March 2, 2026
Published by admin on March 2, 2026
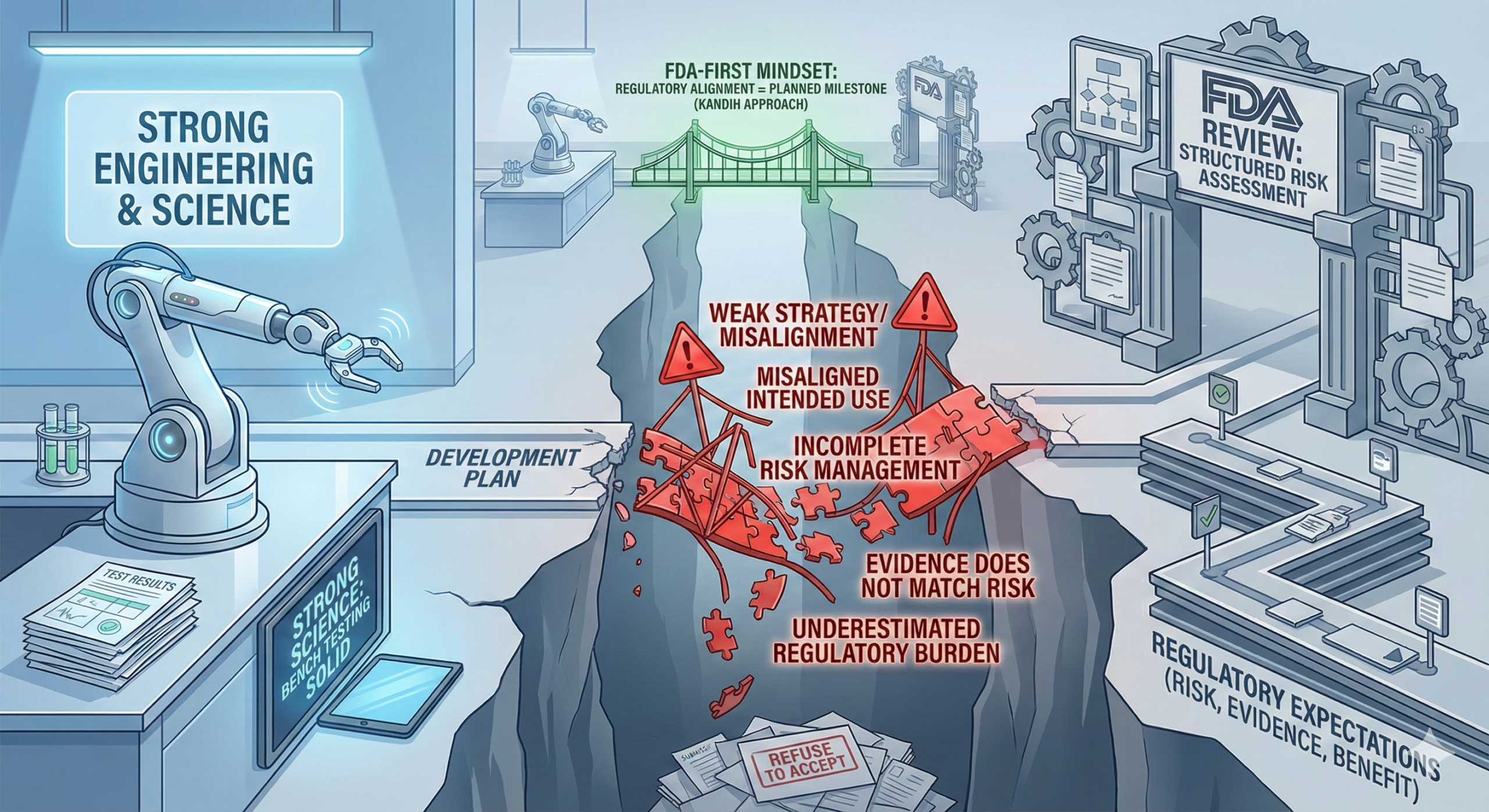
Direct answer: most medical devices do not fail FDA review because the science is weak. They fail because the strategy was weak. In many cases, the engineering works. The bench testing looks solid. The prototype performs as expected. But the submission fails because the development plan was not aligned with how the U.S. Food and Drug Administration evaluates risk, evidence, and benefit
February 27, 2026
Published by admin on February 27, 2026
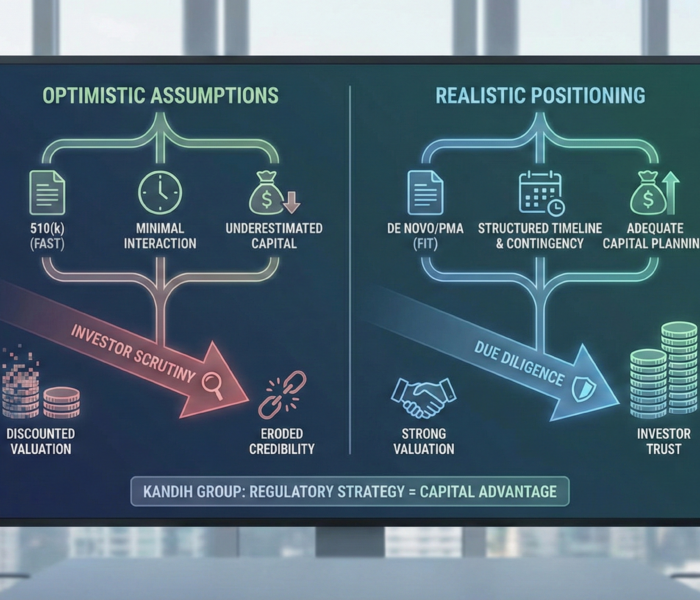
Short answer: investors do not just look at your FDA pathway. They look at how realistic your pathway assumptions are. If your regulatory strategy sounds optimistic, […]
