Uncategorized
February 13, 2026
Published by admin on February 13, 2026
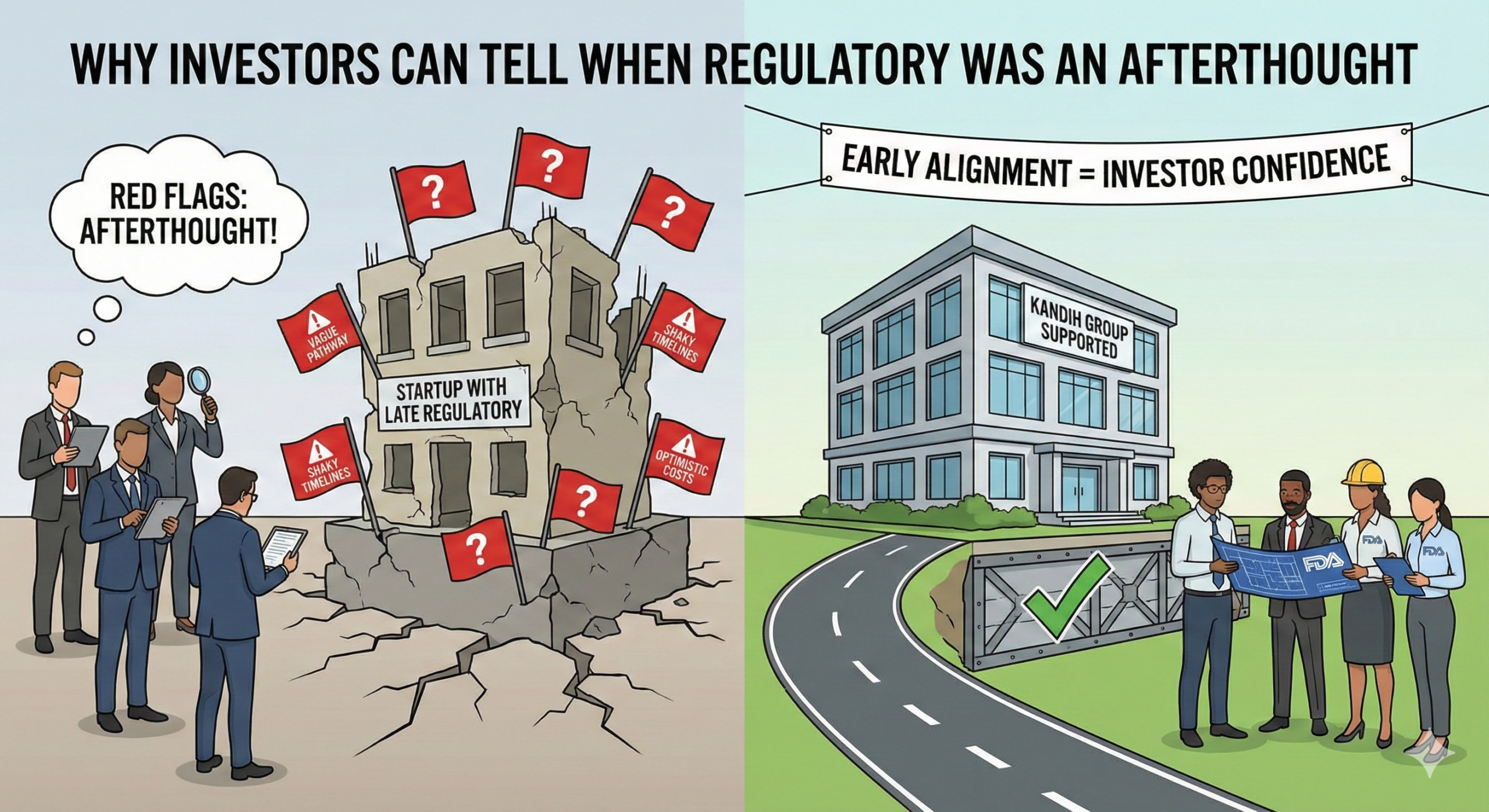
Hard reality: experienced investors can tell—often within minutes—when regulatory strategy was bolted on late instead of built in early. You don’t need to say “we figured […]
February 6, 2026
Published by admin on February 6, 2026
How rushing to close issues creates shallow solutions—and repeat failures Speed is often praised in regulated environments. Fast responses look decisive. Quick closures look efficient. Leaders […]
February 5, 2026
Published by admin on February 5, 2026
(And What Regulators Are Actually Looking For) The Everyday Problem A safety issue shows up during testing.The team investigates, applies a fix, and documents the decision. […]
February 4, 2026
Published by admin on February 4, 2026
Paper compliance isn’t the same as real-world performance On paper, everything can look perfect. The testing plan is written.The documentation is complete.The boxes are checked.The audit […]