March 6, 2026
Published by admin on March 6, 2026
Short answer: FDA failure is not just a regulatory setback. It is a valuation event. When a medical device company receives a major deficiency letter, Refuse-to-Accept decision, or pathway escalation from the U.S. Food and Drug Administration, investors do not see a technical issue. They see risk materializing. And risk directly affects exits.
March 5, 2026
Published by admin on March 5, 2026
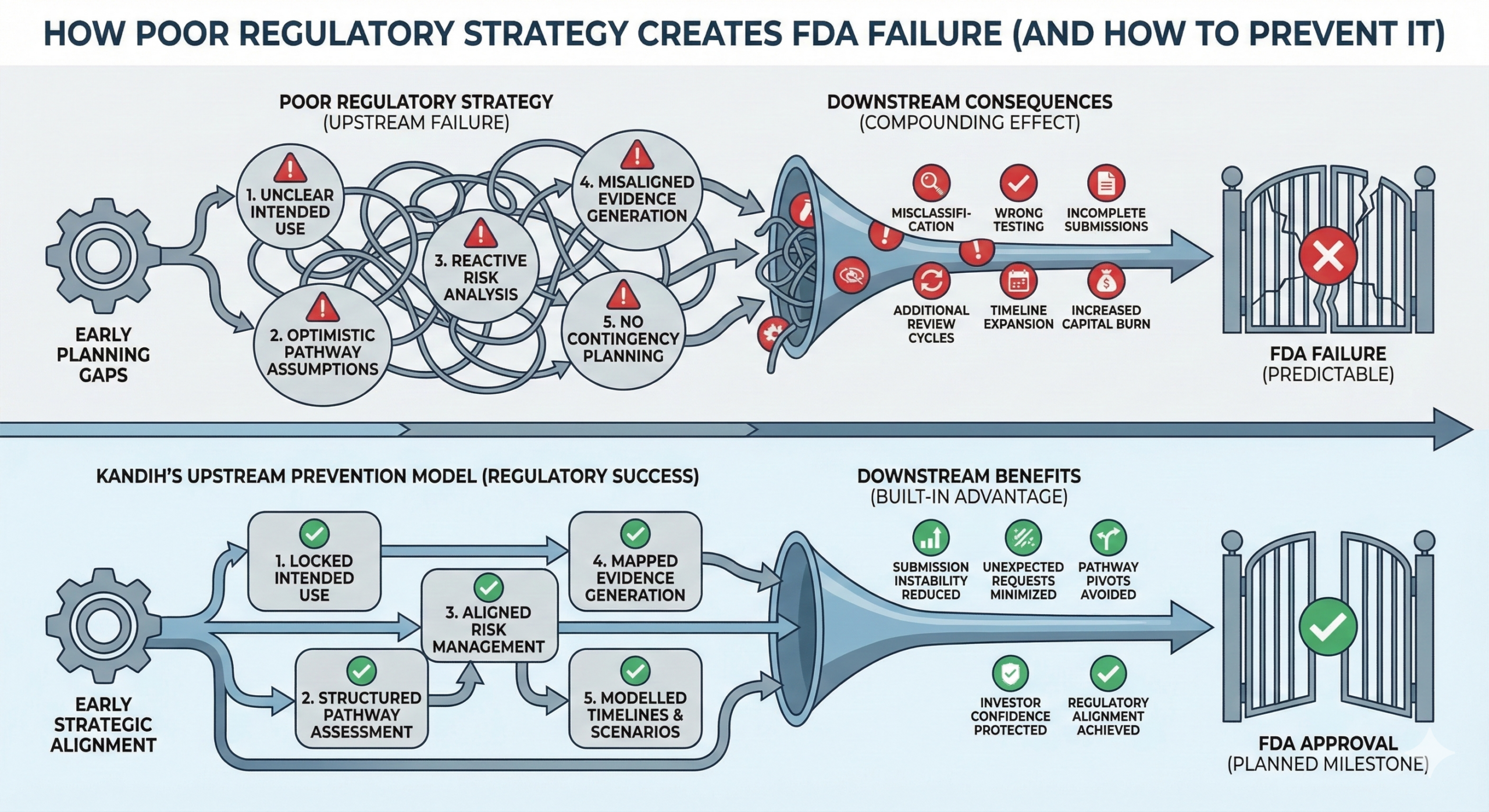
