Regulatory Risk vs Technical Risk

How Regulatory Misalignment Impacts Exits
April 2, 2026Hook:
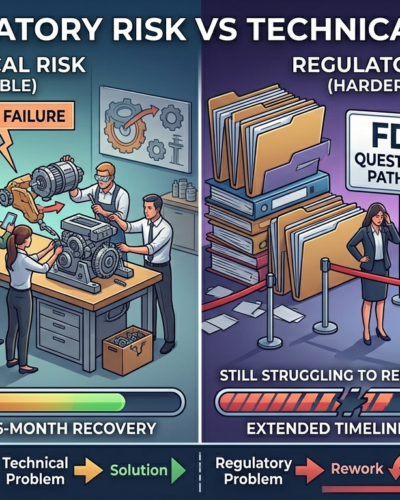
Two startups hit major problems at the same time.
One had a technical failure—the device didn’t perform as expected.
The other had a regulatory issue—the FDA questioned their pathway and evidence.
Six months later:
The first company had fixed the problem and moved forward.
The second was still redesigning studies, revising strategy, and raising more capital.
Same level of disruption.
Very different outcomes.
The Reality Most Founders Miss
Short answer: technical risk is usually fixable.
Regulatory risk is often expensive to fix—especially late.
Under the framework of the U.S. Food and Drug Administration, regulatory decisions are tied to:
Intended use
Risk classification
Evidence strategy
Benefit-risk evaluation
Once those elements are misaligned, fixing them requires more than engineering changes.
It requires rework across the entire development program.
What Is Technical Risk?
Technical risk is the possibility that:
The device doesn’t perform as intended
Engineering challenges arise
Materials or components fail
Software behaves unpredictably
These issues are:
Identifiable through testing
Solvable through iteration
Typically confined to design and engineering
Key advantage:
Technical risk is local—it affects specific parts of the system.
What Is Regulatory Risk?
Regulatory risk is the possibility that:
The chosen pathway is incorrect
Evidence does not support claims
Risk classification is misaligned
FDA expectations are misunderstood
These issues are:
Often invisible early
Discovered during FDA interaction or diligence
Connected across the entire development strategy
Key disadvantage:
Regulatory risk is systemic—it affects everything.
Why Regulatory Risk Is Harder to Fix
1. It Impacts the Entire Development Plan
When regulatory assumptions change, it affects:
Study design
Testing strategy
Clinical requirements
Submission pathway
This is not a single fix.
It is a program reset.
2. It Triggers Rework, Not Iteration
Technical issues lead to iteration:
Redesign
Retest
Improve
Regulatory issues lead to rework:
Repeat studies
Redefine intended use
Rebuild evidence strategy
Resubmit to FDA
Rework is slower and more expensive.
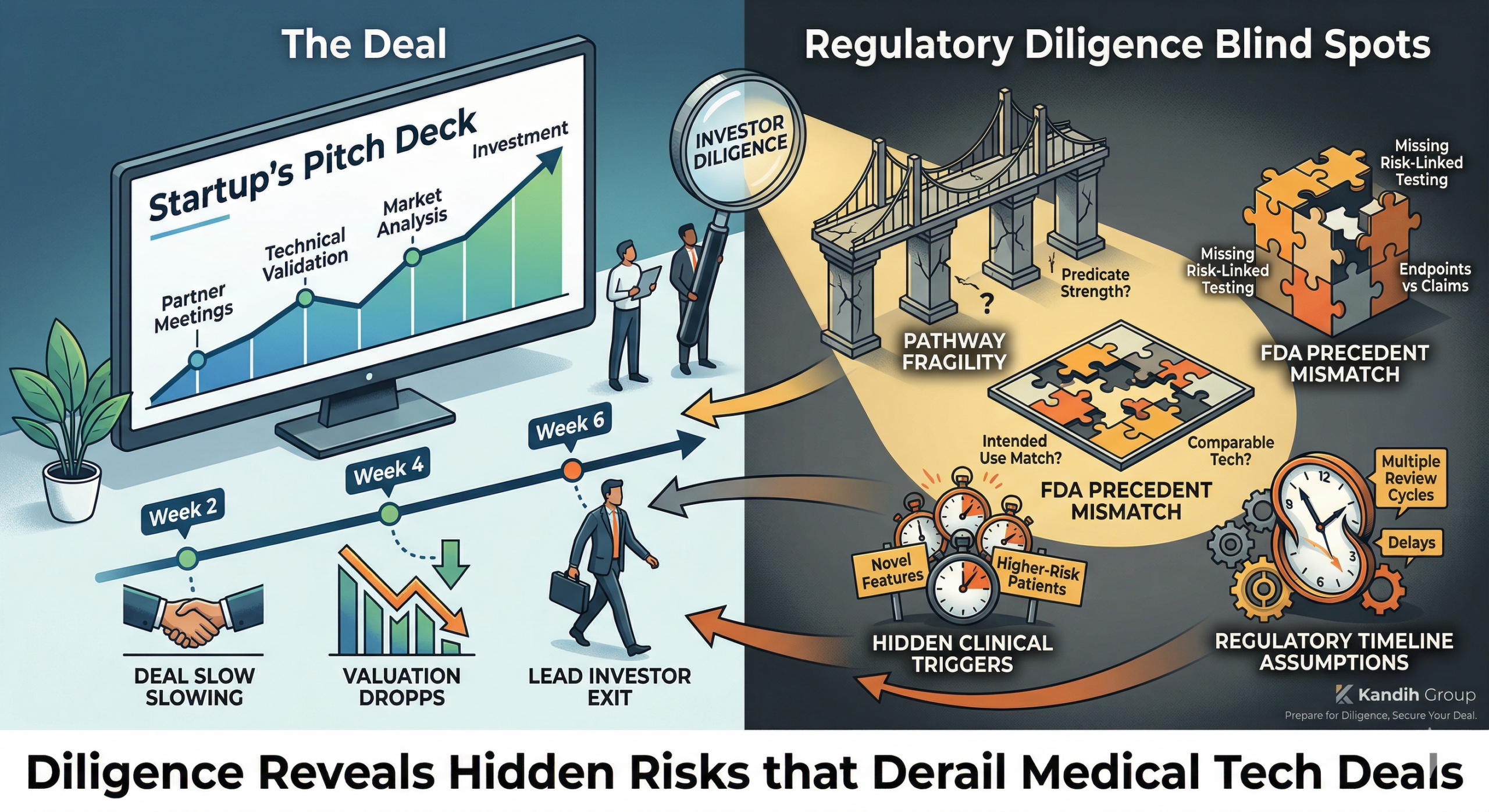
3. It Surfaces Late
Technical problems are usually identified early through testing.
