FDA Failure Through the Investor Lens
How Poor Regulatory Strategy Creates FDA Failure
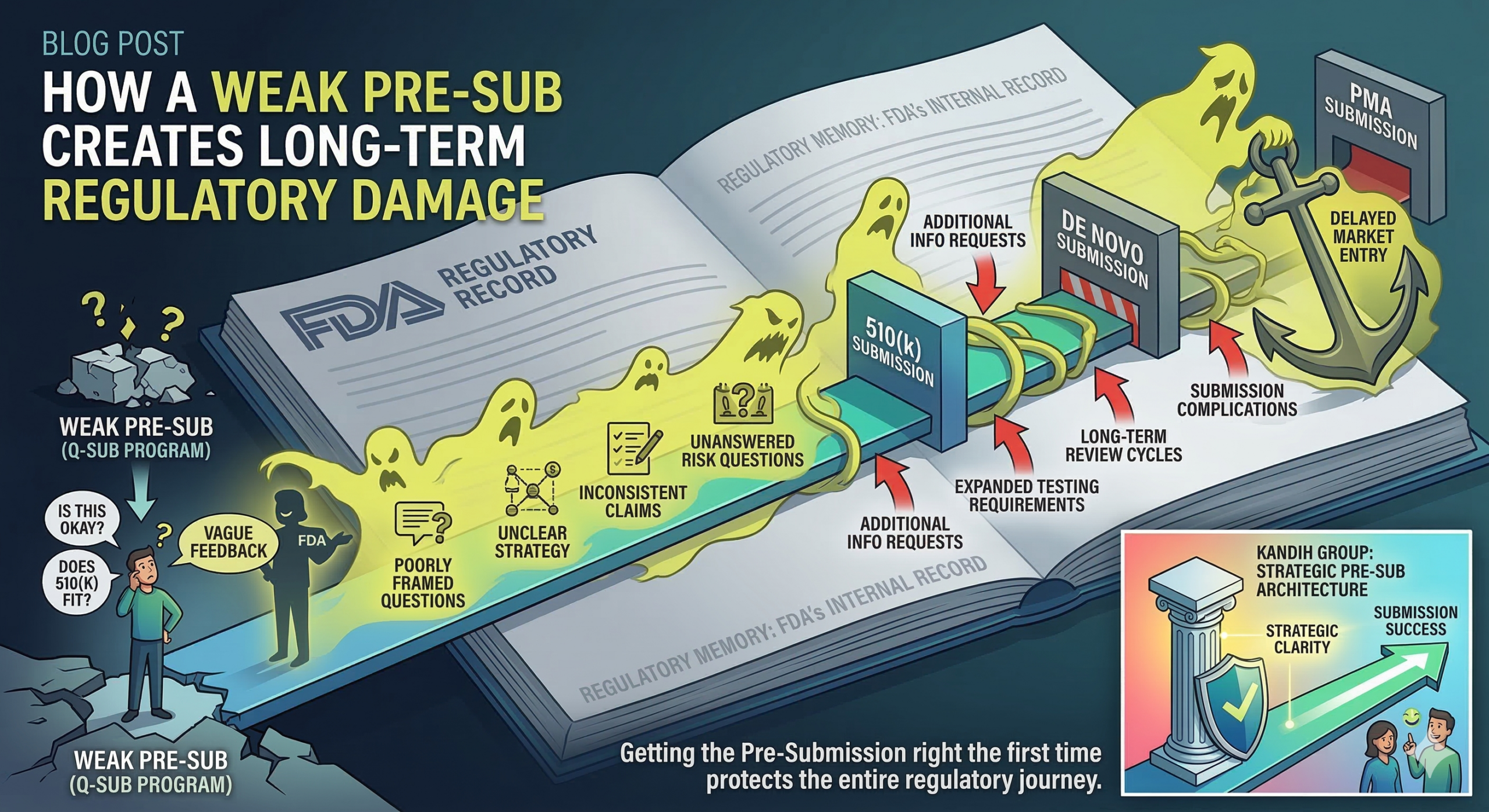
March 5, 2026What Does FDA Actually Expect in a Medical Device Pre-Submission?
March 9, 2026Short answer: FDA failure is not just a regulatory setback. It is a valuation event.
When a medical device company receives a major deficiency letter, Refuse-to-Accept decision, or pathway escalation from the U.S. Food and Drug Administration, investors do not see a technical issue. They see risk materializing.
And risk directly affects exits.
