March 18, 2026
Published by admin on March 18, 2026
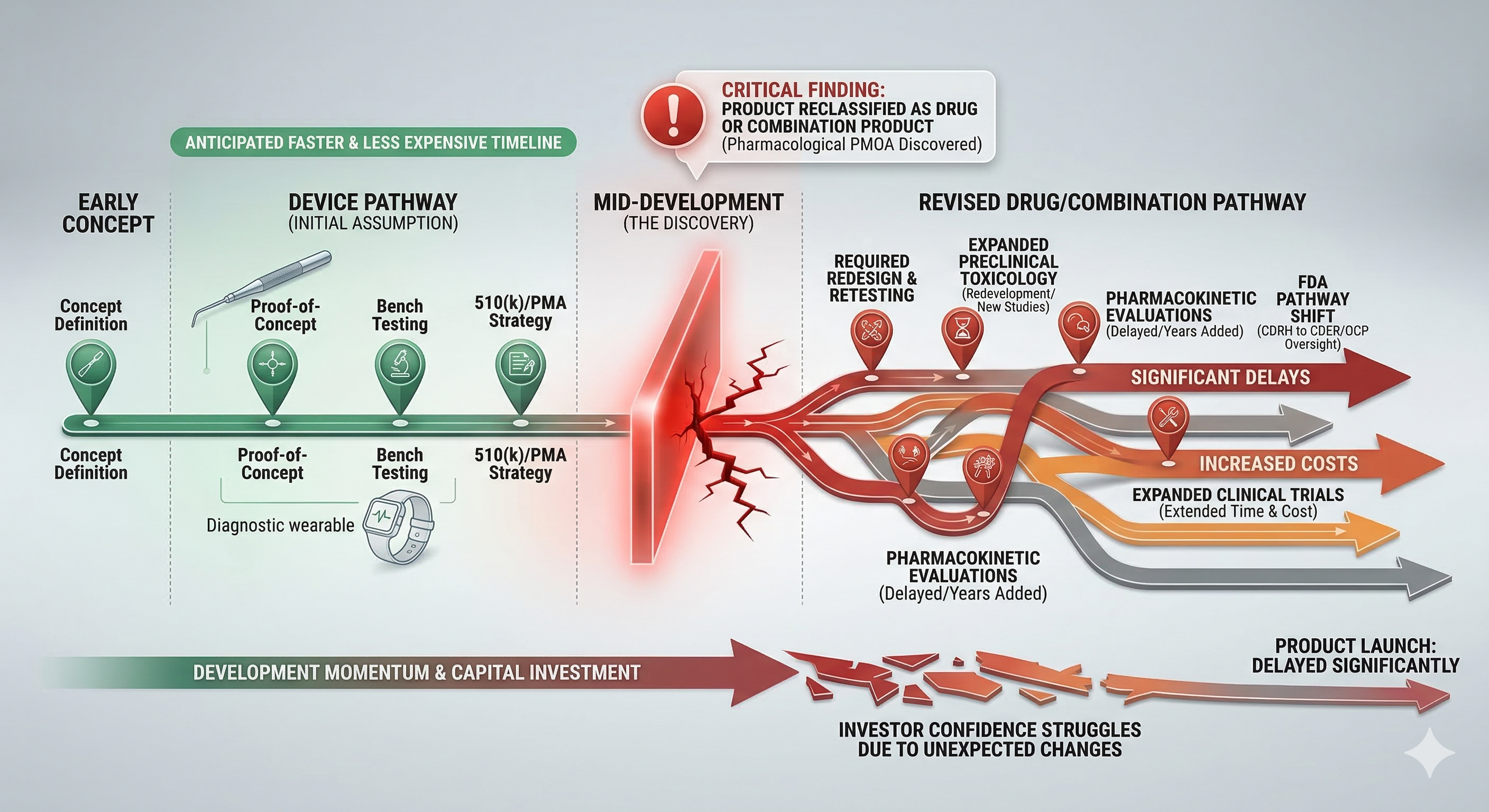
Short answer: product misclassification rarely causes immediate failure—but it quietly destroys timelines later. Many companies begin development assuming their product is a medical device because that pathway often appears faster and less expensive. Months—or even years—later, they discover the product functions more like a drug or combination product
March 17, 2026
Published by admin on March 17, 2026
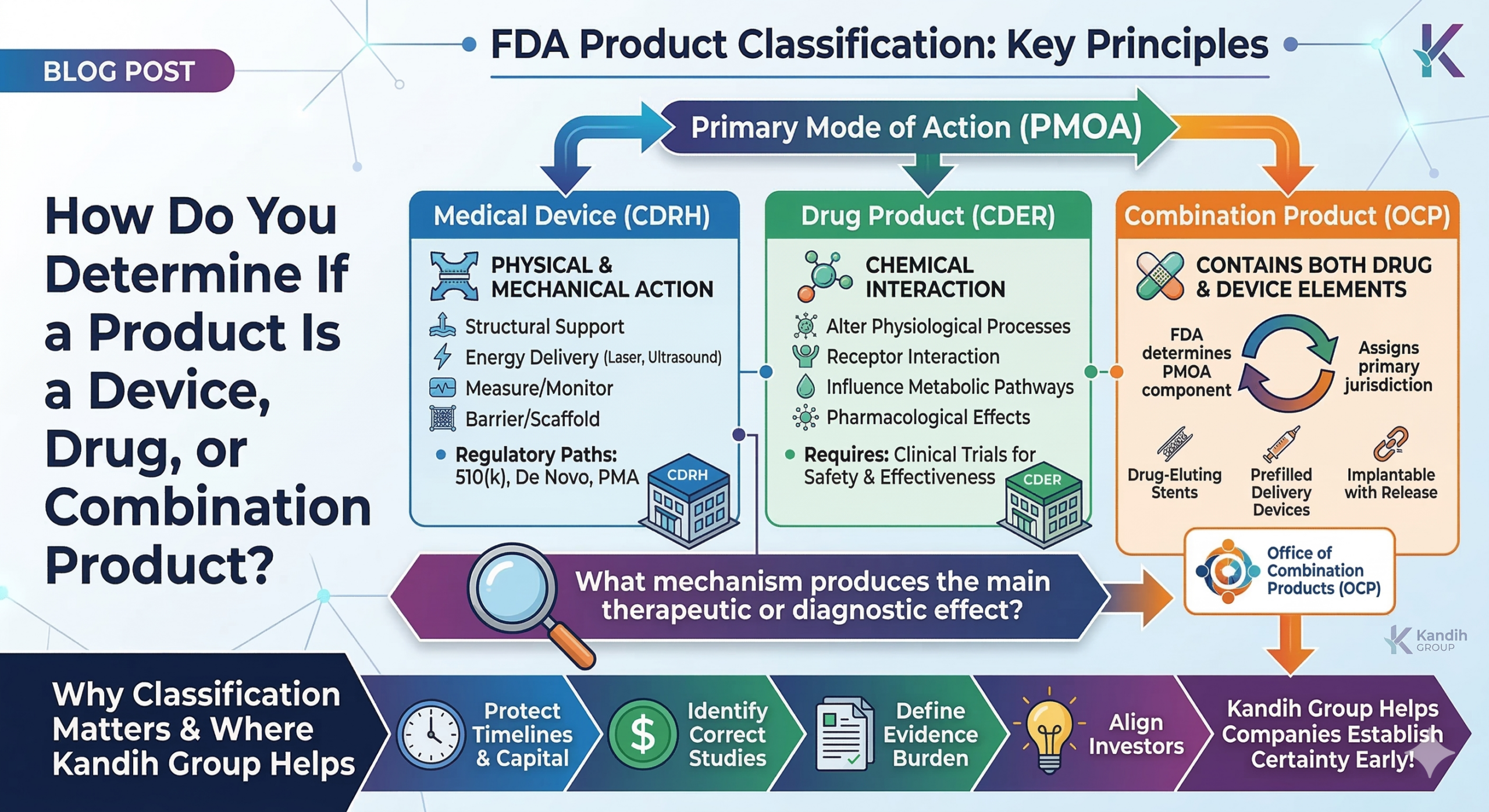
Short answer: the classification depends on the product’s primary mode of action (PMOA)—how the product achieves its main intended effect in the body. This determination matters because it decides which center of the U.S. Food and Drug Administration will regulate the product and what type of evidence will be required.
March 16, 2026
Published by admin on March 16, 2026
By Harriet Kamendi | Regulatory Toxicologist | Kandih Group Imagine you are a founder. You have compelling efficacy data, a clean mechanismof action, and enough runway […]
March 13, 2026
Published by admin on March 13, 2026
Short answer: Pre-Submissions (Pre-Subs) often fail not because the science is weak, but because the wrong regulatory questions were asked. Many companies approach a Pre-Submission thinking the goal is to prove the device works. In reality, the goal is to align the development plan with the expectations of the U.S. Food and Drug Administration before major investment decisions are made