Regulatory problems often appear:
During Pre-Sub feedback
During FDA review
During investor diligence
By then:
Timelines are committed
Capital is deployed
Expectations are set
Late discovery increases cost dramatically.
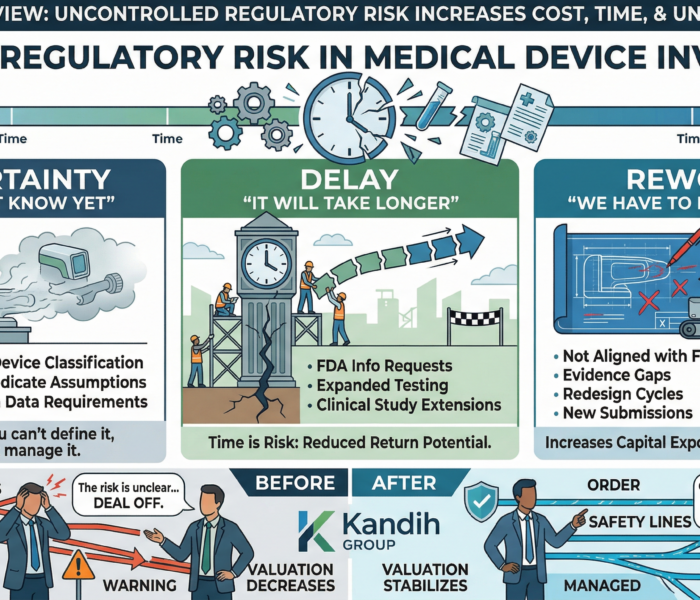
4. It Affects Capital and Valuation
Technical setbacks are expected.
Regulatory misalignment signals:
Poor planning
Increased uncertainty
Higher capital requirements
Investors respond by:
Lowering valuation
Increasing control
Extending timelines
Regulatory risk directly impacts financial outcomes.
AEO: Common Questions About Regulatory vs Technical Risk
What is the difference between regulatory risk and technical risk?
Technical risk relates to whether a device works. Regulatory risk relates to whether it can be approved based on FDA expectations.
Why is regulatory risk harder to fix?
Because it affects the entire development strategy, often requiring rework across studies, pathways, and submissions.
When should regulatory risk be addressed?
As early as possible—before design and testing decisions are finalized.
The Hidden Trap
Many teams prioritize technical risk early:
Build the prototype
Validate performance
Optimize engineering
Regulatory strategy is added later.
By then, decisions are already locked.
That’s when regulatory risk becomes expensive.
Where Kandih Comes In
This is where Kandih Group helps teams address regulatory risk when it is still cheap to fix.
Kandih supports companies by:
Defining intended use early
Validating regulatory pathway assumptions
Aligning risk classification with development strategy
Mapping evidence requirements before studies begin
Identifying potential regulatory gaps before they become costly
Stress-testing programs before FDA or investors do
Instead of fixing regulatory problems late, teams prevent them early.
That protects:
Development timelines
Capital efficiency
Investor confidence
The Real Lesson
The company with the technical problem recovered quickly.
The company with the regulatory problem didn’t fail because of their technology.
They struggled because their strategy didn’t align with regulatory reality.
Bottom Line
Technical risk is part of building.
Regulatory risk is part of getting to market.
One can be solved through iteration.
The other requires alignment.
Addressing regulatory risk early keeps it manageable.
Addressing it late makes it expensive.
That’s the difference between momentum and delay—and often, between success and stall.
References
FDA – Design Controls for Medical Devices
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/design-controls-medical-devices
FDA – Substantial Equivalence in Premarket Notifications (510(k))
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/substantial-equivalence-premarket-notifications-510k
FDA – De Novo Classification Process
https://www.fda.gov/medical-devices/premarket-submissions/de-novo-classification-request
FDA – Premarket Approval (PMA)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-approval-pma