7 Costly FDA Mistakes EU Medical Device Companies Make

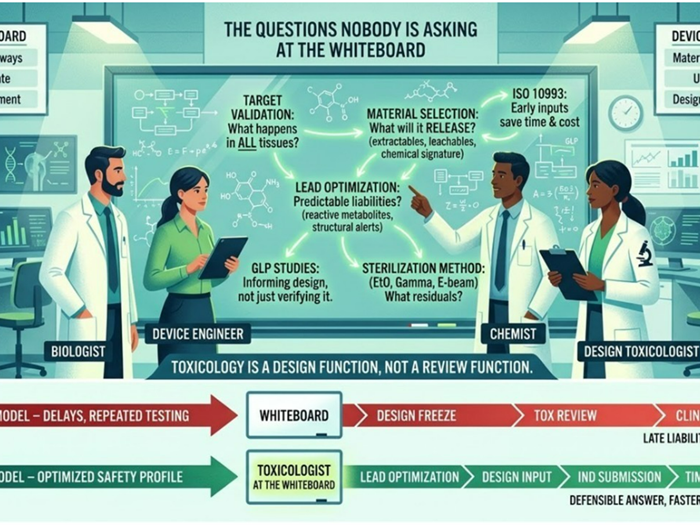
Infant Botulism Outbreak Linked to Infant Formula: A Toxicology & Product-Design Alarm
November 10, 2025
Portable Breast MRI
November 11, 2025Entering the U.S. market requires a fundamental shift from the EU’s CE Marking philosophy. Underestimating the differences in the FDA’s regulatory framework leads to costly delays, Refuse-to-File letters, and Form 483s.
Here are the top 7 regulatory and quality mistakes EU device companies make:
1. Assuming CE Marking Strategy Translates to FDA Strategy
- The Mistake: Treating the FDA’s “Substantial Equivalence” (510(k)) or “Safety & Effectiveness” (PMA) requirements as a simple translation of your CE Marking Technical File. The data requirements, predicate device justification, and clinical evidence expectations are fundamentally different.
- The Fix: Conduct a pre-submission gap analysis against the precise FDA requirements for your device class and regulatory pathway. Do not assume your existing clinical data will be sufficient.
2. Underestimating Data Integrity (ALCOA+) and Part 11 Compliance
- The Mistake: Relying on EU data governance practices that don’t meet the FDA’s stringent ALCOA+ standard for all GxP data. This is especially critical for electronic records (21 CFR Part 11), where missing audit trails, shared logins, or inadequate system validation are major red flags.
- The Fix: Implement and validate electronic systems (eQMS, ERP, LMS) with robust data integrity controls before generating data for your U.S. submission. Prepare for FDA inspectors to scrutinize audit trails.
3. Treating ISO 13485 as an FDA-QSR Substitute
- The Mistake: Believing an ISO 13485-certified QMS is fully compliant with 21 CFR Part 820 (Quality System Regulation). While the new QMSR rule aligns Part 820 with ISO 13485, specific FDA expectations for the Device Master Record (DMR), Design History File (DHF), and complaint handling files remain distinct and are heavily emphasized in inspections.
- The Fix: Create a detailed gap analysis/cross-reference matrix between your QMS and 21 CFR Part 820. Conduct mock FDA audits focused on these specific record sets.
4. Submitting an Unsupported Indications for Use Statement
- The Mistake: Carrying over a broad, commercially-driven “Intended Purpose” from the EU labeling. The FDA requires a precise, narrow Indications for Use statement. An overreaching claim is the fastest way to derail a 510(k) or De Novo, as it breaks the link to your chosen predicate and questions the adequacy of your data.
- The Fix: Define your U.S. Indications for Use first, based on the data you have or will generate. Use a least-burdensome approach: get cleared for a narrow indication and plan for future supplements to expand the label.
5. Ineffective U.S. Implementation of SOPs
- The Mistake: Having SOPs that are not adequately implemented for U.S. operations. This includes failures in training, translation, or ensuring U.S. personnel consistently follow procedures. “Failure to establish/maintain procedures” is a top FDA 483 observation.
- The Fix: Ensure all critical SOPs (e.g., CAPA, Management Review, Complaint Handling) are implemented, training is documented, and compliance is verified by an independent Quality function. Assume the FDA will check this.
6. A Non-Compliant U.S. Post-Market Surveillance System
- The Mistake: Applying EU MDR vigilance timelines and processes to U.S. Medical Device Reporting (MDR). The FDA’s reporting timelines (e.g., 30-day for serious injuries, 5-day for deaths) are stricter, and the forms (e.g., Form 3500A) are unique. Failure to report is a severe enforcement target.
- The Fix: Establish a dedicated U.S. Post-Market system, appoint a U.S. Agent, and train your global vigilance team specifically on 21 CFR Part 803. Integrate this process with your CAPA system.
7. Misclassifying the Device or Choosing a Weak Predicate
- The Mistake: Incorrectly self-classifying your device or selecting an obsolete or poorly matched predicate for a 510(k). A flawed predicate strategy is a critical error that can invalidate your entire submission, forcing you to start over or pursue a more burdensome pathway (like a De Novo).
- The Fix: Use the FDA’s Product Classification database and guidance documents. Engage regulatory counsel or consultants early to validate your device classification and predicate device selection before you write the first page of your submission.