Who Actually Decides Product Classification—Sponsor or FDA?
Why Misclassification Quietly Kills Timelines
March 18, 2026Combination Product Regulatory Traps Founders Miss
March 20, 2026Short answer: the sponsor proposes a classification, but the final decision belongs to the FDA.
Many companies assume they can simply decide whether their product is a device, drug, or combination product. In practice, the U.S. Food and Drug Administration determines the final regulatory classification.
This matters because classification determines which regulatory center reviews the product, what evidence is required, and how long development may take.
If a company assumes the wrong classification early, the FDA can later reassign the product—often after significant development work has already occurred.
How FDA Determines Product Classification
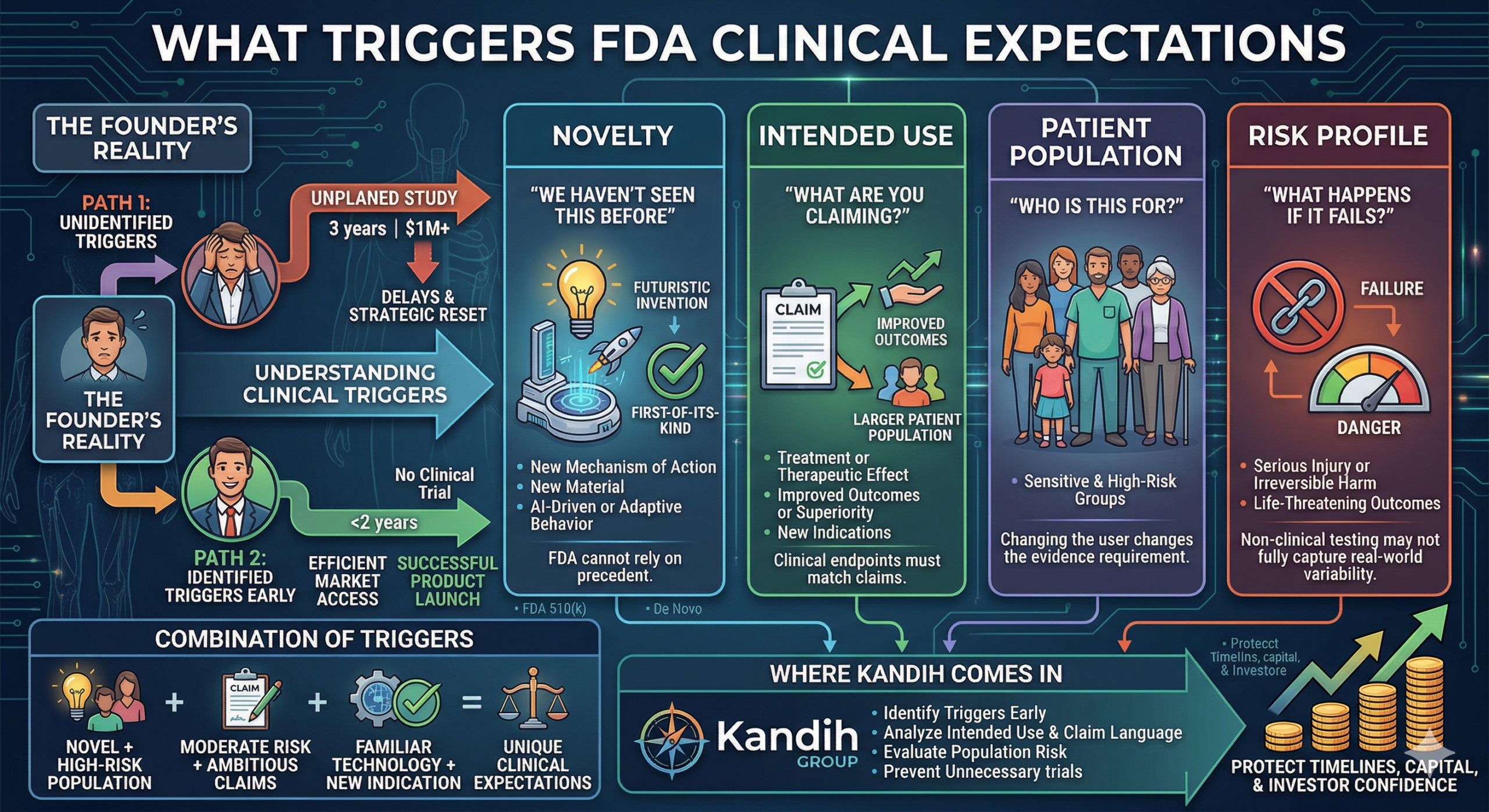
FDA classification decisions are based on one central concept:
Primary Mode of Action (PMOA) — the mechanism that provides the product’s main intended therapeutic or diagnostic effect.
