February 24, 2026
Published by admin on February 24, 2026
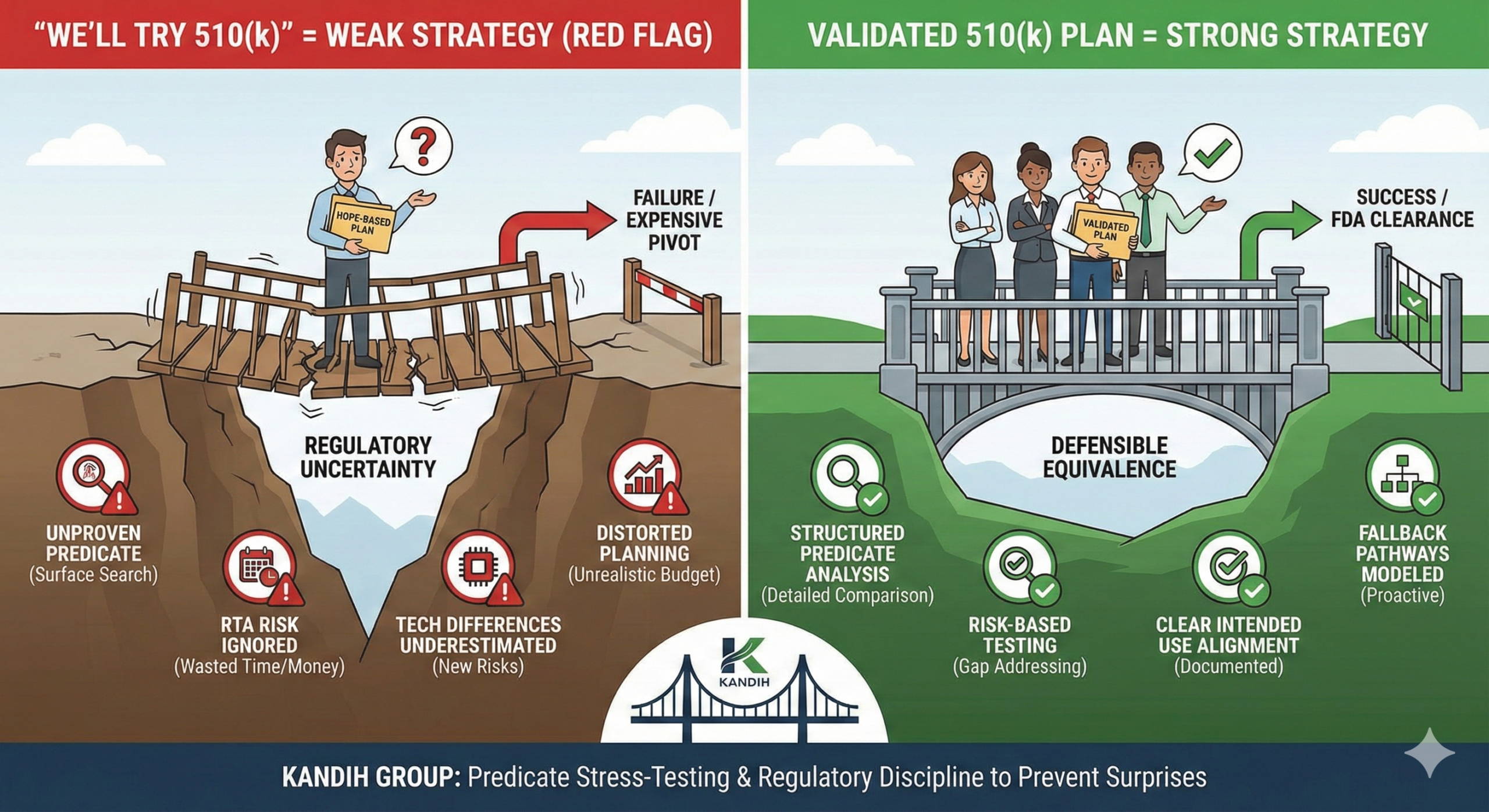
Short answer: when a team says, “We’ll try 510(k),” it usually means the regulatory strategy is not fully developed. A 510(k) is not something you “try.” It is something you qualify for. Under the framework of the U.S. Food and Drug Administration, the 510(k) pathway depends on a very specific standard: substantial equivalence to a legally marketed predicate device. If that logic is not solid, the pathway collapses.
