Why FDA Treats CAPA as a Risk-Control System, Not a QA Task

Disseminated Intravascular Coagulation – Causes
December 19, 2025
CAPA Failures as Evidence of a Broken Risk-Control System
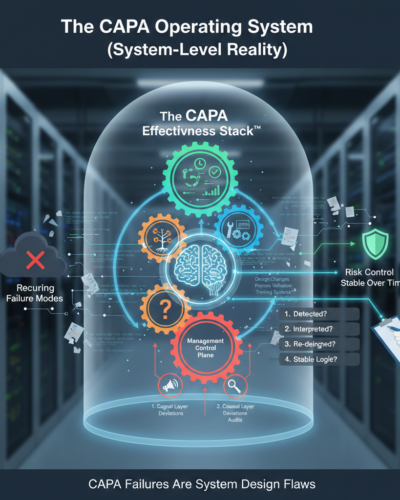
January 13, 2026The CAPA Operating System (System-Level Reality)
Most CAPA programs don’t fail because of weak documentation.
They fail because they were never architected as systems.
Organizations routinely design CAPA as a QA artifact—a form, a workflow, a closure event. FDA inspects something very different: a live, end-to-end risk-control system that proves an organization can detect, absorb, and eliminate recurring failure modes over time.
That gap explains a familiar paradox:
Spotless CAPA files. Repeated 483s for ineffective corrective action.
From an inspection standpoint, CAPA is not a task. It is the central nervous system of the quality management architecture.
How FDA Actually Inspects CAPA
FDA inspectors do not start with CAPA forms.
They start with signals—complaints, deviations, OOS/OOT, adverse events, audit findings—and walk those signals through your CAPA operating system to answer four questions:
Was the risk detected?
Was it correctly interpreted at the system level?
Was the system re-designed to control it?
Can you prove the control is stable over time?
CAPA records are evidence artifacts, not the system itself.
When CAPAs “close” on paper but risk pathways remain open, FDA interprets that as loss of management control, not a clerical issue—explicitly consistent with FDA expectations and ICH Q10 principles.
The CAPA Risk-Loop Architecture
CAPA is best understood as a closed-loop control system—not a checklist.
The CAPA Risk Loop
A continuous feedback architecture connecting:
1. Signal Inputs
Complaints, nonconformances, deviations, OOS/OOT, audit findings, supplier issues, postmarket data—feeding a unified detection funnel, not siloed inboxes.
If signals don’t converge, risk never propagates.
2. Risk Interpretation Engine
Structured causality analysis that evaluates:
Patient impact
Recurrence probability
Systemic scope
—not reflexive defaults to “operator error” or one-off explanations.
Misdiagnosed risk is misrouted risk.
3. Systemic Linkage Layer
Bi-directional integration with:
Design controls
Process validation
Change management
Supplier controls
Training systems
Digital QMS configuration
If CAPA actions don’t re-wire how risk flows, nothing was corrected.
4. Verification & Monitoring Loop
Time-bound, metric-driven confirmation that the risk pathway is:
Controlled
Stable
Trending appropriately
Temporary silence is not effectiveness.
5. Management Control Plane
Management review functioning as a decision engine, not a reporting meeting:
Resource allocation
Escalation
Portfolio-level risk prioritization
This is where FDA looks for executive ownership under QSR and ICH Q10.
When any layer is under-designed, the system leaks—and FDA reads the leak as a governance failure.
The CAPA Effectiveness Stack™️ (Named Framework)
Move beyond “good vs bad CAPA.”
Use a language FDA already understands—system performance.
The 4-Layer CAPA Effectiveness Stack
1. Signal Layer — Are we seeing risk?
Integrated intake and trending across complaints, deviations, audits, and suppliers.
Red flags FDA notices:
Issues re-coded after CAPA
Missing or superficial trend analysis
Product, site, or functional silos
2. Causality Layer — Do we understand risk?
Root cause analysis spanning:
Process
Design
Human factors
Materials
Environment
Supplier controls
Red flags:
“Training only”
“Isolated operator error”
No evidence alternatives were explored and ruled out
3. Control Layer — Did we re-design the system?
CAPA actions embedded into:
Design changes
Process controls
Supplier qualification
Digital QMS logic
Red flags:
SOP edits without system change
Fixes confined to one product, line, or site
4. Verification Layer — Can we prove stability?
Pre-defined effectiveness criteria with:
Metrics
Time windows
Trending logic
Aligned with FDA and ICH expectations for process performance and product quality monitoring.
Red flags:
Immediate CAPA closure
Qualitative statements like “no further issues observed”
No supporting data
System language you can use in any room:
“The issue isn’t this CAPA—the Control and Verification layers of your CAPA Effectiveness Stack are under-designed.”
CAPA Failures Are System Design Flaws
Most recurring CAPA findings map cleanly to architectural weaknesses:
Narrow root cause → Broken causality layer
Training-only actions → Unchanged control layer
Weak effectiveness checks → Missing verification layer
Isolated fixes → No propagation logic
Passive management review → Disabled control plane
FDA does not see these as execution mistakes.
They see them as structural inability to control risk.
What “Good” CAPA Systems Actually Share
Organizations that consistently clear inspections don’t have better forms—they have better architectures:
Risk-based governance: CAPA prioritization scales with patient impact and recurrence risk
Unified data spine: SOPs, training, deviations, CAPA, change control, and complaints are digitally linked
Outcome-anchored ownership: Owners are accountable for risk reduction metrics, not task closure
Management as system operators: Leadership uses CAPA trends to decide where to invest, redesign, or escalate
This is exactly how FDA and ICH Q10 define management responsibility—whether or not the organization realizes it.
References
1. U.S. Food and Drug Administration (FDA).
Quality System Regulation (QSR), 21 CFR Part 820 — §820.100 Corrective and Preventive Action.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-820
2. U.S. Food and Drug Administration (FDA).
Guidance for Industry: Quality Systems Approach to Pharmaceutical CGMP Regulations.
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/quality-systems-approach-pharmaceutical-current-good-manufacturing-practice-regulations
3. International Council for Harmonisation (ICH).
ICH Q10: Pharmaceutical Quality System.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8689590/
4. International Council for Harmonisation (ICH).
ICH Q9(R1): Quality Risk Management.
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/q9r1-quality-risk-management
5. FDA Compliance Program Guidance Manual (CPGM).
CPGM 7346.832 – Medical Device Quality System Inspection Technique (QSIT).
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-guides/quality-systems
6. FDA Warning Letters and Form FDA 483 Observations (Public Database).
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/compliance-actions-and-activities/warning-letters
7. ISO 13485:2016.
Medical devices — Quality management systems — Requirements for regulatory purposes.
https://www.iso.org/standard/59752.html
8. ISO 9001:2015.
Quality management systems — Requirements.
https://www.iso.org/standard/62085.html