August 25, 2025
Published by admin on August 25, 2025
In June 2025, the European Medicines Agency (EMA) confirmed what many clinicians suspected: non-arteritic anterior ischemic optic neuropathy (NAION) is now recognized as a very rare but real side effect of semaglutide (Ozempic). For patients, this means sudden, irreversible vision loss.
For drug developers, it’s a wake-up call: standard GLP toxicology will almost never catch NAION.
August 20, 2025
Published by admin on August 20, 2025
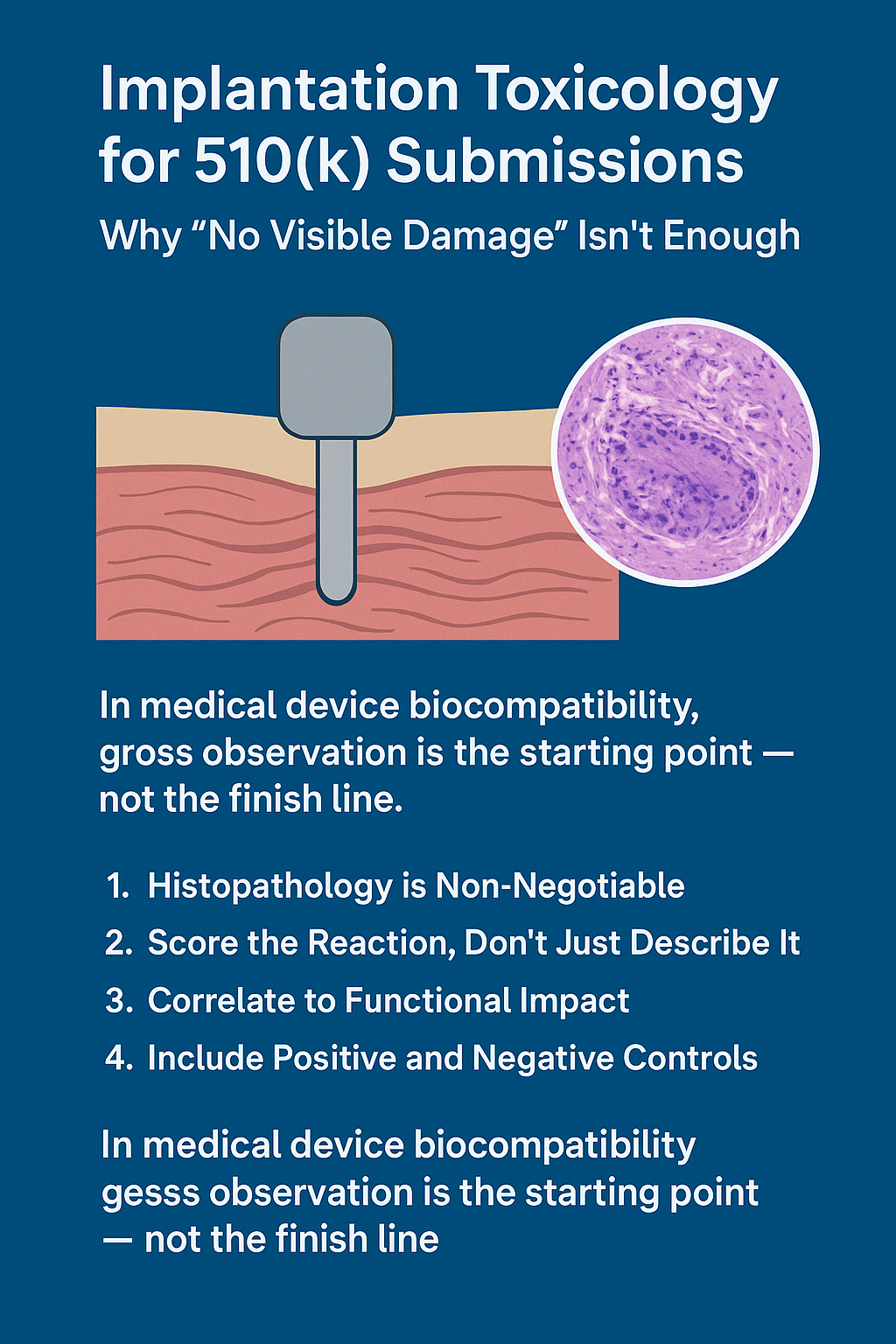
In medical device biocompatibility, gross observation is the starting point — not the finish line.
If your implantation study report for a 510(k) simply says “no visible tissue reaction”, you’re setting yourself up for FDA pushback. Why? Because regulators know what seasoned toxicologists know: devices can cause microscopic and functional changes long before they cause visible tissue damage.
August 13, 2025
Published by admin on August 13, 2025
Choosing the wrong tissue site in an implantation study is one of the fastest ways to derail your 510(k) submission.
We’ve seen FDA request repeat testing—adding months and tens of thousands of dollars—simply because the chosen implantation site didn’t match the device’s intended clinical use.
August 11, 2025
Published by admin on August 11, 2025
Many medical device developers pass cytotoxicity, irritation, and sensitization testing—only to hit a wall at the implantation stage.
In fact, one of the most common reasons 510(k) submissions face delays is a poorly designed ISO 10993-6 implantation study.