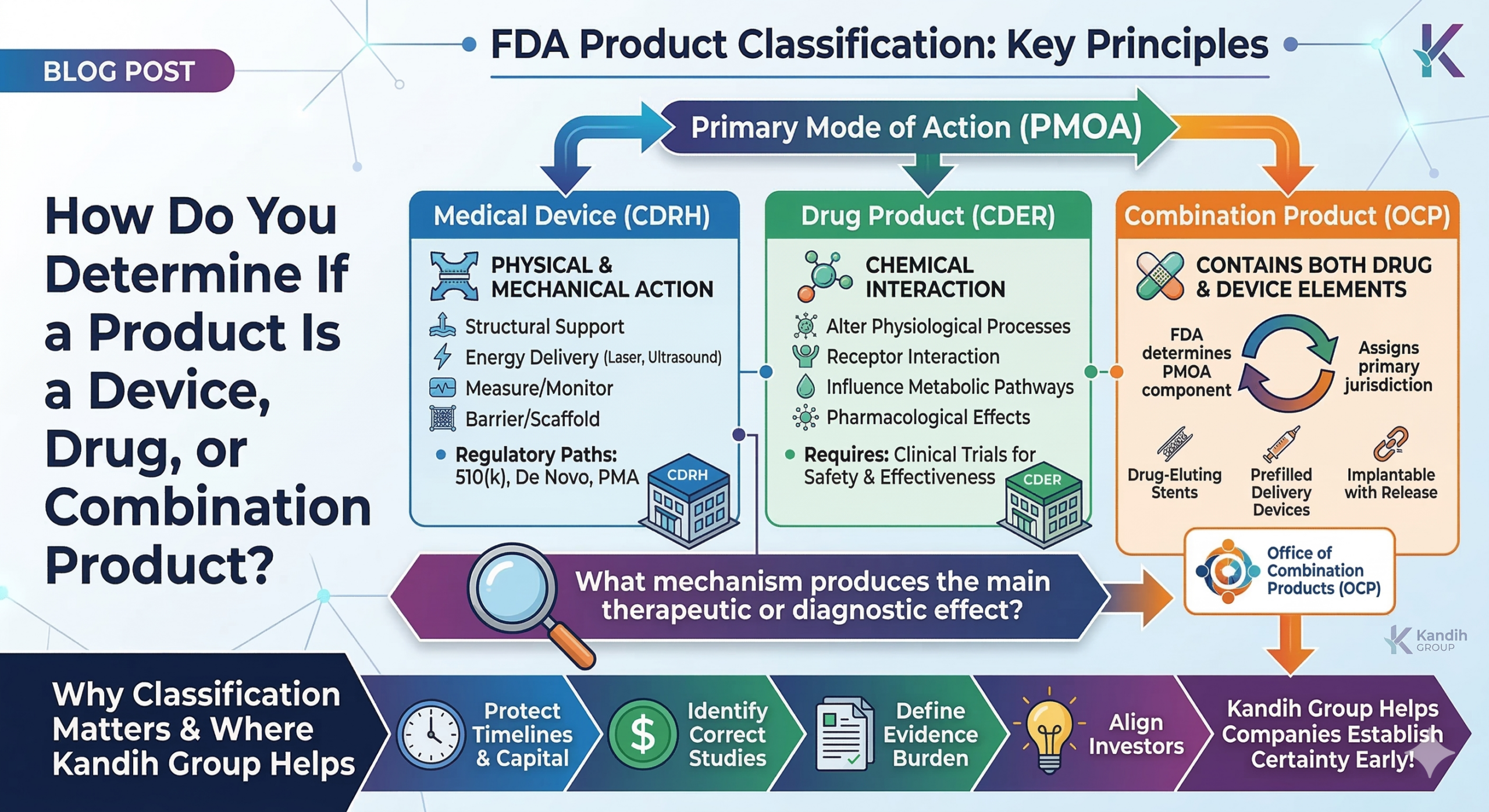
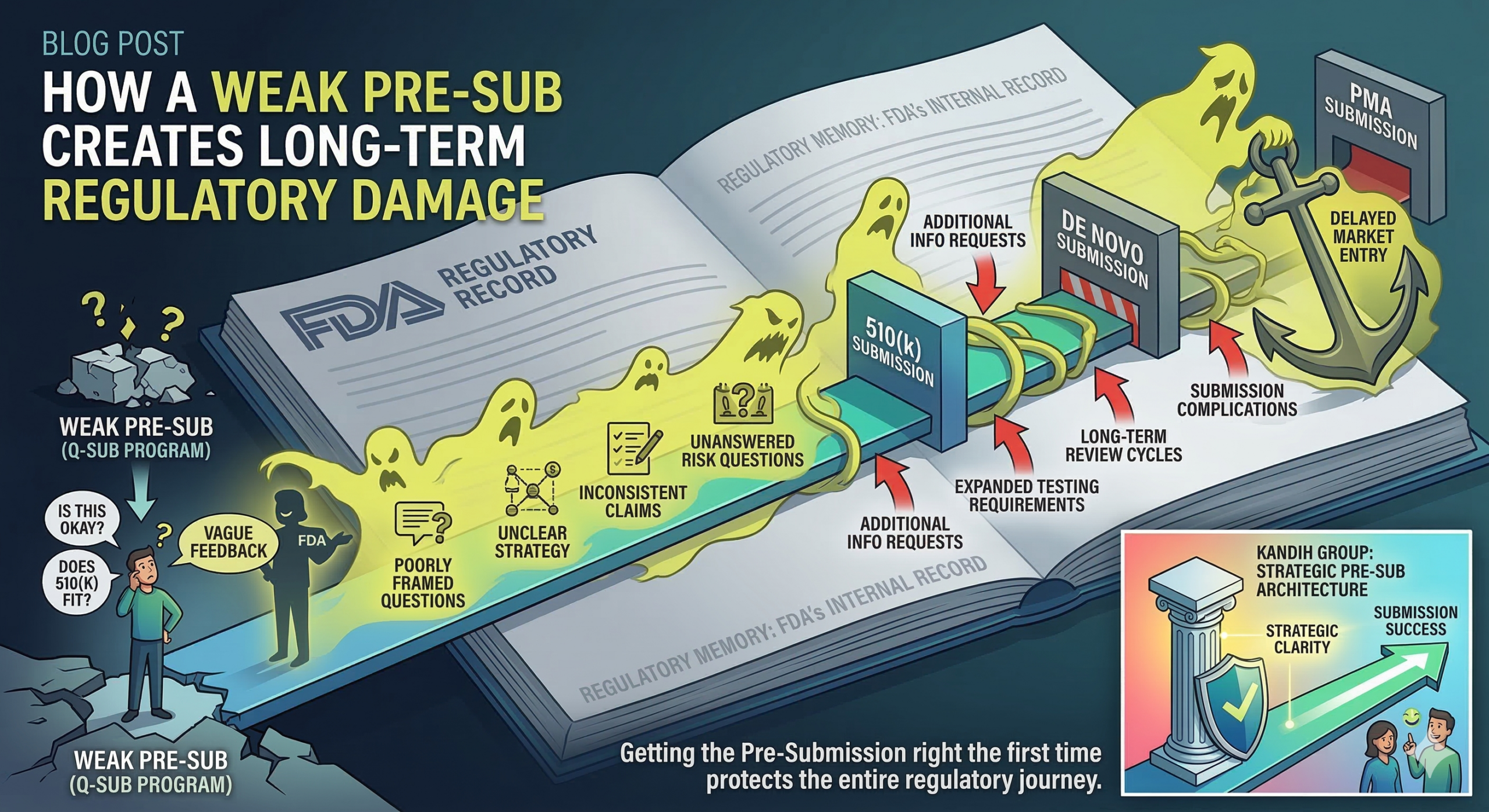
CAPA Failures as Evidence of a Broken Risk-Control System

Why FDA Treats CAPA as a Risk-Control System, Not a QA Task
January 12, 2026
CAPA ≠ Paperwork. It’s a Risk-Control Feedback Loop
January 14, 2026(How FDA documents system collapse, not paperwork gaps)
FDA consistently uses CAPA failures as proxy evidence that a firm’s end-to-end risk-control architecture is nonfunctional. For medical devices, this is most often cited as a violation of 21 CFR 820.100(a). For drugs and combination products, the same systemic failure is documented through CGMP investigation and CAPA expectations under 21 CFR Part 211, even when the word “CAPA” is not used explicitly.
Across warning letters, the pattern is stable: FDA does not isolate CAPA errors. It uses them to demonstrate loss of management control over quality.
