Do You Trend Complaints Before CAPA?

Complaint → Weak Root Cause → Repeat 483
January 21, 2026
The Complaint-to-CAPA Mapping Checklist
January 23, 2026Yes—FDA expects complaint trending before CAPA initiation.
From an inspection perspective, complaint trending is a diagnostic function, not an effectiveness check. If trending only occurs after CAPA opens, FDA assumes the escalation decision was not risk-informed and that the system reacted without understanding the signal.
Why This Question Matters More Than Most Teams Realize
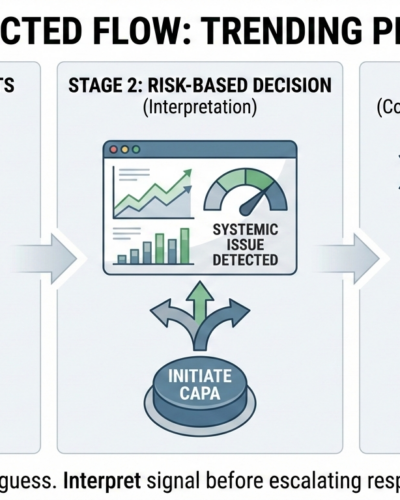
If CAPA is your first tool, you already missed the signal.
Many organizations treat CAPA as the mechanism that reveals trends.
FDA expects CAPA to respond to interpreted risk, not raw complaint noise.
Key mismatch:
Organizations: “We trend complaints after CAPA to show it worked.”
FDA: “Why didn’t you understand the signal before you escalated?”
From an inspection standpoint, initiating CAPA without prior trending is not proactive control.
It is reactive guesswork.
The FDA Systems Perspective
Complaints are leading indicators.
CAPA is a control response.
Between the two sits a critical system function:
Trend Analysis
The U.S. Food and Drug Administration does not expect CAPA to compensate for weak signal interpretation. Inspectors expect to see that complaint data is:
Aggregated across time
Grouped by failure mode
Stratified by product, use condition, and severity
Interpreted before escalation decisions
If trending appears only after CAPA opens, FDA assumes the decision to escalate—or not escalate—was not risk-based.
What FDA Inspectors Actually Evaluate
Inspectors are not checking whether you have a trending SOP.
They evaluate whether trending influences decisions.
Specifically, FDA looks for evidence that:
Complaint trends are reviewed on a defined cadence
CAPA initiation is tied to trend behavior, not anecdotes
Similar complaints are grouped and escalated consistently
The absence of CAPA is defensible using documented trend data
When inspectors ask:
“Why wasn’t CAPA opened earlier?”
They are asking for your signal-interpretation logic, not your closure metrics.
Failure to show that logic is interpreted as loss of control.
Why This Fails Systemically (Not Procedurally)
1. Complaint Handling Is Treated as Case Management
Each complaint is investigated, closed, and filed—independently.
FDA interpretation:
You are managing records, not risk.
2. CAPA Becomes the Diagnostic Tool
Organizations rely on CAPA to discover trends that should have been visible upstream.
FDA interpretation:
Your system requires escalation to understand itself. That is backward.
3. Trending Is Used to Justify Closure
Trending is performed only to prove CAPA worked.
FDA interpretation:
You are verifying paperwork, not restoring control.
Administrative closure may be complete.
Regulatory effectiveness is not.
The Regulatory Insight (Clear and Uncomfortable)
If you cannot show that complaint trends informed the CAPA decision, FDA assumes the decision was arbitrary.
Arbitrary escalation—or failure to escalate—is viewed by FDA as a system weakness, not a judgment call.
This is why inspections often cite:
“Failure to adequately analyze sources of quality data.”
That language is not about missing charts.
It is about missing signal interpretation.
The Design Principle (Your One Takeaway)
Trending belongs upstream of CAPA—not downstream of it.
Trending answers:
Is this noise, drift, or systemic failure?
CAPA answers:
Which control failed, and how do we restore it?
If your system cannot clearly show how complaint trends triggered—or did not trigger—CAPA, then CAPA initiation is not designed.
It is improvised.
FAQ
Q: Does FDA require complaint trending before CAPA?
A: FDA expects complaint data to be trended and interpreted before CAPA decisions are made. Trending after CAPA is not sufficient.
Q: Is complaint trending part of CAPA effectiveness?
A: No. Trending is a diagnostic activity. Effectiveness is demonstrated by post-CAPA outcome changes.
Q: What happens if CAPA is opened without prior trending?
A: FDA may interpret the escalation decision as arbitrary and the system as reactive.
Q: Can FDA cite lack of trending even if CAPA exists?
A: Yes. FDA frequently cites failure to analyze quality data even when CAPAs are present.
If your team struggles to explain why CAPA started when it did—or why it didn’t, that is a diagnostic signal worth taking seriously.
That is where system-level review, CAPA remediation, and quality architecture redesign create regulatory predictability.
If your CAPA system can’t explain itself to an inspector, it’s not designed yet.
References:
U.S. Food and Drug Administration — 21 CFR §820.100(a)
Corrective and Preventive Action
Requires analysis of quality data sources before CAPA initiation to identify existing and potential causes of nonconforming product.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-820/section-820.100
U.S. Food and Drug Administration — 21 CFR §820.198
Complaint Files
Establishes complaints as a formal quality data source that must be evaluated for trends and systemic issues—not merely investigated individually.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-820/section-820.198
U.S. Food and Drug Administration — Quality System Inspection Technique (QSIT)
FDA’s inspection playbook describing how investigators trace complaints → data analysis → CAPA → management controls.
International Council for Harmonisation — ICH Q10: Pharmaceutical Quality System
Positions CAPA as a response to interpreted quality signals, supported by continual monitoring and trending.
https://database.ich.org/sites/default/files/Q10_Guideline.pdf