FDA Recalls Cholesterol Medication — What Toxicology Can Teach Developers About Hidden Risks
Pacemakers – How Is a Pacemaker Placed?
October 24, 2025
Can LED Light Really Kill Cancer? What Toxicology Teaches Us About Safety, Innovation, and Smart Design
October 28, 2025FDA Recalls Cholesterol Medication — What Toxicology Can Teach Developers About Hidden Risks
When “Safe” Products Become a Safety Lesson
The U.S. Food and Drug Administration (FDA) recently announced a recall of several cholesterol-lowering medications after routine testing detected nitrosamine impurities — chemicals that can increase cancer risk following long-term exposure.
For patients, the immediate health risk is low.
For scientists, regulators, and developers, however, this is a powerful reminder:
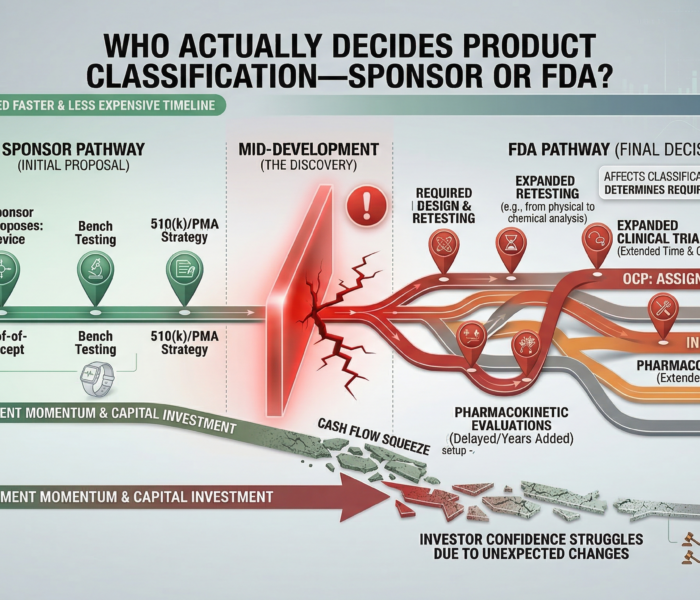
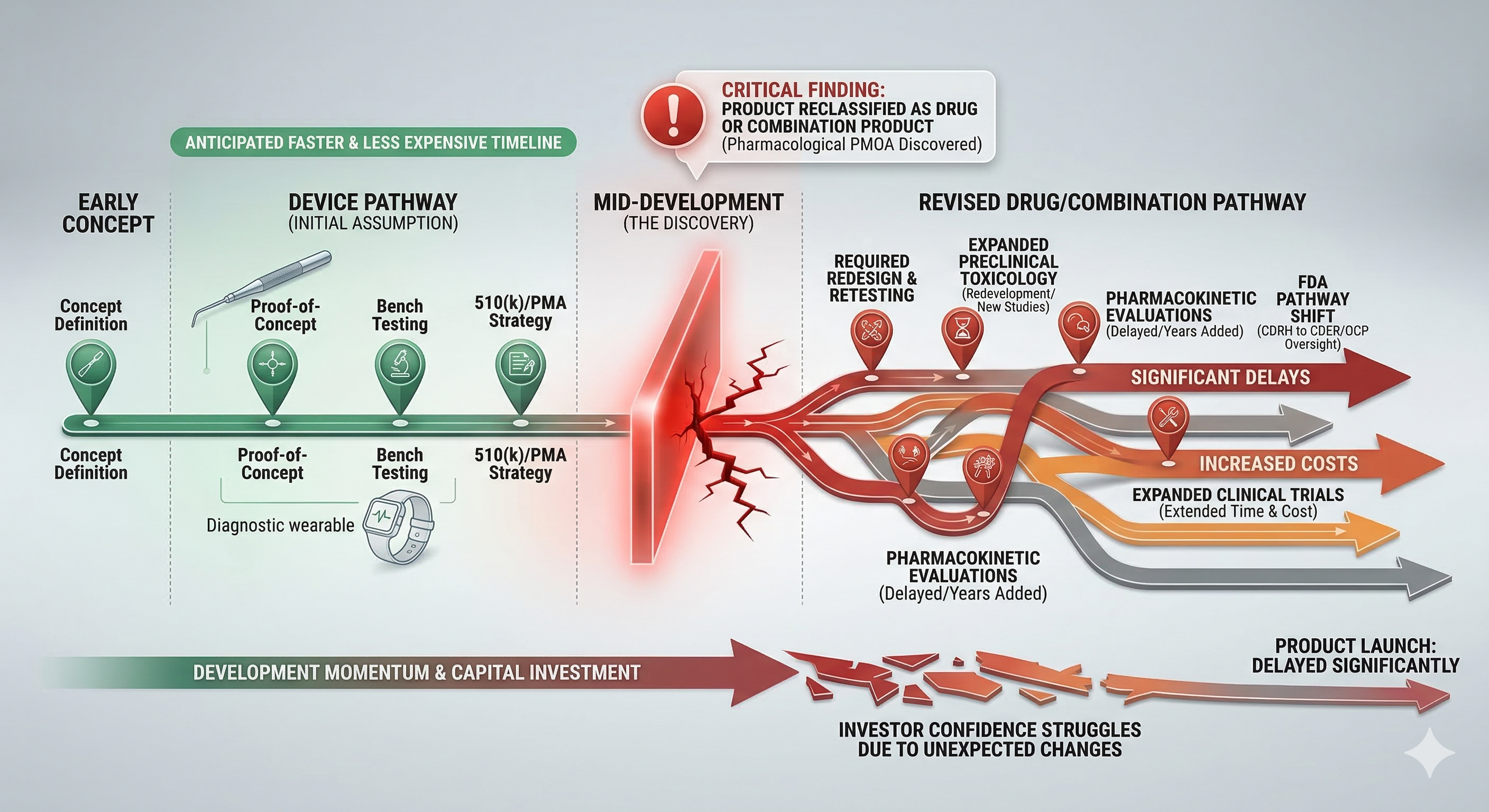
Even well-established drugs can fail if toxicology isn’t integrated early — and continuously revisited throughout a product’s lifecycle.