Short answer: most FDA failures are not created during review. They are created months—or years—earlier during poor planning. By the time a submission reaches the U.S. Food and Drug Administration, the outcome is often already predictable. FDA does not “surprise” companies. Companies surprise themselves by discovering too late that their strategy never aligned with regulatory expectations
March 4, 2026
Published by admin on March 4, 2026
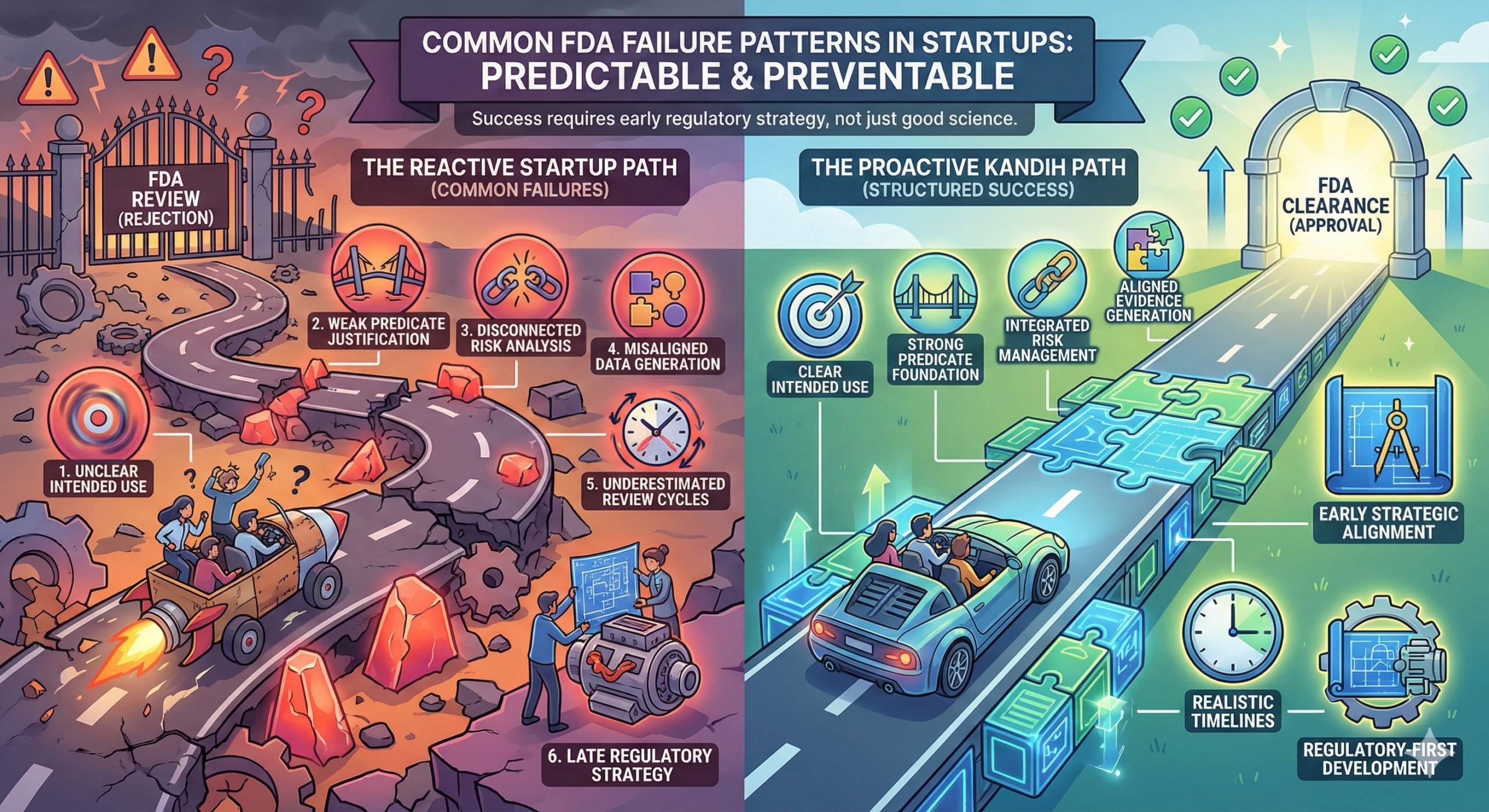
Short answer: most FDA submission failures follow predictable patterns. They are rarely random. They are rarely