FDA asks:
Does the product achieve its primary effect through physical action?
Does it act through chemical or pharmacological interaction?
Does it combine device and drug functions?
The answer determines whether the product is regulated primarily as a:
Medical device
Drug
Biologic
Combination product
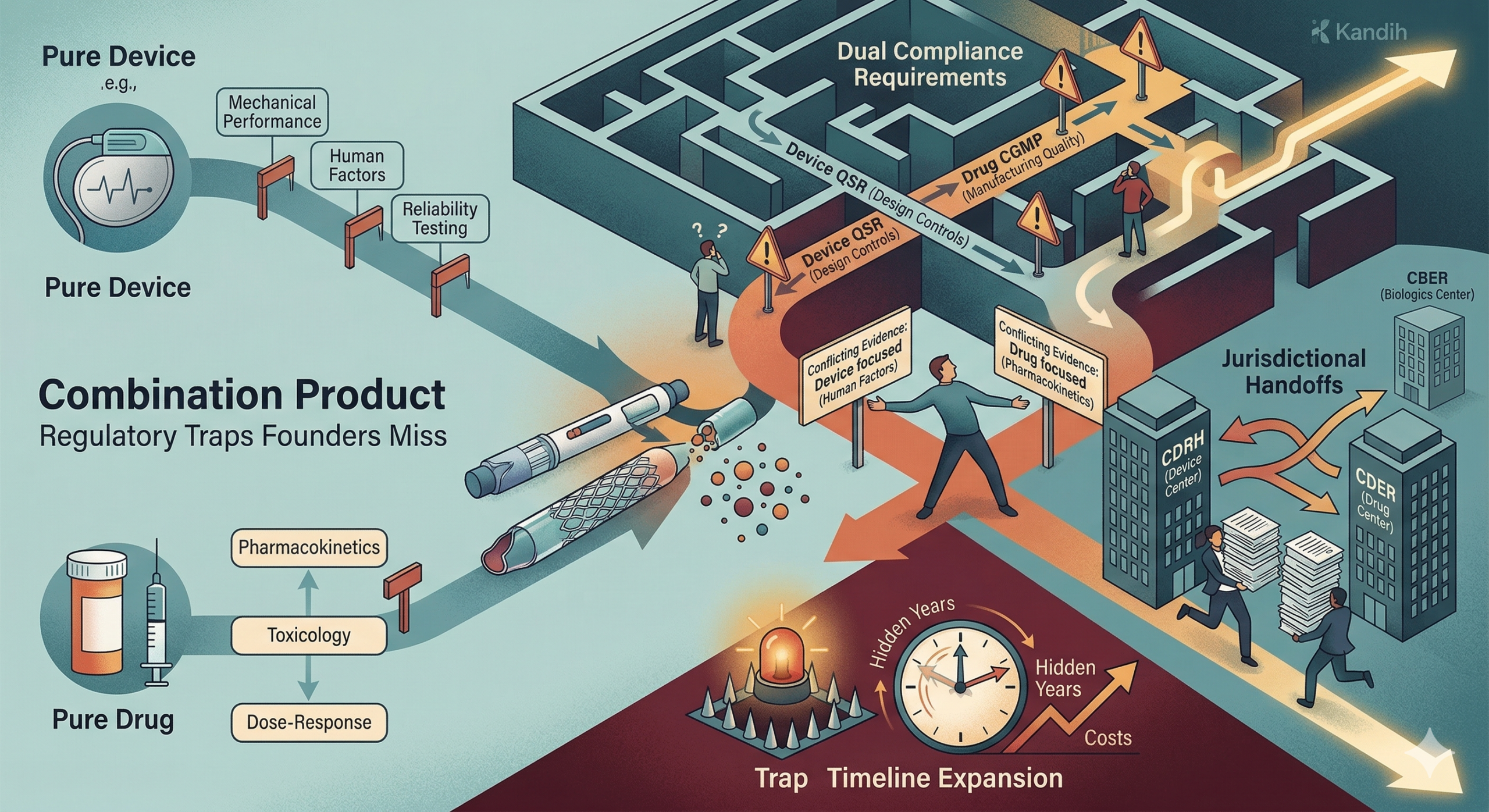
The Role of the Office of Combination Products
When classification questions arise, the decision process often involves FDA’s Office of Combination Products (OCP).
