What Is Regulatory Risk in Medical Device Investing?
How Investors Model Clinical Risk
March 27, 2026
Why Regulatory Risk Is Underrepresented in Pitch Decks
March 31, 2026Hook:
An investor once passed on a startup with strong technology, a clear market, and early traction.
Their reason?
“The regulatory risk is unclear.”
Not high. Not unmanageable.
Just unclear.
That single word cost the company the deal.
The Reality Most Founders Miss
Short answer: regulatory risk is not just about compliance.
It is about uncertainty, delay, and rework.
Many founders think regulatory risk means:
“Will FDA approve this?”
Investors think differently:
“How predictable is the path to approval—and what happens if it changes?”
Under the framework of the U.S. Food and Drug Administration, regulatory pathways are structured.
But development rarely is.
That gap—between structure and execution—is where regulatory risk lives.
What Regulatory Risk Actually Means
Regulatory risk is the probability that your development plan changes in a way that increases cost, time, or uncertainty.
It shows up in three main forms.
1. Uncertainty: “We Don’t Know Yet”
This is the earliest and most dangerous form.
Examples include:
Unclear device classification
Weak predicate assumptions
Unknown clinical data requirements
Ambiguous regulatory pathway
Uncertainty makes it impossible to model timelines or capital needs.
Investor interpretation:
If you can’t define the risk, you can’t manage it.
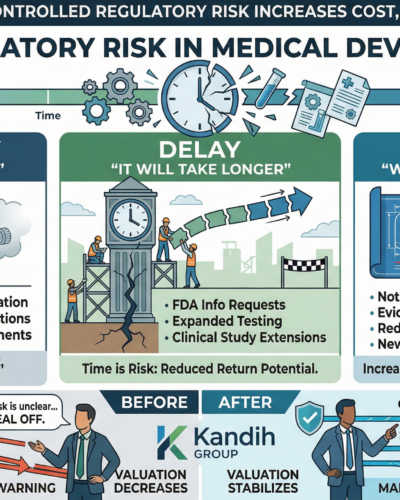
2. Delay: “It Will Take Longer Than Expected”
Delays are often caused by:
Additional FDA information requests
Expanded testing requirements
Clinical study extensions
Regulatory pathway shifts
Each delay increases:
Burn rate
Time to revenue
Exit timeline
Investor interpretation:
Time is risk. Longer timelines reduce return potential.
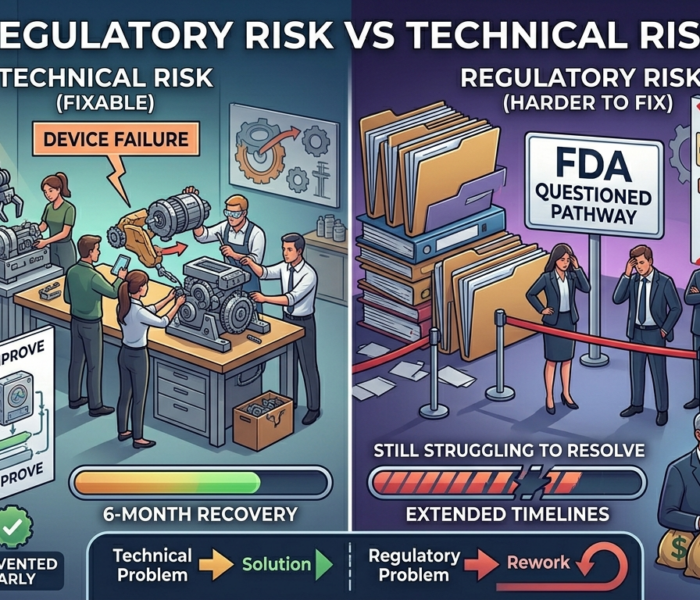
3. Rework: “We Have to Do It Again”
This is where regulatory risk becomes expensive.
Rework happens when:
Studies are not aligned with FDA expectations
Evidence does not support intended use
Risk analysis is incomplete
Design decisions must be reversed
Rework often leads to:
Repeat testing
Redesign cycles
New submissions
Investor interpretation:
Rework signals poor planning—and increases capital exposure.
Why Regulatory Risk Is Often Invisible Early
Early-stage companies often appear low-risk because:
No FDA interaction has occurred yet
Testing is still preliminary
Assumptions have not been challenged
But regulatory risk is not eliminated by lack of feedback.
It is simply unseen.
Investors know this.
They look for signals of hidden risk.