February 26, 2026
Published by admin on February 26, 2026
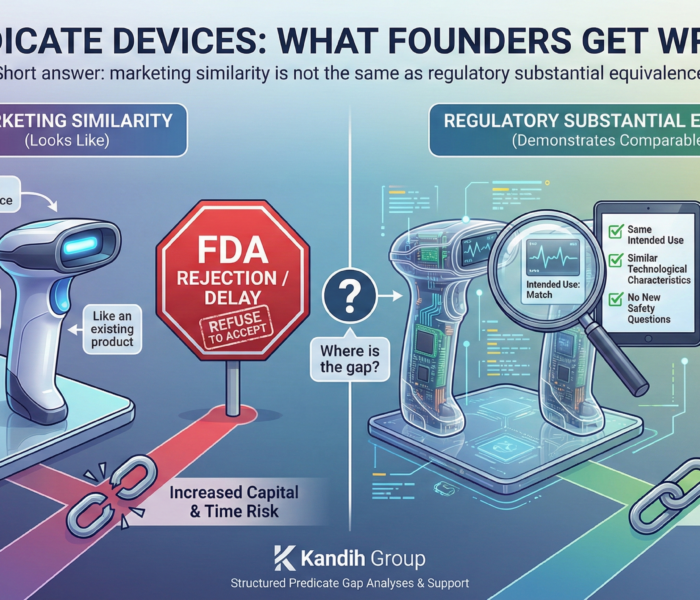
Short answer: marketing similarity is not the same as regulatory substantial equivalence. Many founders believe that if their device “looks like” something already on the market, they can file a 510(k). That assumption is one of the most common regulatory mistakes in medical device development.
February 25, 2026
Published by Daniel Ndiki on February 25, 2026
We often hear that modern science has "doubled" the human lifespan. We look at the statistics from 1840—where life expectancy was a mere 40 years—and compare them to our 80-year average today, assuming we’ve fundamentally reengineered human biology.
But the data tells a different, more sobering story.