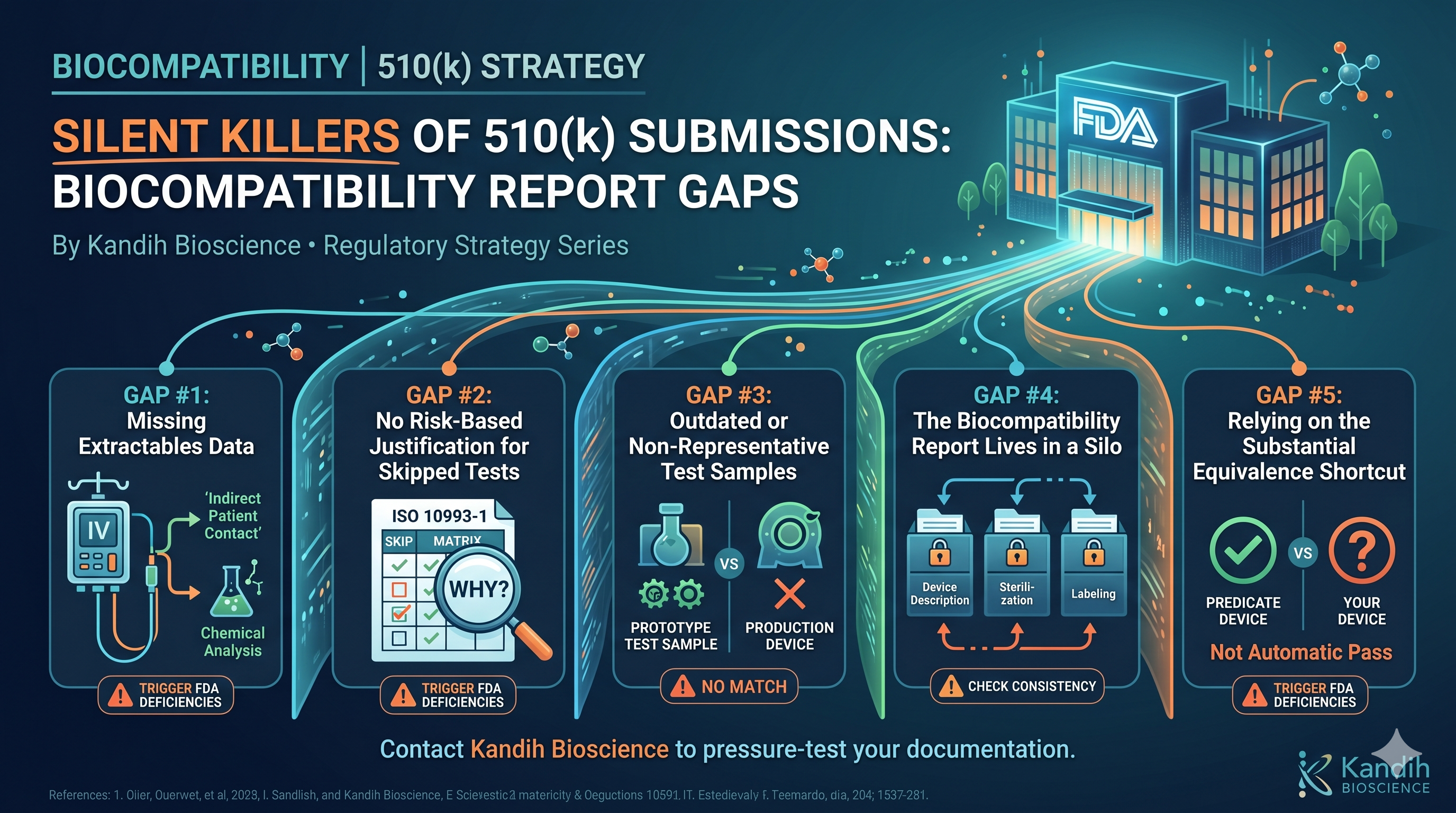
Why Speed Is the Enemy of Good Fixes

Why the Same Problems Keep Showing Up in Medical Device Development
February 5, 2026
When Should Regulatory Strategy Actually Start for a Medical Device?
February 9, 2026How rushing to close issues creates shallow solutions—and repeat failures
Speed is often praised in regulated environments. Fast responses look decisive. Quick closures look efficient. Leaders feel reassured when issues disappear from dashboards.
But here’s the uncomfortable reality: the faster you rush to close an issue, the more likely it is to come back.
In quality systems, study execution, testing programs, and regulatory operations, speed frequently works against real improvement. It creates fixes that look good on paper, satisfy immediate pressure, and quietly fail in practice.
Why urgency feels right—but leads to weak fixes
When an issue surfaces, the pressure is immediate:
Audits are coming
Studies are ongoing
Timelines are at risk
Leadership wants answers
Urgency creates a narrow focus: “How do we close this—now?”
That mindset is dangerous because it changes the goal. The objective quietly shifts from solving the problem to making the problem disappear.
Once that happens, quality becomes transactional instead of structural.
The hidden costs of rushing
1. Closure replaces understanding
Good fixes require understanding why something happened—not just what happened.
Rushed investigations often stop at:
“Human error”
“Procedure not followed”
“Training gap”
These explanations are fast, familiar, and easy to document. They also avoid the harder work of asking:
Was the process realistic under real workload?
Were roles and handoffs clear?
Was the decision point obvious at the time?
Did competing priorities create risk?