Why FDA Outcomes Matter to Investors
For investors, regulatory milestones are inflection points. Clearance or approval often unlocks:
Strategic partnerships
Revenue acceleration
Follow-on funding at higher valuation
Acquisition interest
Conversely, regulatory failure triggers:
Delayed revenue
Increased burn rate
Emergency capital raises
Down rounds
Exit uncertainty
In venture-backed device companies, FDA outcomes often determine whether an exit happens at 3–5 years—or stretches to 7–10.
How FDA Failure Impacts Exit Scenarios
1. Acquisition Timing Shifts
Many strategic acquirers prefer:
Cleared or approved devices
De-risked regulatory pathways
Predictable reimbursement strategy
If FDA clearance is delayed, acquisition discussions often stall.
Buyers discount regulatory uncertainty heavily.
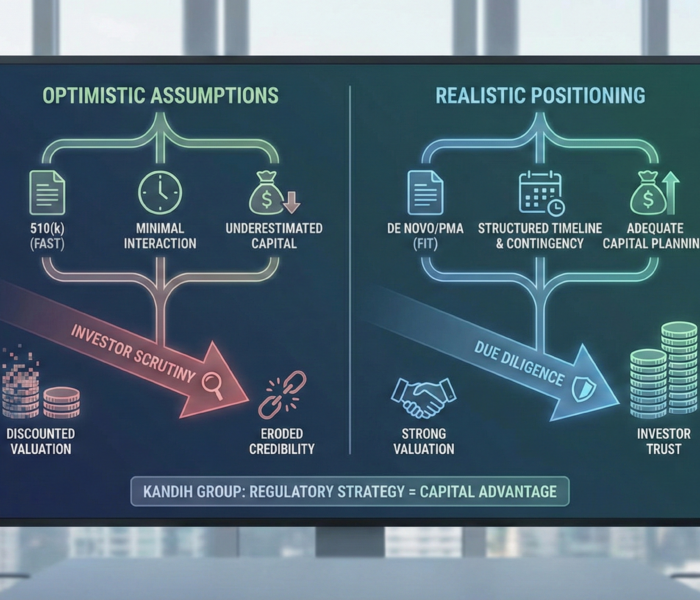
2. Valuation Compression
If a company experiences:
Multiple review cycles
Unexpected pathway changes
Expanded clinical requirements
investors adjust valuation models.
More time to market = more capital required.
More capital required = greater dilution.
Greater dilution = reduced founder and early investor return.
Regulatory instability lowers enterprise value.
3. Fundraising Under Pressure
If FDA outcomes are negative or delayed:
Burn continues
Revenue is deferred
Investor leverage shifts
Companies may raise at lower valuations to extend runway.
That impacts long-term ownership structure and exit multiples.
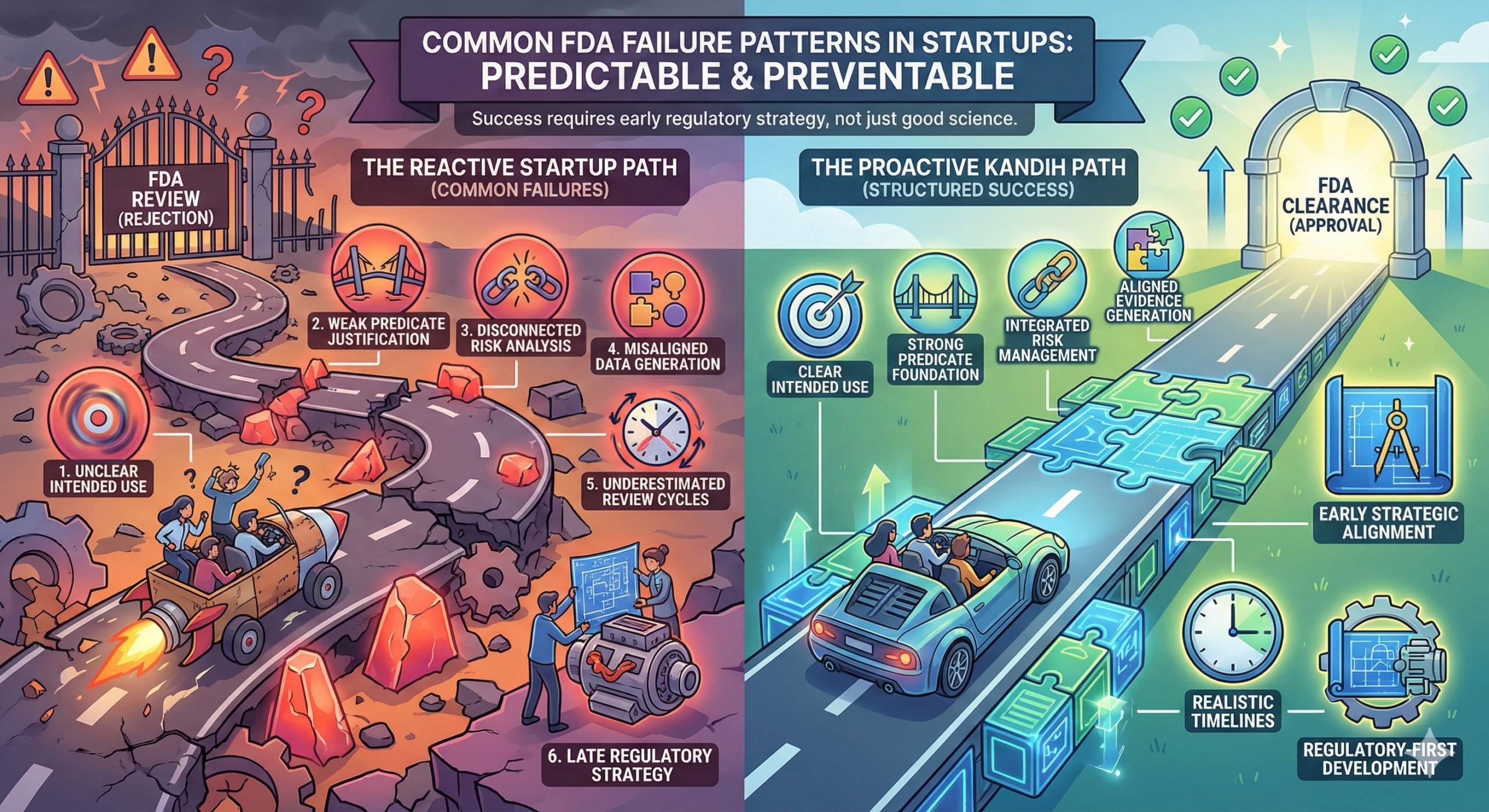
Why Investors Focus on Regulatory Discipline
Sophisticated investors evaluate:
Clarity of regulatory pathway
Strength of predicate strategy (if 510(k))
Alignment between intended use and evidence
Contingency planning
Realistic timeline modeling
They are not just assessing FDA success probability.
They are modeling exit probability.
Regulatory credibility influences acquisition credibility.
AEO: How Does FDA Approval Affect Startup Valuation?
Does FDA failure reduce valuation?
Yes. Delays increase capital needs and reduce exit predictability.