This recall isn’t just a manufacturing story.
It’s a toxicology case study — showing how chemistry, formulation, and vigilance intersect to protect public health.
The Recall: What Happened and Why It Matters
Nitrosamines — compounds such as NDMA (N-nitrosodimethylamine) and NDEA (N-nitrosodiethylamine) — are probable human carcinogens. They can form unintentionally during drug synthesis, packaging, or storage, particularly under heat and humidity.
These impurities have previously been detected in antihypertensives, antidiabetics, and now, statins — core therapies for cardiovascular disease.
The issue isn’t acute toxicity.
It’s chronic exposure — microgram-level doses that accumulate and cause DNA alkylation and mutagenesis over time.
The FDA’s recall underscores a timeless principle:
Toxicology doesn’t end after IND approval — it continues through process chemistry, manufacturing, and quality control.
How Toxicology Detects and Prevents Hidden Contaminants
Toxicologists are often invisible in the drug development narrative — yet they’re the reason these recalls happen before harm occurs.
Here’s how their expertise safeguards modern therapeutics:
1.
Understanding Impurity Mechanisms
Nitrosamines form through secondary amine–nitrite reactions, often accelerated by solvent residues, excipients, or packaging components.
Toxicologists collaborate with chemists to model degradation pathways and modify conditions that promote nitrosation — stopping problems before they start.
2.
Setting Toxicological Limits
Before regulators can define “safe” impurity thresholds, toxicologists establish acceptable intake levels (AILs) — daily exposures that carry a theoretical lifetime cancer risk of ≤1 in 100,000.
These assessments blend animal carcinogenicity data, structure–activity modeling, and exposure duration to anchor regulatory decisions.
3.
Translating Chemistry Into Risk Management
Analytical chemistry detects parts-per-billion levels of contaminants — but only toxicology interprets what those numbers mean for patient risk.
A few nanograms of NDMA could be trivial or trigger a recall, depending on dose, duration, and patient population.
4.
Informing Regulatory Action
FDA recall decisions hinge on toxicological interpretation:
Is the impurity dose-dependent, mechanistically genotoxic, or avoidable?
The stronger the toxicology data, the faster regulators can act with scientific confidence.
Product Development Takeaways: Toxicology as a Design Partner
This event reinforces that toxicology isn’t an endpoint — it’s a continuous design function embedded across development, manufacturing, and post-market surveillance.
Design with Degradation in Mind
Model potential impurity formation during synthesis, formulation, and storage.
Conduct forced-degradation studies under stress conditions to reveal early toxic liabilities.
Integrate Toxicology into Supply Chain Review
Evaluate solvents, excipients, catalysts, and packaging for nitrosating potential.
Build vendor qualification systems that include toxicology risk evaluation.
Leverage Predictive Toxicology Tools
Use in silico modeling (e.g., DEREK Nexus, OECD QSAR Toolbox) to forecast mutagenic potential before scale-up.
Confirm results through in vitro genotoxicity assays and in vivo validation.
Plan for Lifecycle Monitoring
Establish post-market toxicology reviews as part of stability testing.
Partner with analytical labs for routine impurity trend analysis and early warning detection.
Rooted in Experience
In one consulting engagement, a polymer-coated oral formulation showed excellent stability — until a packaging material change introduced trace nitrosamine formation.
The toxicology review identified the issue before launch, prompting a packaging redesign that eliminated the contaminant entirely.
That’s the real value of toxicology — not reaction, but prevention.
The Bigger Picture: Toxicology as a Risk Compass
Cholesterol drugs save millions of lives every year.
This recall doesn’t discredit their value — it reinforces a critical truth:
Toxicology isn’t about identifying harm — it’s about predicting, preventing, and designing it out.
For developers, continuous toxicological integration isn’t just regulatory housekeeping.
It’s brand protection, patient trust, and scientific integrity — all built on a foundation of foresight.
References
1. U.S. Food and Drug Administration (FDA). FDA Updates and Press Announcements on Nitrosamine Impurities in Medications. https://www.fda.gov/drugs/drug-safety-and-availability/information-about-nitrosamine-impurities-medications
2. Centers for Disease Control and Prevention (CDC). Nitrosamines and Human Health. https://stacks.cdc.gov/view/cdc/103627
3. European Medicines Agency (EMA). Assessment Report: Nitrosamine Impurities in Medicines. EMA/369136/2020. https://www.ema.europa.eu/en/documents/report/report-european-medicines-regulatory-networks-response-nitrosamine-impurities-human-medicines_en.pdf
4. Graham DJ, et al. Statin Safety and Toxicology: Lessons from Regulatory Surveillance. Drug Saf. 2022;45(9):875–888. https://www.sciencedirect.com/science/article/abs/pii/S0002914905021430
5. U.S. Pharmacopeia (USP). Guidance on Impurity Testing and Nitrosamine Control. 2023 Edition. https://qualitymatters.usp.org/nitrosamine-impurities-latest-usp-tool-further-aids-understanding-and-control