Speed favors surface-level answers. Root causes take time—and pressure discourages time.
2. Fast fixes rely on people, not systems
The quickest fixes almost always depend on behavior:
“Be more careful”
“Double-check next time”
“Follow the SOP more closely”
These solutions are appealing because they don’t require redesign. No workflow changes. No system investment. No uncomfortable conversations.
But behavior-based fixes are fragile. They assume people will perform perfectly under the same pressure that caused the issue in the first place.
That’s not risk reduction. That’s hope.
3. Urgency rewards paperwork, not effectiveness
Under time pressure, teams gravitate toward actions that are:
Easy to write
Easy to approve
Easy to close
Retraining, SOP updates, and reminder communications check all those boxes.
What they don’t guarantee is:
Fewer errors
More consistent execution
Earlier detection of problems
Speed rewards documentation over design—and the system stays exactly as vulnerable as before.
4. Quick closure creates false confidence
When an issue is closed quickly, it creates a sense of safety:
“We handled it.”
But often:
The same failure mode still exists
The same shortcuts are still tempting
The same assumptions remain untested
Risk hasn’t been reduced. It’s just been reclassified as “resolved.”
That false confidence is dangerous because it delays real improvement—and allows repeat issues to feel surprising when they shouldn’t be.
A common real-world example: rushed corrective actions
Consider a testing deviation during a development program.
The response is fast:
Root cause: protocol not followed
Action: retrain staff
Prevention: remind team to review procedures
The CAPA is closed on time.
But nothing changes in how the work is actually done:
The protocol is still overly complex
Critical steps are still buried in dense text
Reviews still happen at the end, not at decision points
Timelines are still unrealistic
Three months later, a similar issue appears.
The system didn’t fail again.
It was never fixed.
Why slowing down feels risky—but isn’t
Many organizations fear that slowing down will:
Delay projects
Increase scrutiny
Signal weakness
In reality, slowing down strategically does the opposite.
It:
Reduces repeat issues
Decreases cumulative rework
Builds regulator confidence
Saves time over the full lifecycle
The goal is not to move slowly.
The goal is to move deliberately.
What good fixes actually require
Strong fixes take slightly more time upfront because they:
Examine how work actually happens
Challenge assumptions baked into procedures
Identify pressure points and failure modes
Change the system so errors are harder to repeat
Good fixes answer this question honestly:
“If nothing else changes, can this still happen?”
If the answer is yes, the fix isn’t finished—no matter how fast it was closed.
Where Kandih comes in
Kandih helps teams slow down just enough to fix the right thing.
We understand the pressure to move fast. We also understand the cost of shallow solutions. Our role is to help organizations balance urgency with effectiveness.
Specifically, Kandih helps teams:
Separate time pressure from root cause
Identify whether a proposed fix actually reduces risk
Stress-test corrective actions against real-world use
Redesign studies, testing plans, and workflows instead of piling on reminders
Close issues in a way that prevents recurrence—not just documentation gaps
We don’t add bureaucracy.
We remove fragility.
By taking a structured, system-focused approach, Kandih helps teams close issues with confidence, not crossed fingers.
The real trade-off: speed now vs time later
Rushing saves time today.
Good fixes save time for years.
Organizations that chase speed often find themselves:
Reopening the same CAPAs
Rewriting the same procedures
Explaining the same issues to new auditors
Training the same teams on the same mistakes
Organizations that slow down just enough:
See fewer repeat issues
Build stronger, simpler systems
Spend less time firefighting
Gain credibility with regulators and partners
Bottom line
Speed feels responsible.
But speed without depth creates fragile fixes.
If your priority is closing issues quickly, expect them to return quietly and repeatedly.
Kandih helps teams pause at the right moment—long enough to understand the real problem, fix the real system, and move forward stronger.
Because the fastest way forward is fixing it right the first time.
References
FDA – Quality System Regulation Preamble (CAPA Effectiveness)
FDA explicitly states that corrective actions must address root cause and prevent recurrence—not just close issues quickly.
https://www.fda.gov/files/drugs/published/Overview-of-Quality-System-Regulation.pdf
ICH Q10 – Pharmaceutical Quality System
Requires evaluation of corrective action effectiveness and discourages superficial fixes driven by timelines.
https://database.ich.org/sites/default/files/Q10_Guideline.pdf
ISO 9001:2015 – Clause 10.2 (Corrective Action)
Mandates that organizations evaluate whether actions taken actually prevent recurrence—not just implement them quickly.
https://www.iso.org/standard/62085.html
Reason, J. (2000). Human Error: Models and Management. BMJ
Foundational paper explaining why rushed, behavior-based fixes fail when systems remain unchanged.
https://www.bmj.com/content/320/7237/768
ASQ – Root Cause Analysis: Why Quick Fixes Fail
Explains how urgency leads to symptom treatment instead of true root cause correction.
https://asq.org/quality-resources/root-cause-analysis
ISPE – Risk-Based Approach to Quality and Investigations
Discusses the danger of timeline-driven investigations and superficial corrective actions.
https://ispe.org/publications/guidance-documents
HSE (UK) – Investigating Accidents and Incidents
Demonstrates that rushed investigations lead to missed root causes and repeat failures.
https://www.hse.gov.uk/pubns/books/hsg245.htm
FDA – Investigations Operations Manual (IOM)
FDA inspection guidance emphasizing thorough investigations over rapid closure under time pressure.