Lynkuet (Elinzanetant): What Toxicology and Product Development Teach Us About Safe Innovation

Can LED Light Really Kill Cancer? What Toxicology Teaches Us About Safety, Innovation, and Smart Design
October 28, 2025
New Coronavirus Found in Brazilian Bats: What Toxicology and Product Development Must Learn Now
October 30, 2025Lynkuet (Elinzanetant): What Toxicology and Product Development Teach Us About Safe Innovation
Mechanism Meets Safety
Lynkuet (elinzanetant) is one of the first non-hormonal medicines approved to treat moderate to severe hot flashes and night sweats caused by menopause (EMA, 2025).
It’s a major step forward — and a powerful reminder for developers of one core truth:
When you create a new kind of drug, toxicology isn’t a formality. It’s your compass for designing safe innovation.
Developing Lynkuet wasn’t just about calming the brain circuits that trigger hot flashes.
It was about making sure that turning down one biological switch didn’t accidentally flip another — and cause harm elsewhere.
That’s what toxicology does: it balances innovation with safety, all the way from molecule to market.
Understanding Lynkuet: How It Works and Why Safety Matters
Lynkuet blocks two brain receptors — NK-1 and NK-3 — that sit inside a cluster of neurons known as KNDy neurons.
During menopause, these neurons become overactive, sending faulty “heat” signals that cause hot flashes.
By quieting those signals, Lynkuet helps restore balance.
But this new mechanism comes with unique challenges for toxicologists and developers:
It acts deep in the central nervous system (CNS) — which means even small off-target effects could affect the brain or mood.
It doesn’t use hormones, so there’s no historical safety data to rely on.
It’s first-in-class, meaning every safety study had to start from scratch.
When you’re pioneering something new, safety science has to pioneer alongside you.
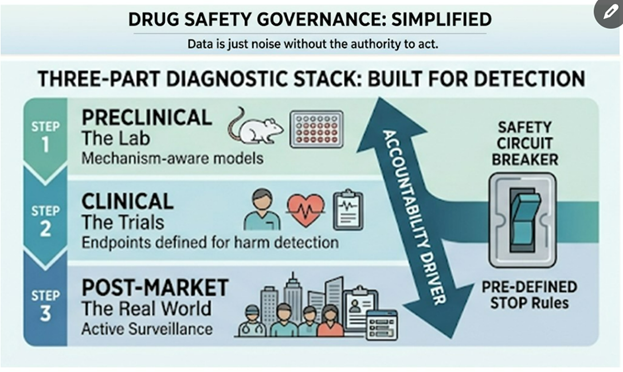
How Toxicology Guided Lynkuet’s Development
1. Finding the Body’s Weak Spots
Animal studies help reveal which organs might be sensitive to a drug over time.
For Lynkuet, long-term studies pointed to possible effects in the reproductive system, brain, and digestive tract (Bayer, 2025).
At very high doses, rats showed muscle spasms and primates had mild hormonal changes.
This helped toxicologists and clinicians design smarter human trials — with careful dose escalation, safety monitoring, and early warning systems for side effects.
Lesson: Toxicology defines your safety boundaries before the first patient ever takes a pill.
2. Understanding How the Drug Moves and Interacts
Lynkuet is broken down by the liver enzyme CYP3A4 and is transported by P-glycoprotein, two systems that handle many common drugs.
That means potential drug–drug interactions with medications like antifungals or antibiotics.
Toxicology and pharmacology teams used these insights to:
Adjust dosage for people with liver or kidney problems
Test for buildup in skin and eye pigments (since melanin can trap some drugs)
Watch for light sensitivity — a potential “phototoxic” effect linked to light exposure
Lesson: Toxicology turns complex metabolism data into clear, actionable safety strategies — dosing rules, monitoring plans, and label warnings.
3. Planning for the Long Haul
Because Lynkuet is meant for chronic use, its safety story doesn’t end at approval.
Toxicologists continue monitoring for liver, reproductive, and light-related effects through long-term studies and post-market surveillance.
This “lifecycle toxicology” ensures that as more patients use the drug, new safety data is analyzed and reflected in label updates and patient guidance.
Lesson: Safe innovation isn’t one-and-done — it’s continuous learning.
A Real-World Lesson in Safety Design
In one neuroscience drug program, a compound caused skin reactions during animal testing.
The culprit? A stabilizer molecule that bonded to melanin, making the skin more sensitive to light.
By reformulating early, the toxicology team prevented what could have become a major safety recall.
Lynkuet’s journey reflects that same proactive mindset — identify risks early, fix them fast, and protect both patients and the product.
Why This Matters
For biotech founders, R&D teams, and regulatory leaders, Lynkuet represents more than a new menopause therapy.
It’s a case study in responsible innovation — proving that toxicology isn’t the brake pedal; it’s the navigation system.
Safe innovation means:
Anticipating problems before they appear
Designing products that prevent harm by default
Building trust through transparency and science
Efficacy builds excitement. Toxicology builds longevity.
If you’re developing a first-in-class therapy, remember:
The molecule is only half the story.
The other half is how you protect patients and your product’s future with a toxicology-driven roadmap.
References
1. European Medicines Agency (EMA). Lynkuet (elinzanetant) – Summary of Product Characteristics. Accessed Sept 19, 2025. https://www.ema.europa.eu/en/medicines/human/EPAR/lynkuet
2. Bayer AG. Lynkuet (elinzanetant) Product Monograph Including Patient Medication Information. July 23, 2025. https://www.bayer.com/sites/default/files/lynkuet-pm-en.pdf
3. U.S. Food and Drug Administration (FDA). Guidance for Industry: Nonclinical Toxicology Evaluation for Drugs. https://www.fda.gov/media/71542/download
4. Centers for Disease Control and Prevention (CDC). Drug Development and Safety Monitoring for Novel Therapies. https://wwwnc.cdc.gov/eid/article/30/11/23-1416_article
5. International Council for Harmonisation (ICH). S6(R1): Preclinical Safety Evaluation of Biotechnology-Derived Pharmaceuticals. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/s6r1-preclinical-safety-evaluation-biotechnology-derived-pharmaceuticals