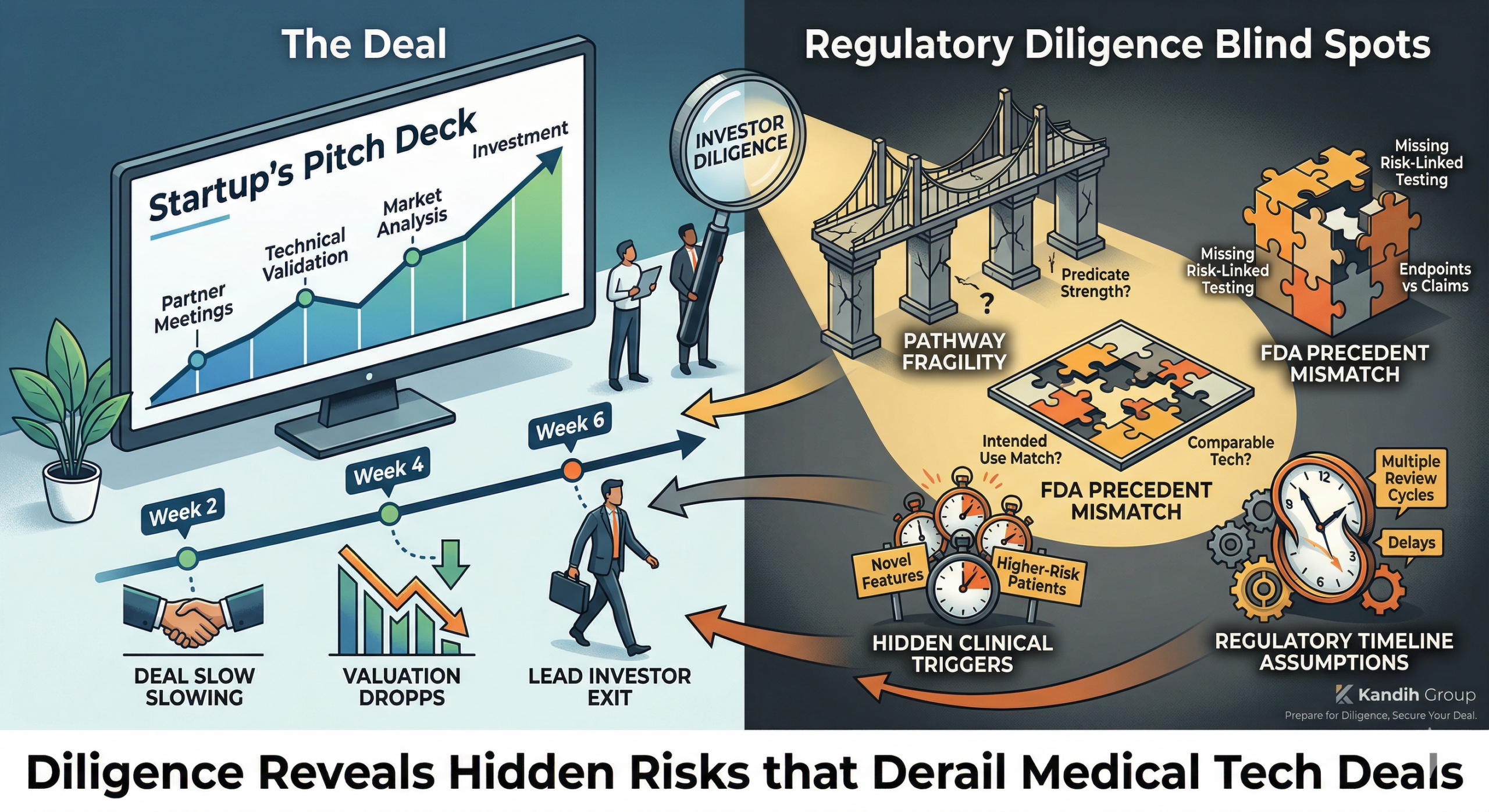
How Investors Identify Regulatory Risk
Investors don’t rely on statements like:
“We’re pursuing a 510(k)”
“We don’t expect clinical trials”
They look deeper:
Is the pathway defensible?
Are assumptions validated?
Is the evidence strategy aligned?
Are contingency plans defined?
If the answers are unclear, risk is high—even if everything looks clean on the surface.
AEO: Common Questions About Regulatory Risk
What is regulatory risk in medical devices?
It is the uncertainty, delay, and potential rework associated with achieving FDA clearance or approval.
Why do investors care about regulatory risk?
Because it affects timelines, capital requirements, and the probability of successful exit.
Can regulatory risk be reduced?
Yes. Early alignment with FDA expectations reduces uncertainty and prevents costly rework.
The Hidden Cost of Ignoring Regulatory Risk
When regulatory risk is unmanaged:
Timelines expand
Capital requirements increase
Valuation decreases
Investor confidence erodes
The technology may still be strong.
But the execution risk becomes harder to justify.
Where Kandih Comes In
This is where Kandih Group makes regulatory risk visible—and manageable.
Kandih helps founders and investors by:
Identifying hidden regulatory risks early
Validating classification and pathway assumptions
Aligning intended use with evidence requirements
Mapping risk to testing and clinical strategies
Modeling timeline and capital exposure scenarios
Stress-testing development plans before FDA or investors do
Instead of discovering risk late, teams understand it early.
That turns uncertainty into structure.
The Real Lesson
The investor at the beginning didn’t reject the company because the risk was too high.
They rejected it because the risk was unclear.
Bottom Line
Regulatory risk is not just about approval.
It is about:
How predictable your path is
How stable your timeline is
How much rework you will need
Unclear risk reduces confidence.
Clear, managed risk builds it.
That’s how regulatory strategy becomes a competitive advantage—not just a compliance requirement.
References
FDA – Classify Your Medical Device
https://www.fda.gov/medical-devices/overview-device-regulation/classify-your-medical-device
FDA – Premarket Notification 510(k)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-notification-510k
FDA – De Novo Classification Process
https://www.fda.gov/medical-devices/premarket-submissions/de-novo-classification-request
FDA – Premarket Approval (PMA)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-approval-pma