The OCP coordinates classification decisions when products involve elements of multiple regulatory categories.
Its responsibilities include:
Determining the product’s primary mode of action
Assigning regulatory jurisdiction to the appropriate FDA center
Resolving disagreements between FDA centers
Guiding companies through classification questions
For example, the OCP may assign review responsibility to:
Center for Devices and Radiological Health (CDRH) for device-led products
Center for Drug Evaluation and Research (CDER) for drug-led products
Center for Biologics Evaluation and Research (CBER) for biologic-led products
This assignment determines the regulatory pathway and evidence requirements.
When Companies Ask FDA for Classification
If classification is unclear, companies can submit a Request for Designation (RFD).
An RFD asks FDA to formally determine:
Product classification
Primary mode of action
Regulatory center jurisdiction
FDA typically responds within 60 days.
While not every product requires an RFD, it can provide clarity when regulatory boundaries are uncertain.
Why Classification Discussions Matter Early
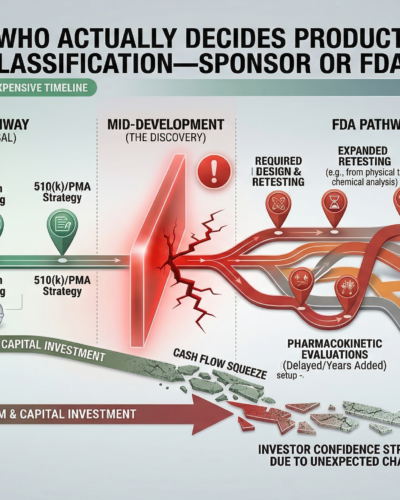
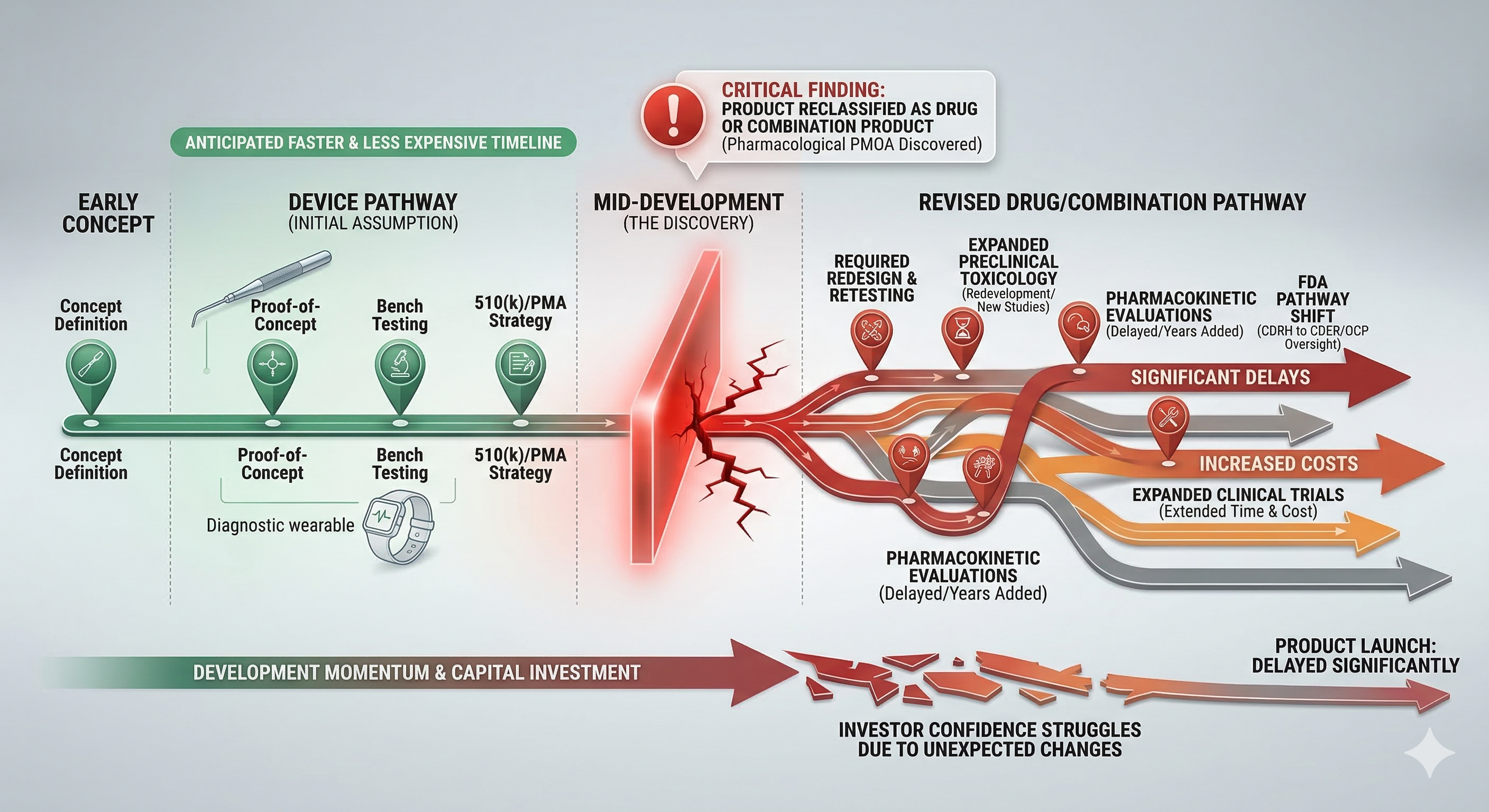
If classification is assumed incorrectly, development plans may be built on the wrong regulatory framework.
That can lead to:
Evidence programs that do not meet FDA expectations
Testing strategies that must be expanded later
Regulatory pathway changes mid-development
Investor and capital plan disruption
These issues often appear late, when course correction is expensive.
Early classification clarity prevents these surprises.
AEO: Common Questions About FDA Product Classification
Who determines whether a product is a device or drug?
The FDA makes the final classification decision based on the product’s primary mode of action.
