What Is the FDA Regulatory Pathway for Medical Devices?
Why Investors Can Tell When Regulatory Was an Afterthought
February 13, 2026
How Device Classification Controls Timelines
February 17, 2026Short answer: the FDA regulatory pathway for medical devices depends on risk, intended use, and whether a similar device already exists.
The three main pathways are:
510(k) – for devices that are substantially equivalent to an existing legally marketed device
De Novo – for novel, low-to-moderate risk devices with no predicate
PMA (Premarket Approval) – for high-risk devices requiring clinical evidence
But definitions are not enough. What matters is decision logic.
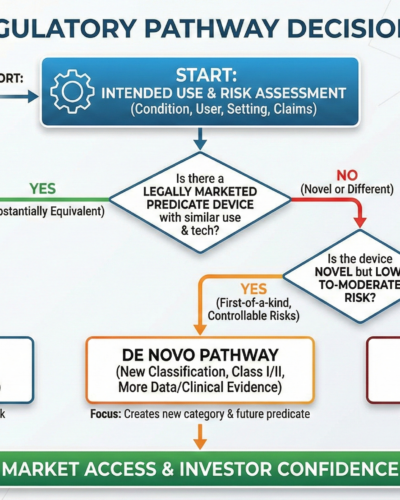
The Decision Logic Behind FDA Pathways
The U.S. Food and Drug Administration does not choose pathways based on innovation level. They choose based on risk and existing regulatory history.
Here is how the decision actually works.
Step 1: What Is the Intended Use?
This is the starting point. Everything flows from intended use.
FDA first asks:
What condition does the device address?
Who will use it?
In what setting?
What claims are being made?
Intended use determines risk classification. And classification drives pathway.
Step 2: Is There a Predicate Device?
If a legally marketed device exists with:
The same intended use
Similar technological characteristics
No new safety concerns
Then a 510(k) pathway may be possible.
510(k) Logic:
If your device can show substantial equivalence to a predicate, you do not need to prove safety and effectiveness from scratch. You need to show it is not materially different in risk profile.
Typical profile:
Moderate-risk devices (Class II)
Bench and performance testing
Sometimes limited clinical data
If no appropriate predicate exists, 510(k) is not an option.
Step 3: Is the Device Low-to-Moderate Risk but Novel?
If there is no predicate, but the device presents low-to-moderate risk, the likely route is De Novo.
De Novo Logic:
If the device is new but risks can be mitigated through controls, FDA may create a new classification for it.
Typical profile:
First-of-a-kind technologies
Requires more data than 510(k)
Often includes clinical evidence
Longer review time
De Novo creates a new regulatory category. Future devices may then use it as a predicate.
Step 4: Is the Device High Risk?
If the device:
Sustains or supports life
Is implanted long-term
Presents significant potential harm if it fails
Then the pathway is likely PMA (Premarket Approval).
PMA Logic:
If risk is high, FDA requires valid scientific evidence, usually including well-controlled clinical trials.
Typical profile:
Class III devices
Extensive bench, animal, and clinical data
Most expensive and time-intensive pathway
Why Optimism Is Dangerous
Founders often choose pathways based on:
What they hope is easiest
What competitors did
What feels cheaper
FDA does not care about optimism.
They care about risk classification and evidence sufficiency.
Misclassifying your device early can result in:
Forced pathway changes
Repeated testing
12–24 months of delay
Millions in unexpected costs
The regulatory pathway is not a marketing decision. It is a risk decision.
Where Kandih Comes In
This is where Kandih Group supports founders and investors early.
Kandih helps teams:
Analyze intended use against FDA classification logic
Assess realistic predicate options
Identify hidden technological risk escalators
Evaluate whether De Novo is viable
Pressure-test whether PMA is unavoidable
Build pathway decisions based on risk, not optimism
Instead of guessing which pathway is easiest, teams make decisions grounded in regulatory reality.
That reduces surprises, protects capital, and improves investor confidence.
Bottom Line
The FDA regulatory pathway is not chosen.
It is determined by risk, intended use, and regulatory precedent.
Understanding the decision logic early prevents:
Costly pivots
Repeated testing
Investor hesitation
Strong regulatory strategy means choosing the right path the first time.
References
FDA – Classify Your Medical Device
https://www.fda.gov/medical-devices/overview-device-regulation/classify-your-medical-device
FDA – Premarket Notification 510(k)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-notification-510k
FDA – De Novo Classification Process
https://www.fda.gov/medical-devices/premarket-submissions/de-novo-classification-request
FDA – Premarket Approval (PMA)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-approval-pma