New Coronavirus Found in Brazilian Bats: What Toxicology and Product Development Must Learn Now

Lynkuet (Elinzanetant): What Toxicology and Product Development Teach Us About Safe Innovation
October 29, 2025
Outbreak of Lung Injury Associated with the Use of E-Cigarette, or Vaping, Products
October 31, 2025New Coronavirus Found in Brazilian Bats: What Toxicology and Product Development Must Learn Now
When Discovery Meets Responsibility
Scientists in Brazil have discovered a previously unknown coronavirus in bats — one that carries genetic similarities to MERS-CoV, the virus responsible for deadly outbreaks in the Middle East.
(University of Nebraska Medical Center, 2025
)
For professionals in biotech, toxicology, and product development, the message is urgent and clear:
Emerging pathogens aren’t just a virology issue. They’re a toxicology and product-safety challenge long before they become a clinical crisis.
When animal viruses jump to humans — what scientists call spillover — the race begins to create diagnostics, antivirals, and vaccines. But moving fast without an equally rapid safety framework is a recipe for unintended harm.
Why This Discovery Matters for Toxicology and Product Development
1. Expanding Exposure Risks
The new coronaviruses found in Brazilian bats belong to the alpha- and beta-coronavirus families — the same groups that include SARS, MERS, and COVID-19 (Magalhães et al., 2025
).
That means the geographic and biological risk zones just expanded. For developers, this redefines the “exposure map” — who might come in contact, where, and how.
For toxicologists, it means running early safety assessments on products that may interact with these new viral proteins — even before human cases appear.
Because if a vaccine, drug, or diagnostic later crosses into a different species or population, we need to know how it behaves in new biology, not just the old.
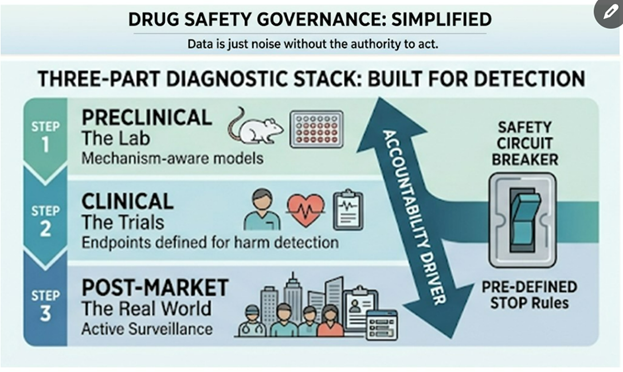
2. Preclinical Safety in Unknown Terrain
When new viruses emerge, timelines compress — but safety science can’t skip steps.
Every aspect of preclinical testing must be re-evaluated:
Animal models: Does the infection behave differently in species used for safety studies?
Biodistribution: Where do vaccine components or antibodies travel once in the body?
Immunotoxicity: Could an immune overreaction cause harm (like cytokine storms)?
Off-target effects: Could antibodies or antivirals mistakenly bind to human tissues?
In the rush to respond, even small oversights can have large consequences.
For instance, a live vaccine vector might replicate safely in one species but trigger inflammation in another. A broadly neutralizing antibody could bind not just the virus — but a host protein critical for normal function.
Lesson: Speed matters. But precision — through toxicology — saves lives.
3. Manufacturing and Lifecycle Risks
Pandemic urgency often drives “warp-speed” manufacturing. That’s when formulation and contamination risks spike.
Toxicologists ensure that:
Manufacturing impurities or residual viral vectors are identified early.
Adjuvants (immune boosters) are tested for unexpected reactions.
Product shelf life, stability, and transport conditions don’t alter safety.
In other words — when the pathogen changes, the product template must change too. Reusing old designs without revalidating safety is like using a seatbelt from one car model in another — it might not fit when it matters most.
Tactical Steps for Product Teams
Map Exposure Early: Identify at-risk populations (e.g., communities near bat habitats in Brazil). Model worst-case spillover scenarios to plan safety thresholds.
Integrate Toxicology at Sequence Stage: Start safety screens — including immune overactivation and cross-reactivity tests — as soon as new viral genetic data is available.
Build Modular Safety Frameworks: Use validated platforms (like mRNA or adenovirus vectors), but re-run the safety modules for this new pathogen context.
Communicate Transparently: Regulators, clinicians, and the public must see safety as part of design — not an afterthought. Publish plain-language summaries of toxicology data to build trust.
Update Risk Management Plans: Add “emerging zoonotic virus” as a formal risk trigger in your development roadmap. Link it to supply-chain, manufacturing, and regulatory risk mitigation plans.
From Experience: When Speed Outran Safety
During a previous outbreak, a diagnostic kit was fast-tracked to market. Later, it was found to cross-react with human proteins, producing false positives. Patients were misdiagnosed; confidence in testing collapsed.
The root cause? Toxicology screens hadn’t been updated for the new viral proteins.
The harm was avoidable — and entirely predictable.
Takeaway: In emergencies, you can accelerate development — but you can’t outsource safety.
The Bottom Line
The discovery of a new coronavirus in Brazilian bats is more than a scientific headline — it’s a warning shot for every R&D, toxicology, and regulatory team.
When new pathogens appear, the safety rulebook must be rewritten — not reused.
Emergent pathogens demand emergent toxicology.
Innovation must evolve as fast as the virus does — safely.
Fast innovation saves time.
Safe innovation saves lives.
References
1. Magalhães T.B.S., et al. First Detection of Alphacoronavirus in Bats from the World’s Largest Wetland, the Pantanal, Brazil. Pathogens. 2025;14(1):58. https://www.mdpi.com/2076-0817/14/1/58
2. “Close Relative of Highly Fatal Coronavirus Discovered in Brazil’s Bats.” University of Nebraska Medical Center Global Center for Health Security. March 19, 2025. https://www.unmc.edu/healthsecurity/transmission/2025/03/19/close-relative-of-highly-fatal-coronavirus-discovered-in-brazils-bats
3. Góes L.G.B., et al. Novel Bat Coronaviruses, Brazil & Mexico. Emerg Infect Dis. 2013;19(12):2176–2179. https://pubmed.ncbi.nlm.nih.gov/24050144
4. U.S. Food and Drug Administration (FDA). Guidance for Industry: Nonclinical Safety Evaluation of Biotechnology-Derived Products. https://www.fda.gov/files/drugs/published/Drug-Products–Including-Biological-Products–that-Contain-Nanomaterials—Guidance-for-Industry.pdf
5. Centers for Disease Control and Prevention (CDC). Zoonotic Diseases and Emerging Infectious Diseases. https://www.cdc.gov/ncezid/index.html
6 TimesAlert! New Bat Coronavirus Found in Brazil Raises Pandemic Monitoring Importance. https://timesofindia.indiatimes.com/life-style/health-fitness/health-news/new-pandemic-alert-another-bat-coronavirus-found-in-brazil-sparks-concerns-over-human-infection/articleshow/119127512.cms