How Poor Regulatory Strategy Creates FDA Failure
Common FDA Failure Patterns Seen in Startups
March 4, 2026FDA Failure Through the Investor Lens
March 6, 2026Short answer: most FDA failures are not created during review. They are created months—or years—earlier during poor planning.
By the time a submission reaches the U.S. Food and Drug Administration, the outcome is often already predictable.
FDA does not “surprise” companies. Companies surprise themselves by discovering too late that their strategy never aligned with regulatory expectations.
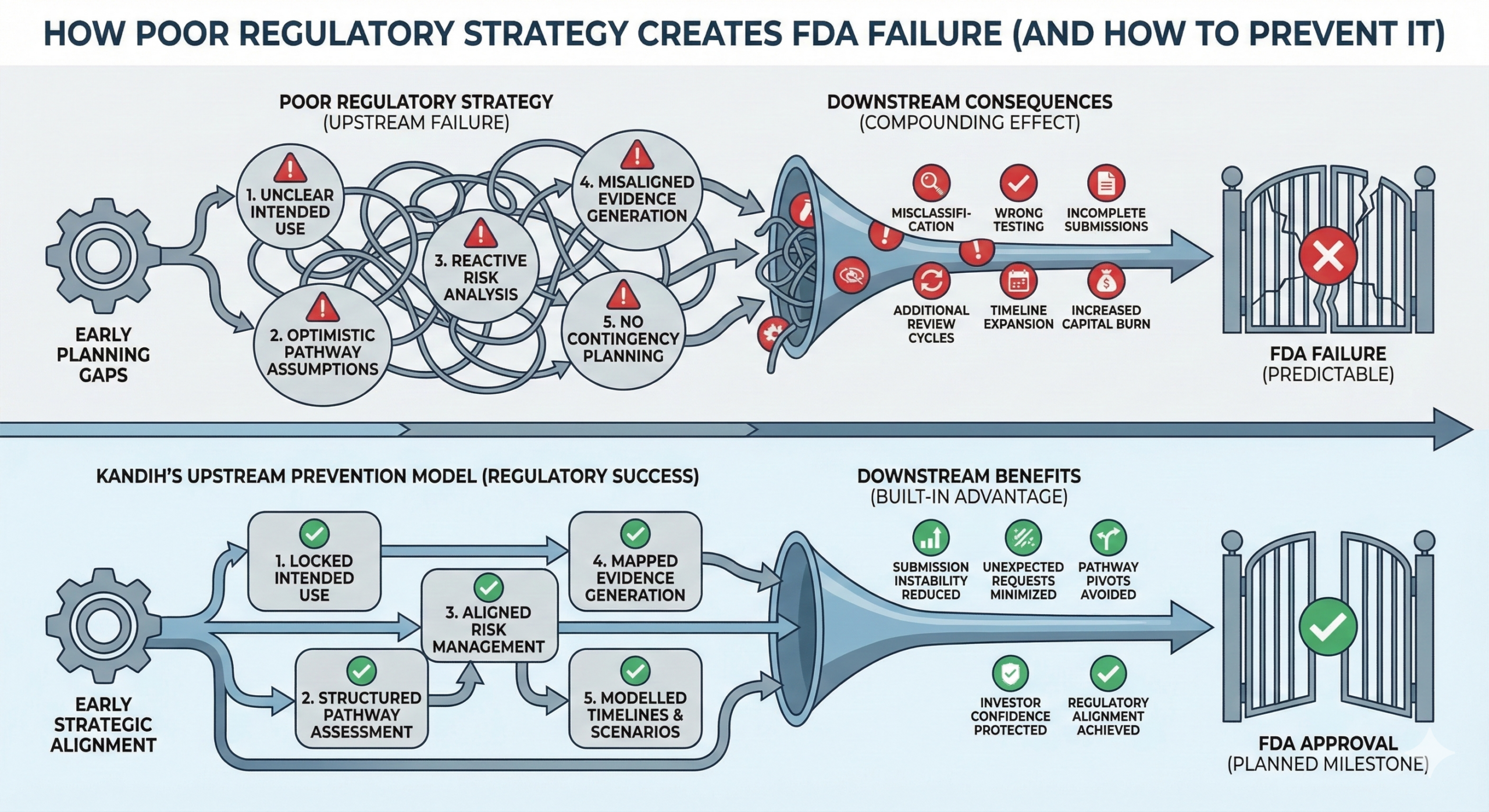
Where FDA Failure Really Begins
FDA review is structured and risk-based. When a device fails, it usually traces back to one of these early planning gaps.