Canonical 21 CFR 820.100(a) Language (Devices)
FDA repeatedly uses near-identical language to signal that the entire CAPA operating system is deficient:
Criticare Technologies, Inc. (2024)
FDA cited “Failure to establish and maintain procedures for implementing corrective and preventive actions, as required by 21 CFR 820.100(a)”, noting that the CAPA procedure lacked sufficient detail to:
Identify causes
Investigate nonconformities
Implement appropriate corrective actions
Verify or validate effectiveness
FDA explicitly stated that the examples “describe a CAPA system that does not meet the above requirements” and emphasized that this was a repeat deficiency following a prior 2017 Warning Letter—classic evidence of ineffective systemic control, not clerical error.
Source: [1]
Jiangsu Caina Medical Co., Ltd. (2024)
FDA cited failure to adequately establish and maintain CAPA procedures under 21 CFR 820.100(a) and specifically invoked §820.100(a)(1) for failing to analyze quality data sources to identify existing and potential causes of nonconforming product.
This is FDA explicitly calling out a broken signal-detection and risk-interpretation layer.
Source: [2]
Contec Medical Systems Co., Ltd. (2025)
FDA cited failure to adequately establish CAPA procedures and then decomposed the failure into system elements:
Causes not adequately investigated
Corrective actions not documented per procedure
Preventive actions (e.g., cybersecurity management plan) not completed prior to CAPA closure
This letter reads as a CAPA verification and governance failure, not a documentation issue.
Source: [4]
Failure to Trigger or Escalate CAPA (Broken Signal Layer)
FDA frequently documents CAPA failures where written triggers exist but are not operationalized—a hallmark of a nonfunctional operating system.
iRhythm Technologies, Inc. (2023)
FDA observed failure to initiate CAPA in response to negative trends and critical complaints, despite CAPA procedures requiring escalation. The agency highlighted repeated device transmission limit exceedances without systemic action—clear evidence of signal detection without system response.
Source: [5]
Royal Philips (2025)
FDA noted that CAPAs were not opened despite defined triggers such as:
Risk to public health
Systemic issues
Trends exceeding defined expectations
FDA’s critique focused on the gap between documented escalation logic and actual behavior, signaling governance failure.
Source: [6]
Exer Labs, Inc. (2025)
FDA described a complaint involving a failed software update where design changes were made but no CAPA evaluation was performed to assess systemic impact—explicitly criticizing the failure to treat the issue as potentially systemic.
Source: [7]
Inadequate Investigation and Root Cause (Broken Causality Layer)
Kabe Labortechnik GmbH (2025)
FDA stated the CAPA procedure did not adequately describe:
Root cause investigation methodology
Effective corrective action implementation
Verification of effectiveness
Use of statistical methods for trend detection
FDA then cited specific CAPAs as case studies of these systemic gaps.
Source: [8]
CAPA 23G4429-1 (Kabe example)
Initiated due to machine-damaged capillaries, the CAPA failed to:
Reference training evidence required by the action plan
Define statistically sound effectiveness criteria before closure
FDA’s criticism explicitly tied weak investigation and verification to continued manufacturing risk.
Source: [8]
Cross-letter synthesis (2025)
Independent reviews of multiple FDA warning letters emphasize that inadequate investigations lead directly to ineffective CAPAs and recurring failures, particularly where quality unit and governance deficiencies are left unaddressed.
Source: [9]
Ineffective Actions and Missing Effectiveness Verification
(Broken Control and Verification Layers)
Criticare Technologies (2024)
FDA emphasized that the CAPA procedure lacked instructions for effectiveness verification and that the firm’s response did not include sufficient detail or timelines to correct the CAPA system itself.
Source: [1]
Kabe Labortechnik (2025)
FDA explicitly linked missing or statistically unsupported effectiveness checks to ongoing manufacturing problems, using CAPAs as proof points.
Source: [8]
Industry trend analyses (2025)
Multiple analyses of recent warning letters identify a recurring enforcement theme: CAPAs closed without demonstrating sustained effectiveness, often tied to weak process validation and incomplete protocols.
Sources: [10], [11]
Drug and Combination-Product CAPA Themes (21 CFR 211 + 820)
Combination product warning letters
FDA cited CAPA deficiencies under 21 CFR 820.100 while also invoking 21 CFR 211.198, criticizing firms for recording defect counts without determining causes—underscoring expectations for systemic investigation and corrective action across regulatory frameworks.