February 23, 2026
Published by admin on February 23, 2026
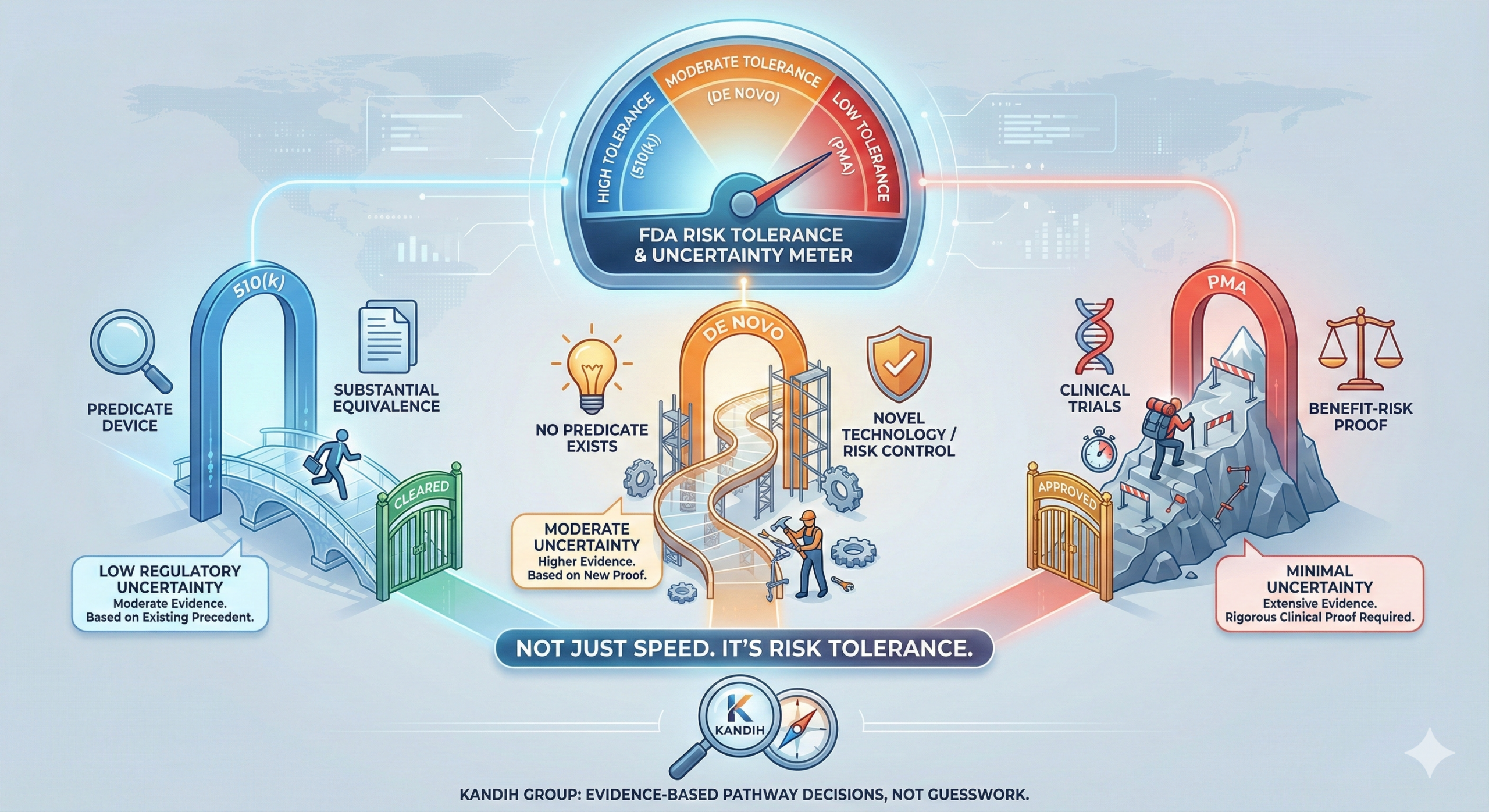
Short answer: the real difference between 510(k), De Novo, and PMA is not speed. It is risk tolerance. Each pathway reflects how much uncertainty the U.S. Food and Drug Administration is willing to accept—and how much evidence they require to reduce that uncertainty.
February 20, 2026
Published by admin on February 20, 2026
Short answer: an FDA regulatory pathway is not just a compliance decision. It is a portfolio risk signal. For investors, the difference between 510(k), De Novo, […]
February 19, 2026
Published by admin on February 19, 2026
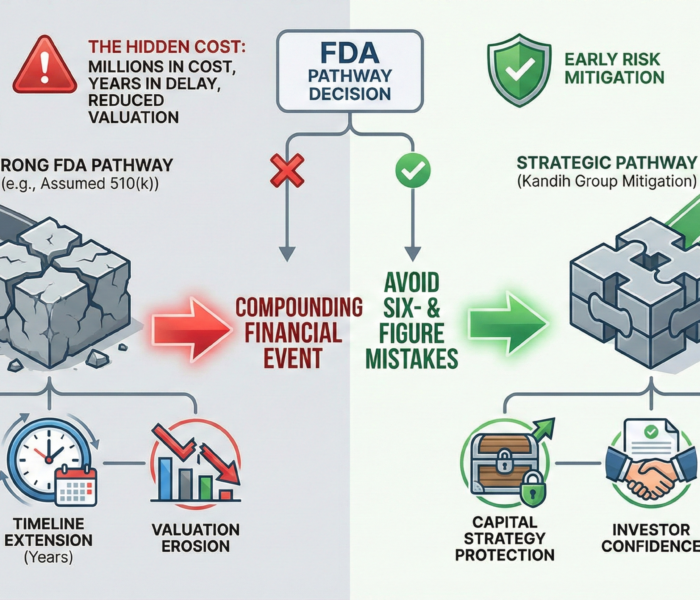
Direct answer: choosing the wrong FDA pathway does not just delay your launch. It can add millions in cost, years in delay, and materially reduce company valuation. Most pathway mistakes happen early—when teams assume a 510(k) is viable without fully pressure-testing risk, intended use, or technological differences.