Do acquirers wait for FDA clearance?
Often, yes. Clearance or approval significantly reduces acquisition risk.
Can regulatory instability kill an exit?
Yes. Unresolved regulatory risk can delay or eliminate acquisition interest.
The Pattern Investors Recognize
When FDA failure occurs, investors often discover:
Pathway optimism
Weak predicate justification
Underestimated clinical burden
Reactive evidence generation
Regulatory failure rarely surprises experienced investors.
It reveals planning gaps.
Where Kandih Comes In
This is where Kandih Group acts as a regulatory risk translator for investors and founders.
Kandih supports investor-aligned development by:
Evaluating pathway realism before capital deployment
Stress-testing predicate assumptions
Modeling regulatory timeline and cost exposure
Aligning evidence plans with FDA expectations
Identifying hidden risk escalators early
Preparing companies for investor diligence conversations
Instead of presenting regulatory strategy as a hope, we translate it into structured risk logic that investors can evaluate confidently.
That protects:
Valuation integrity
Exit timing
Capital planning
Investor trust
Bottom Line
FDA failure is not just a regulatory issue.
It affects:
Burn rate
Ownership structure
Strategic acquisition interest
Exit multiples
From an investor lens, regulatory clarity is exit clarity.
When regulatory strategy is disciplined and aligned early, FDA milestones become value drivers—not valuation threats.
That is how upstream regulatory rigor protects downstream returns.
References
FDA – Premarket Notification 510(k)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-notification-510k
FDA – De Novo Classification Process
https://www.fda.gov/medical-devices/premarket-submissions/de-novo-classification-request
FDA – Premarket Approval (PMA)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-approval-pma
FDA – Refuse to Accept Policy for 510(k)s
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/refuse-accept-policy-510ks