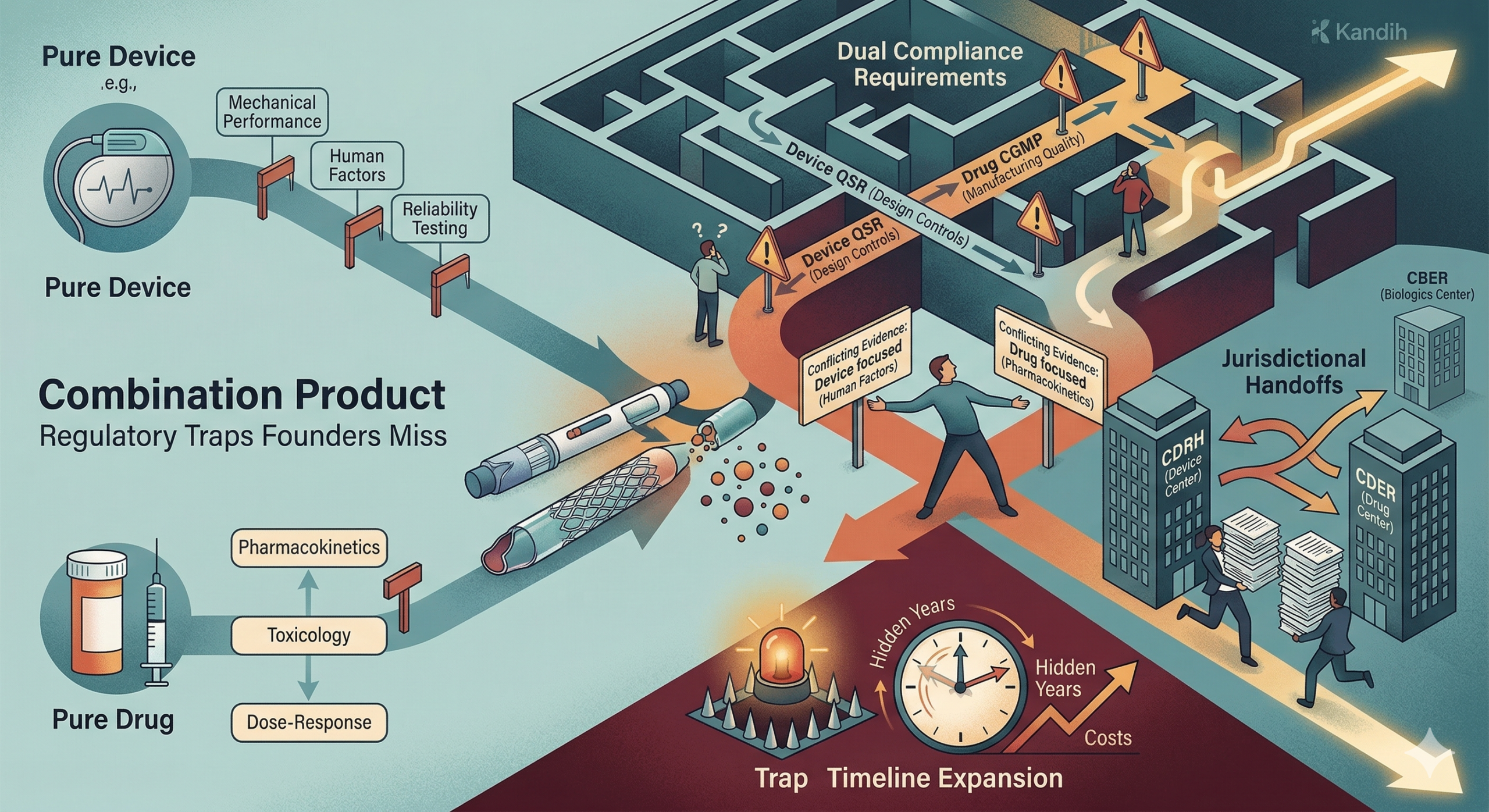
Short answer: combination products are often harder to develop than pure devices or pure drugs because they must satisfy multiple regulatory frameworks at the same time. Many founders assume that combining a device with a drug simply means choosing one regulatory pathway. In reality, combination products often require dual compliance, complex evidence strategies, and coordination across different parts of the U.S. Food and Drug Administration.
March 19, 2026
Published by admin on March 19, 2026
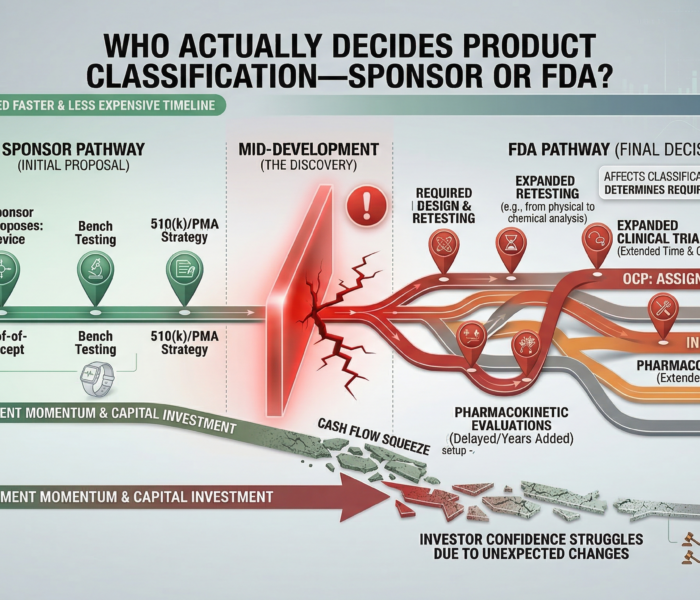
Short answer: the sponsor proposes a classification, but the final decision belongs to the FDA. Many companies assume they can simply decide whether their product is a device, drug, or combination product. In practice, the U.S. Food and Drug Administration determines the final regulatory classification. This matters because classification determines which regulatory center reviews the product, what evidence is required, and how long development may take.