Source: [12]
Pharmaceutical CGMP letters (e.g., Sanofi, 2025)
FDA framed CAPA failures through deficient investigations, deviation handling, and Quality Unit oversight, often requiring comprehensive, independent assessments of the investigation and CAPA system when failures are repetitive or shallow.
Sources: [3], [9]
Regulatory Takeaway (System Framing)
Across devices, drugs, and combination products, FDA uses CAPA failures to establish one conclusion:
If CAPA does not function as a closed-loop risk-control system, management does not control quality.
That conclusion is enforced under:
21 CFR 820.100(a) for devices
21 CFR Part 211 (investigations, QU responsibilities, and corrective action) for drugs
21 CFR Part 4 for combination products
—consistently aligned with FDA’s system-based inspection philosophy.
References
U.S. Food and Drug Administration (FDA).
Warning Letter: Criticare Technologies, Inc. July 12, 2024.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/criticare-technologies-inc-686915-07122024
U.S. Food and Drug Administration (FDA).
Warning Letter: Jiangsu Caina Medical Co., Ltd. July 18, 2024.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/jiangsu-caina-medical-co-ltd-687033-07182024
U.S. Food and Drug Administration (FDA).
Warning Letter: Sanofi. January 15, 2025.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/sanofi-690604-01152025
U.S. Food and Drug Administration (FDA).
Warning Letter: Contec Medical Systems Co., Ltd. October 2, 2025.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/contec-medical-systems-co-ltd-717941-10022025
U.S. Food and Drug Administration (FDA).
Warning Letter: iRhythm Technologies, Inc. May 25, 2023.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/irhythm-technologies-inc-643474-05252023
U.S. Food and Drug Administration (FDA).
Warning Letter: Royal Philips. September 9, 2025.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/royal-philips-709948-09092025
U.S. Food and Drug Administration (FDA).
Warning Letter: Exer Labs, Inc. February 10, 2025.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/exer-labs-inc-699218-02102025
U.S. Food and Drug Administration (FDA).
Warning Letter: Kabe Labortechnik GmbH. May 9, 2025.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/kabe-labortechnik-gmbh-704563-05092025
ECA Academy / GMP-Compliance.org.
Four Warning Letters Concerning CAPA and Root Cause Analysis Published. 2025.
https://www.gmp-compliance.org/gmp-news/four-warning-letter-concerning-capa-and-root-cause-analysis-published
SciLife.io.
Worst FDA Warning Letters in Pharma (CAPA and Investigation Failures). 2025.
https://www.scilife.io/blog/worst-fda-warning-letters-pharma
AssurX.
CAPA Non-Compliance in FDA Warning Letters.
https://www.assurx.com/capa-non-compliance-warning-letters/
ECA Academy / GMP-Compliance.org.
Warning Letter on Combination Products – 21 CFR Part 4.
https://www.gmp-compliance.org/gmp-news/warning-letter-on-combination-products-21-cfr-4
U.S. Food and Drug Administration (FDA).
Warning Letter: Noah Medical Corporation. April 9, 2025.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/noah-medical-corporation-702414-04092025
The FDA Group – Insider.
FDA Warns Four Device Makers Over Quality System Failures.
https://insider.thefdagroup.com/p/fda-warns-four-device-makers
Agarwal, N. (Substack).
Case Study: When Underestimating Risk Leads to an FDA Warning Letter.
https://naveenagarwalphd.substack.com/p/case-study-when-underestimating-risk-leads-to-fda-warning-letter
U.S. Food and Drug Administration (FDA).
Warning Letter: Epicare Acquisitions, LLC. March 21, 2025.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/epicare-acquisitions-llc-702493-03212025
BioSpace.
FDA Sends Warning Letter to Mindray Medical Over Manufacturing Problems.
https://www.biospace.com/fda-sends-warning-letter-to-mindray-medical-over-manufacturing-problems
The FDA Group – Insider.
How Quality Systems Fail at a Medical Device Firm.
https://insider.thefdagroup.com/p/quality-systems-fail-at-a-medical-device-firm
U.S. Food and Drug Administration (FDA).
Warning Letter: Globus Medical, Inc. July 15, 2024.
https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/globus-medical-inc-685606-07152024
Regulatory Affairs Professionals Society (RAPS).
FDA Issues Warning Letters for CGMP Violations. December 2025.
https://www.raps.org/news-and-articles/news-articles/2025/12/fda-issues-warning-letters-for-cgmp-violations-una