Can LED Light Really Kill Cancer? What Toxicology Teaches Us About Safety, Innovation, and Smart Design

FDA Recalls Cholesterol Medication — What Toxicology Can Teach Developers About Hidden Risks
October 27, 2025
Lynkuet (Elinzanetant): What Toxicology and Product Development Teach Us About Safe Innovation
October 29, 2025Can LED Light Really Kill Cancer? What Toxicology Teaches Us About Safety, Innovation, and Smart Design
Light as Medicine — and a Safety Challenge
You may have seen the headline:
“LED light can kill cancer cells.”
It sounds futuristic — shining light on a tumor instead of blasting it with radiation or flooding it with chemotherapy. But this idea isn’t fantasy. It’s called photodynamic therapy (PDT) — and it’s a real area of cancer research that’s gaining traction.
Here’s how it works: scientists use a special light plus a special drug. The drug builds up inside cancer cells. When hit with the right color of light, it activates and becomes toxic — killing the cancer from the inside out.
It’s elegant.
It’s promising.
But it’s also risky if not handled carefully.
That’s where toxicology — the science of safety — comes in.
Why Toxicology Matters Here
Every new therapy has two sides: the power to heal and the potential to harm.
Toxicology is about understanding that balance.
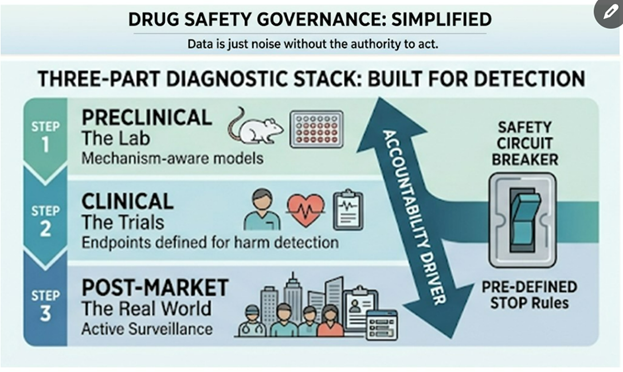
For light-based cancer treatments, toxicologists focus on three key questions:
1. Does It Target Only Cancer Cells?
The goal is precision — killing only the bad cells and leaving healthy ones alone.
Toxicologists study where the drug goes in the body and how long it stays there. If it lingers in healthy skin or organs, light exposure could accidentally cause burns or inflammation.
2. How Much “Light Damage” Is Safe?
When light activates the drug, it creates something called reactive oxygen species (ROS) — short-lived, oxygen-based molecules that kill cells.
Think of them as microscopic sparks.
The trick is using enough “sparks” to destroy cancer, but not so many that they damage surrounding tissue. Toxicologists help define that safe middle ground.
3. How Do the Drug and Device Work Together?
This treatment isn’t just a pill or a machine — it’s both. The drug does the targeting; the LED light does the activation.
That makes it a combination product — part medicine, part device.
Toxicologists work with engineers to make sure the light’s strength, timing, and temperature won’t harm tissue during use.
For Innovators and Developers
If you’re building anything that uses energy — light, sound, or electricity — to affect the body, here’s what toxicology teaches us:
Start safety testing early.
Don’t wait for human trials. Run small lab studies to see how your product behaves under real-world light or heat exposure.
Design for control.
The best medical technologies don’t just “work” — they’re controllable. Toxicologists help you find that safe operating window.
Use modern test models.
New 3D tissue cultures and organ-on-chip systems can predict how human tissue reacts — faster and cheaper than animal testing.
Collaborate from the start.
Bring toxicologists, engineers, and regulatory experts into the design phase. It saves you from expensive course corrections later.
A Real-World Lesson
In one cancer study, a drug used for light therapy worked beautifully in lab dishes.
But when tested in people, patients developed skin inflammation after walking outside — the drug was still in their skin and reacted to sunlight.
A toxicology review found the issue: the compound stayed in the body too long.
The solution? Reformulate the drug and adjust how long patients avoided light after treatment.
Problem solved — and the innovation stayed alive.
That’s toxicology in action: predicting harm before it reaches people.
Limitations and Reality Check
While LED-based cancer therapies look promising, they’re not magic bullets.
Some early lab studies show that under certain light conditions, cancer cells can actually grow faster instead of dying.
This doesn’t mean the idea doesn’t work — it means the details matter: light color, dosage, and timing all affect safety and results.
And as with any cancer treatment, success depends on the individual patient — tumor type, genetics, and overall health all play a role.
Good science is cautious science.
The Takeaway
The clear idea is simple:
Light can heal — but only if science keeps it safe.
For innovators, that means safety isn’t a barrier to progress — it’s the foundation for it.
Smart design is toxicology in disguise: knowing where danger hides and building systems that prevent it.
References
1. U.S. Food and Drug Administration (FDA). Guidance for Industry: Nonclinical Evaluation of Photodynamic Therapy Drugs. https://www.fda.gov/media/71542/download
2. Centers for Disease Control and Prevention (CDC). Cancer Treatment Safety and Exposure Guidelines. https://pmc.ncbi.nlm.nih.gov/articles/PMC3726067
3. Castano AP, et al. Photodiagnosis Photodyn Ther. 2004;1(4):279–293. https://pmc.ncbi.nlm.nih.gov/articles/PMC4108220
4. Xiaoyu Y, et al. Advances in Catalytic Enantioselective Fluorination, Mono-, Di-, and Trifluoromethylation, and Trifluoromethylthiolation Reactions Chem Rev. 2015;115(4):1990–2042. https://pubs.acs.org/doi/10.1021/cr500277b