January 23, 2026
Published by admin on January 23, 2026
CAPA Survival Playbooks — Kandih Bioscience Complaint-to-CAPA mapping is a risk-interpretation system, not an administrative checklist.FDA evaluates whether complaint signals are consistently interpreted, escalated, or deferred […]
January 22, 2026
Published by admin on January 22, 2026
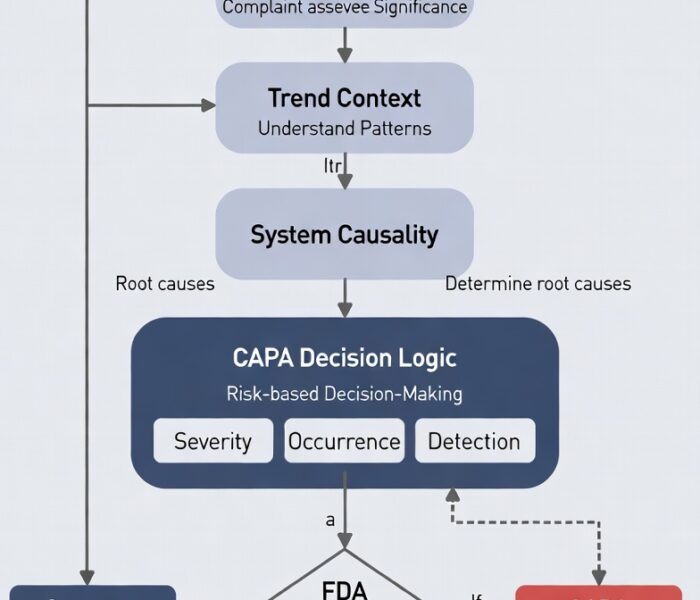
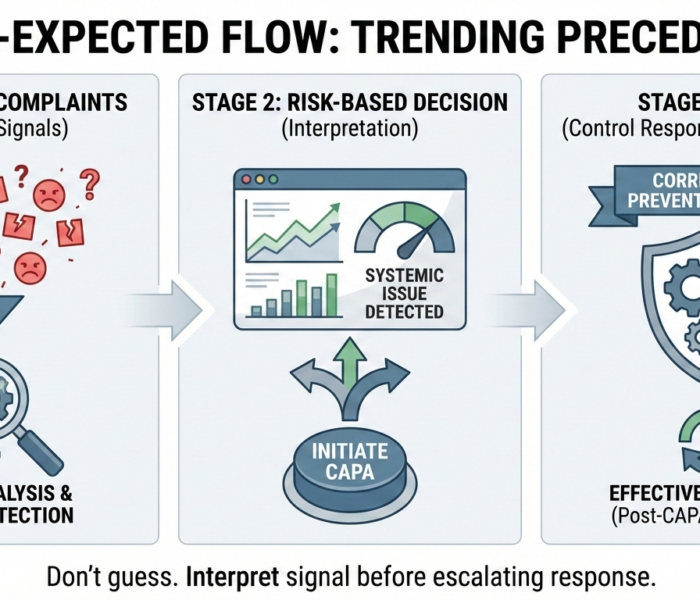
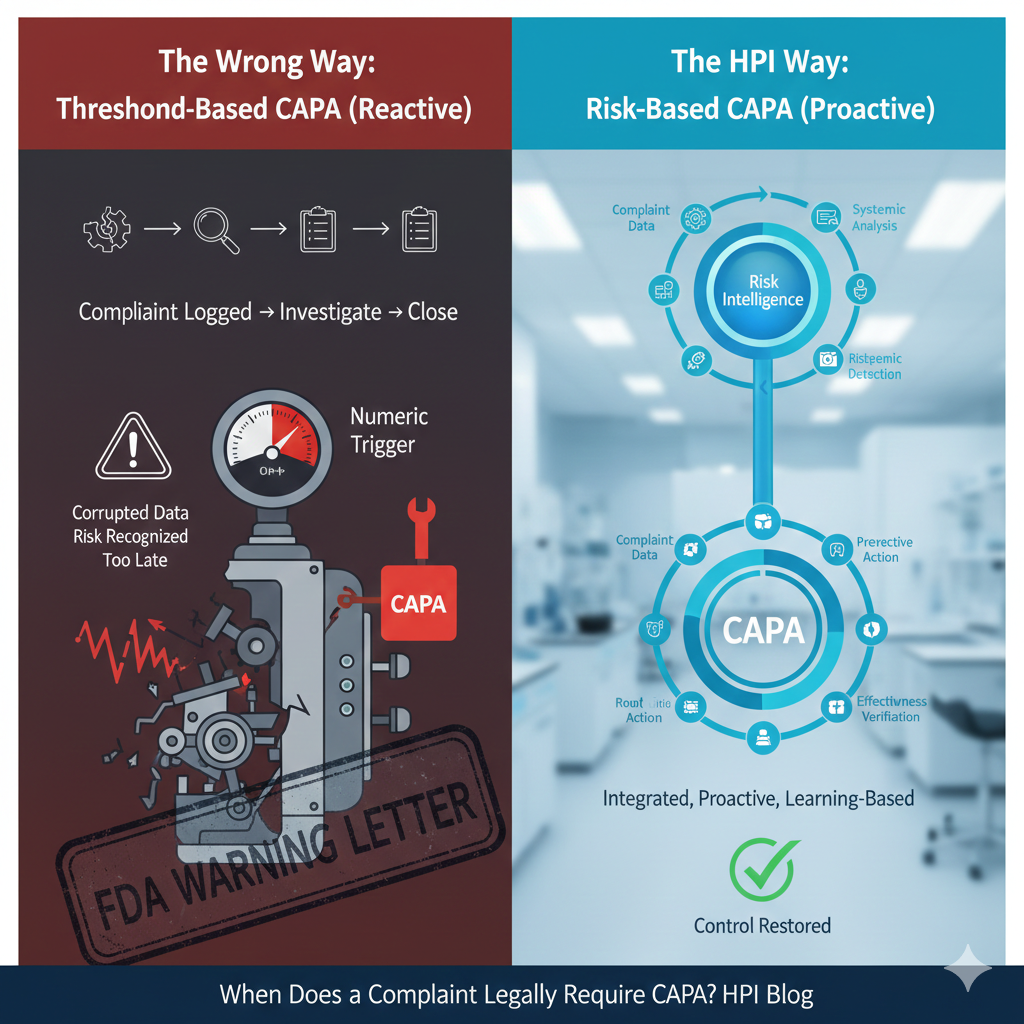
Yes—FDA expects complaint trending before CAPA initiation.From an inspection perspective, complaint trending is a diagnostic function, not an effectiveness check. If trending only occurs after CAPA […]
January 21, 2026
Published by admin on January 21, 2026
CAPA Survival Playbooks — Kandih Bioscience Repeat FDA 483 observations almost always mean the original root cause was wrong.From an FDA enforcement perspective, recurrence equals misdiagnosis. […]
January 20, 2026
Published by admin on January 20, 2026
A complaint legally requires CAPA when it signals loss of control in the quality system—not when it hits a numeric or procedural threshold.From an FDA enforcement […]