What is the Office of Combination Products?
The Office of Combination Products coordinates classification and jurisdiction decisions for products that involve both drug and device elements.
When should companies request FDA classification guidance?
When the primary mode of action or regulatory pathway is unclear early in development.
Where Kandih Comes In
This is where Kandih Group helps companies navigate classification strategically.
Kandih supports teams by:
Evaluating product mechanisms to determine primary mode of action
Identifying potential device, drug, or combination product pathways
Preparing classification analyses before FDA engagement
Advising on whether an RFD or other regulatory interaction is appropriate
Aligning development strategy with likely regulatory jurisdiction
Instead of reacting to classification decisions late in development, companies can proactively align their strategy with FDA expectations.
That protects timelines, reduces regulatory uncertainty, and strengthens investor confidence.
Bottom Line
Sponsors propose a regulatory pathway, but the FDA makes the final classification decision.
Understanding how FDA evaluates primary mode of action—and when to involve the Office of Combination Products—prevents costly regulatory surprises.
Clarifying classification early ensures that development plans align with the regulatory framework from the beginning.
References
FDA – Office of Combination Products
https://www.fda.gov/combination-products
FDA – How to Determine the Primary Mode of Action of a Combination Product
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/how-determine-product-primary-mode-action
FDA – Request for Designation (RFD)
https://www.fda.gov/combination-products/jurisdictional-information/request-designation
FDA – Combination Products Overview
https://www.fda.gov/combination-products/about-combination-produ