February 18, 2026
Published by admin on February 18, 2026
Short answer: incorrect FDA pathway assumptions don’t usually fail at the beginning. They fail when investors start asking hard questions. Early on, optimism hides regulatory gaps. During investor diligence, those gaps get exposed. That’s because regulatory pathway decisions—510(k), De Novo, or PMA—are not just technical. They define your timeline, capital needs, and exit strategy.
February 17, 2026
Published by admin on February 17, 2026
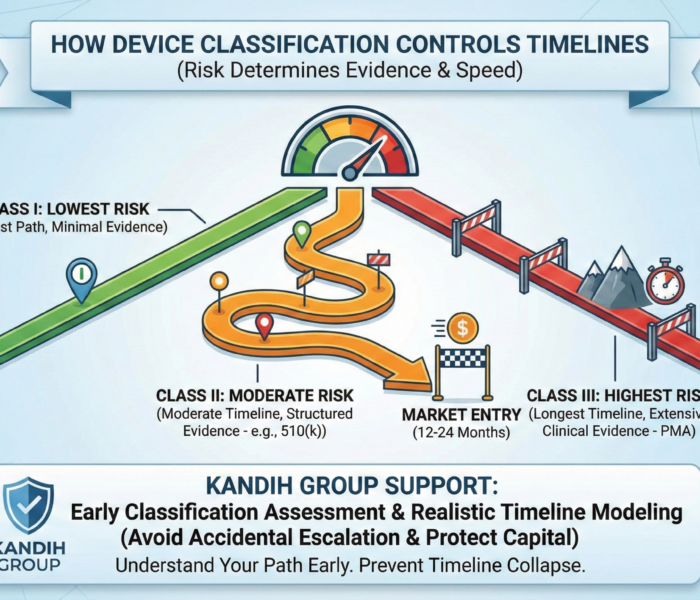
Straight answer: device classification controls how much evidence you must generate—and that controls how fast (or slow) you reach market. If you don’t understand your classification early, your timeline is not a plan. It’s a guess. Under the framework of the U.S. Food and Drug Administration, medical devices are