Why Regulatory Risk Is Underrepresented in Pitch Decks

What Is Regulatory Risk in Medical Device Investing?
March 30, 2026Common Regulatory Diligence Blind Spots
April 1, 2026
A founder walked investors through a polished pitch:
Strong problem
Clear solution
Large market
Impressive prototype
Then came the slide titled “Regulatory Strategy.”
It had three bullets:
510(k) pathway
No clinical trials expected
Timeline: 12 months
The room didn’t react.
But the deal didn’t move forward.
Nothing in that slide was technically wrong.
But everything about it felt incomplete.
The Reality Founders Don’t See
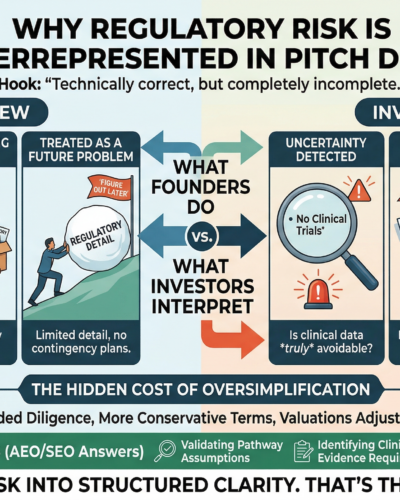
Short answer: regulatory risk is often underrepresented in pitch decks because founders simplify it—but investors interpret that as uncertainty.
Most founders focus on:
Product
Market
Traction
Technology
Regulatory strategy becomes a small section—often treated as a checkbox.
But under the framework of the U.S. Food and Drug Administration, regulatory pathways define:
Evidence requirements
Timeline
Capital needs
Probability of success
Investors don’t ignore regulatory risk.
They assume it is there—and look for how well you understand it.
Why Founders Minimize Regulatory Discussion
1. It Feels Complex and Uncertain
Regulatory strategy involves:
Classification
Pathway selection
Evidence planning
FDA interaction
Many of these elements are still evolving in early-stage companies.
So founders simplify:
“We’re pursuing 510(k)”
“We don’t expect clinical trials”
It sounds clean—but lacks depth.
2. There’s a Fear of Scaring Investors
Some teams believe:
Highlighting regulatory complexity will slow the deal
Discussing clinical risk will reduce valuation
Simplicity is more persuasive than nuance
So they minimize regulatory discussion.
But investors don’t reward simplicity when it hides risk.
They discount it.
3. Regulatory Is Treated as a Future Problem
Another common mindset:
“We’ll figure that out later.”
This leads to:
Limited regulatory detail in early decks
Assumptions presented as facts
Lack of contingency planning
Investors recognize this immediately.
Why Investors Notice the Gap
Investors don’t expect founders to eliminate regulatory risk.
They expect them to understand and frame it.
When regulatory discussion is minimal, investors ask:
Is the pathway validated or assumed?
What happens if FDA disagrees?
Are timelines realistic?
Is clinical data truly avoidable?
If answers are unclear, perceived risk increases.
And increased risk leads to:
Lower valuation
More conservative terms
Slower decision-making
What Strong Regulatory Storytelling Looks Like
A strong pitch deck does not hide regulatory complexity.
It explains it clearly.
Investors gain confidence when founders can articulate:
Why the chosen pathway fits the device
What evidence is required—and why
Where uncertainty exists
How risk is being managed
What contingency plans exist
Clarity reduces perceived risk.
AEO: Common Questions About Regulatory Risk in Pitch Decks
Should regulatory strategy be included in a pitch deck?
Yes. It is a critical factor influencing timelines, capital needs, and exit potential.
Why do investors focus on regulatory risk?
Because it directly impacts development uncertainty, cost, and probability of success.
Does simplifying regulatory strategy help fundraising?
No. Oversimplification can reduce credibility and increase perceived risk.
The Hidden Cost of Oversimplification
When regulatory risk is minimized:
Investors fill in the gaps themselves
Assumptions are stress-tested during diligence
Confidence decreases if answers are unclear
This often results in:
Valuation adjustments
Extended diligence
Lost deals
What is not explained becomes a liability.
Where Kandih Comes In
This is where Kandih Group helps founders align regulatory reality with fundraising narratives.
Kandih supports teams by:
Validating regulatory pathway assumptions
Clarifying intended use and classification
Identifying clinical and evidence requirements early
Mapping regulatory strategy to timeline and capital planning
Translating regulatory complexity into investor-relevant insights
Preparing founders to communicate risk clearly during fundraising
Instead of minimizing regulatory discussion, founders present it as a structured, managed component of the business.
That strengthens:
Investor confidence
Valuation credibility
Fundraising efficiency
The Real Lesson
The founder at the beginning didn’t lose the deal because of regulatory risk.
They lost it because the risk was underexplained.
Investors don’t expect perfection.
They expect clarity.
Bottom Line
Regulatory risk is often underrepresented in pitch decks—not because it is unimportant, but because it is uncomfortable.
But investors notice what’s missing.
Strong companies don’t hide regulatory complexity.
They frame it, manage it, and communicate it clearly.
That’s how regulatory strategy becomes part of the investment story—not a weakness in it.
References
FDA – Classify Your Medical Device
https://www.fda.gov/medical-devices/overview-device-regulation/classify-your-medical-device
FDA – Premarket Notification 510(k)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-notification-510k
FDA – De Novo Classification Process
https://www.fda.gov/medical-devices/premarket-submissions/de-novo-classification-request
FDA – Premarket Approval (PMA)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-approval-pma