mysterious. And they are almost always preventable. Startups often assume failure happens because the science was weak. In reality, failure usually happens because the regulatory strategy was fragmented, optimistic, or reactive
March 3, 2026
Published by admin on March 3, 2026
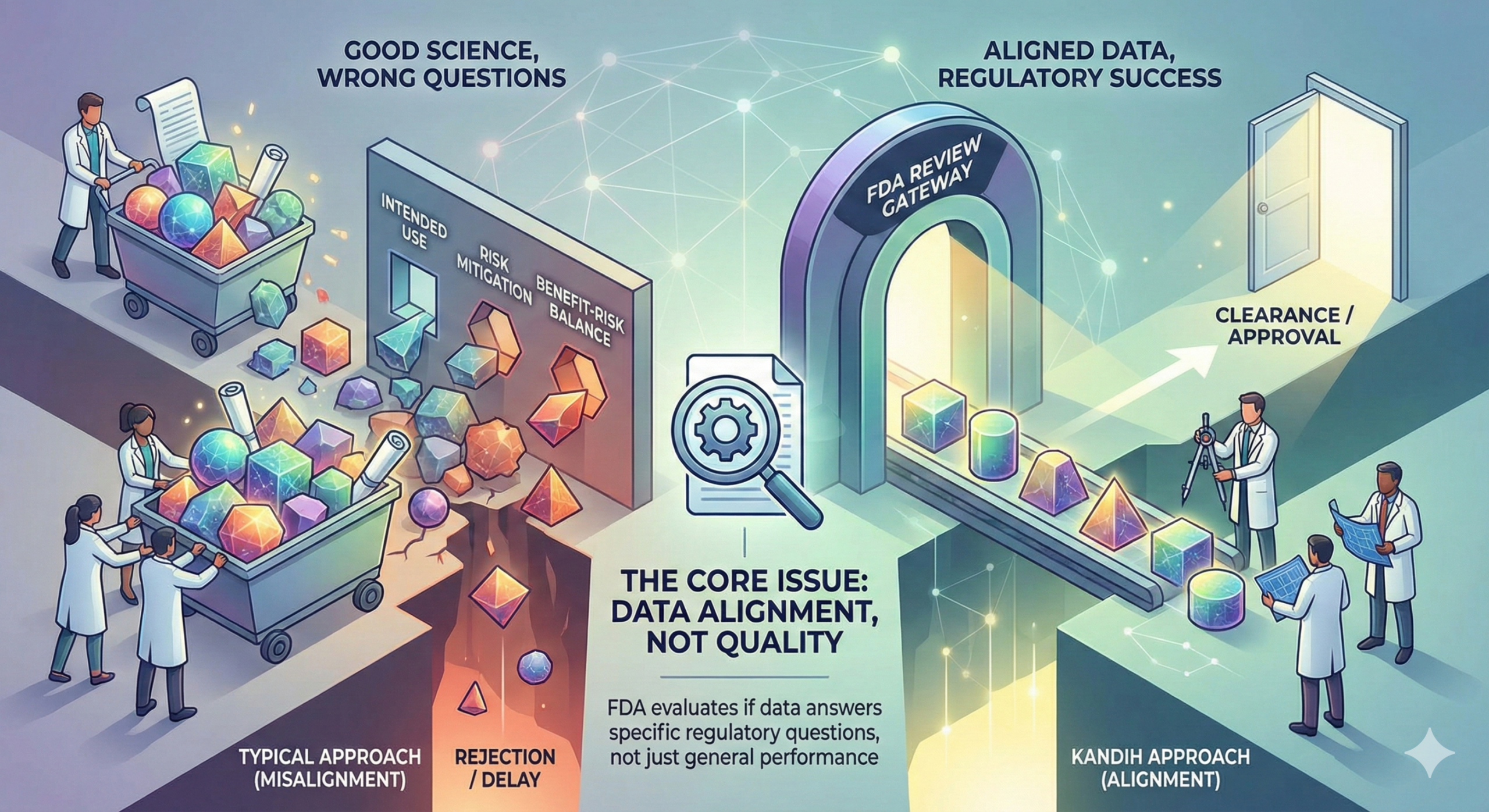
Short answer: FDA rejection is rarely about bad data. It is usually about answering the wrong question. Most companies that struggle during FDA review did generate data. They ran studies. They collected results. They invested money.