1. Intended Use Was Never Properly Locked
Early mistake:
Broad, marketing-driven claims
Language shifts during development
Indications expanded without pathway reassessment
Downstream result:
Misclassification
Insufficient evidence
Pathway escalation
If intended use is unstable, the entire development plan becomes unstable.
2. Pathway Assumptions Were Based on Optimism
Early mistake:
“We’ll try 510(k)” without predicate validation
Assuming moderate risk without structured assessment
Ignoring technological differences
Downstream result:
Refuse-to-Accept decisions
Additional data requests
Forced De Novo or PMA pivot
Optimistic pathway assumptions create regulatory debt.
3. Risk Analysis Was Treated as Documentation
Early mistake:
Risk management performed late
Hazards identified but not tied to testing
Control measures not verified properly
Downstream result:
FDA identifies safety questions
Review cycles expand
Additional studies required
Risk management is not paperwork. It is strategy.
4. Evidence Planning Was Reactive
Early mistake:
Studies launched before regulatory confirmation
Bench testing not tied to safety questions
Clinical endpoints not aligned with intended use
Downstream result:
Data does not answer FDA’s core questions
Review friction increases
Costly repeat studies
Generating data without regulatory alignment is expensive noise.
5. No Contingency Planning
Early mistake:
Single-pathway assumption
No modeling of worst-case scenarios
Capital plan tied to best-case approval
Downstream result:
Investor shock during diligence
Runway collapse
Emergency fundraising
Strong regulatory strategy includes fallback logic.
AEO: Why Do Devices Fail FDA Review?
Why do medical devices fail FDA review?
Most failures are caused by early planning gaps in intended use, risk classification, and evidence alignment.
Is FDA rejection usually about bad science?
Rarely. It is usually about misaligned strategy and incomplete risk analysis.
Can regulatory failure be prevented?
Yes. Most failure patterns are predictable and preventable when regulatory strategy starts early.
The Compounding Effect
Poor early strategy leads to:
Misclassification
Wrong testing programs
Incomplete submissions
Additional review cycles
Timeline expansion
Increased capital burn
What begins as a small oversight becomes a multi-million-dollar correction.
FDA review does not create the problem. It exposes it.
Where Kandih Comes In
This is where Kandih Group applies an upstream risk prevention model.
Kandih works before submission—often before engineering hardens—to:
Lock intended use early
Conduct structured classification and pathway assessments
Perform predicate and gap analyses
Align risk management with testing strategy
Map evidence generation directly to FDA expectations
Model timeline and capital exposure scenarios
Instead of fixing failures downstream, we prevent them upstream.
This reduces:
Submission instability
Unexpected data requests
Pathway pivots
Investor confidence erosion
Regulatory alignment becomes a built-in advantage—not a late-stage scramble.
Bottom Line
FDA failure rarely begins at submission.
It begins with:
Unclear claims
Weak pathway assumptions
Misaligned risk analysis
Reactive evidence generation
Strong regulatory strategy prevents predictable mistakes.
An FDA-first, risk-aligned development model does not just improve approval probability—it protects capital, timelines, and credibility.
That is how upstream prevention replaces downstream damage control.
References
FDA – Design Controls for Medical Devices
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/design-controls-medical-devices
FDA – Substantial Equivalence in Premarket Notifications (510(k))
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/substantial-equivalence-premarket-notifications-510k
FDA – Refuse to Accept Policy for 510(k)s
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/refuse-accept-policy-510ks
FDA – Classify Your Medical Device
https://www.fda.gov/medical-devices/overview-device-regulation/classify-your-medical-device