placed into three risk-based classes: Class I, Class II, and Class III. The higher the risk, the heavier the evidence burden—and the longer the timeline.
February 16, 2026
Published by admin on February 16, 2026
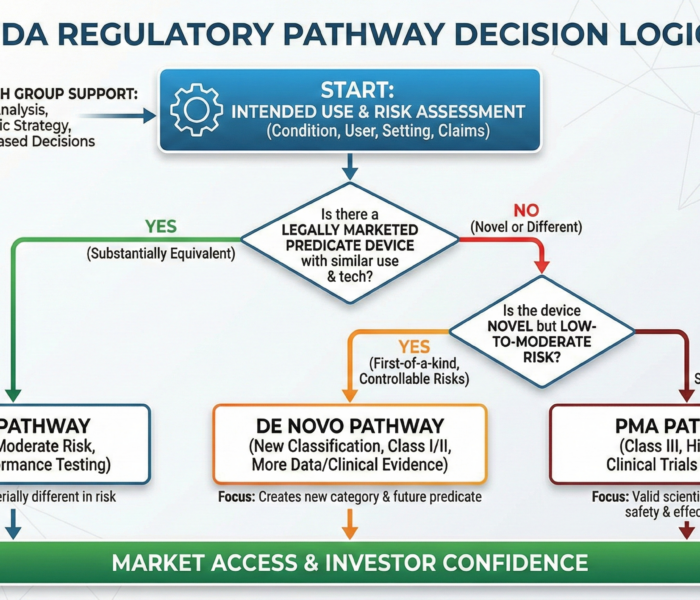
Short answer: the FDA regulatory pathway for medical devices depends on risk, intended use, and whether a similar device already exists. The three main pathways are: 510(k) – for devices that are substantially equivalent to an existing legally marketed device De Novo – for novel, low-to-moderate risk devices with no predicate
February 13, 2026
Published by admin on February 13, 2026
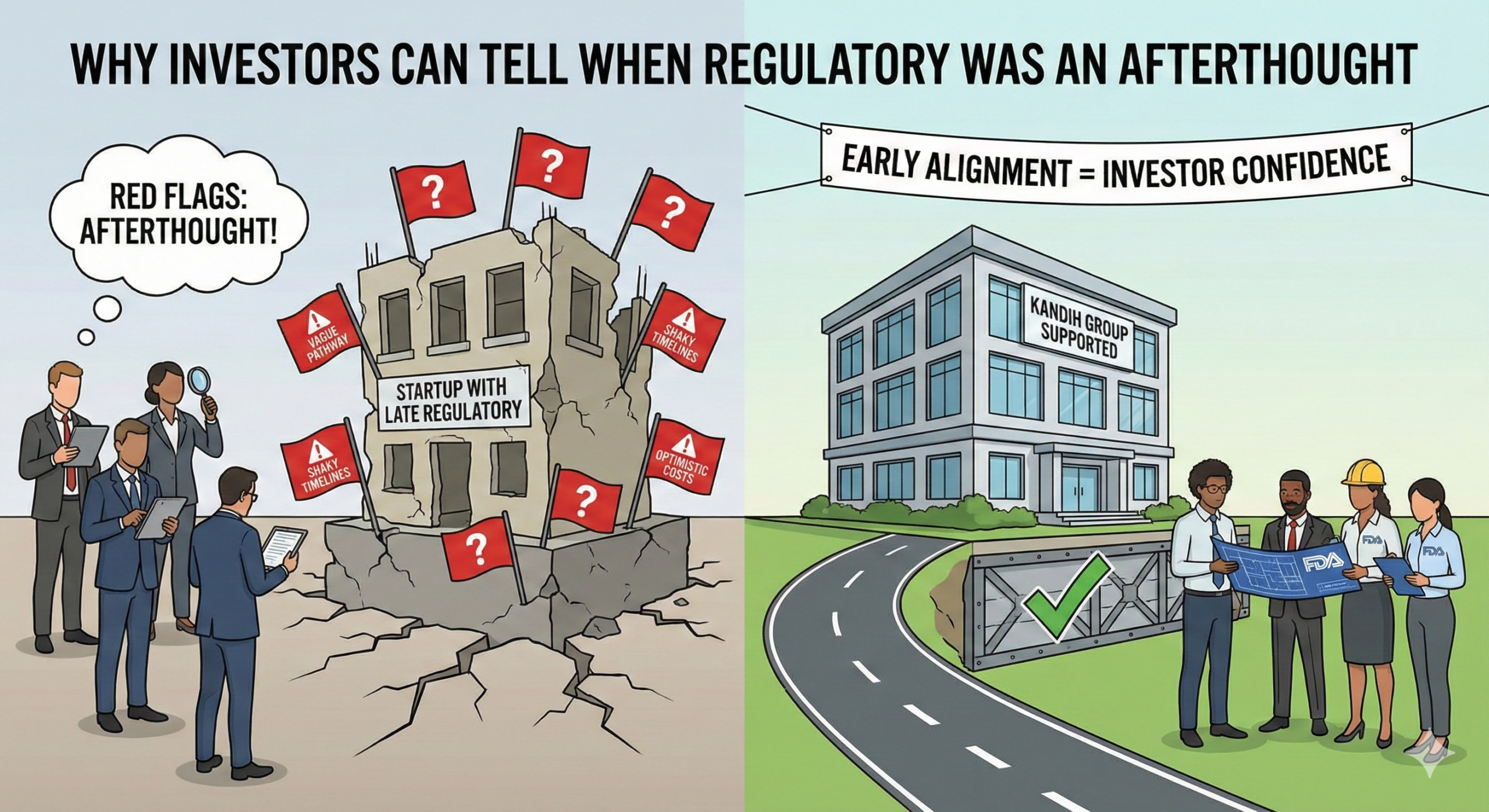
Hard reality: experienced investors can tell—often within minutes—when regulatory strategy was bolted on late instead of built in early. You don’t need to say “we figured […]