How Regulatory Misalignment Impacts Exits
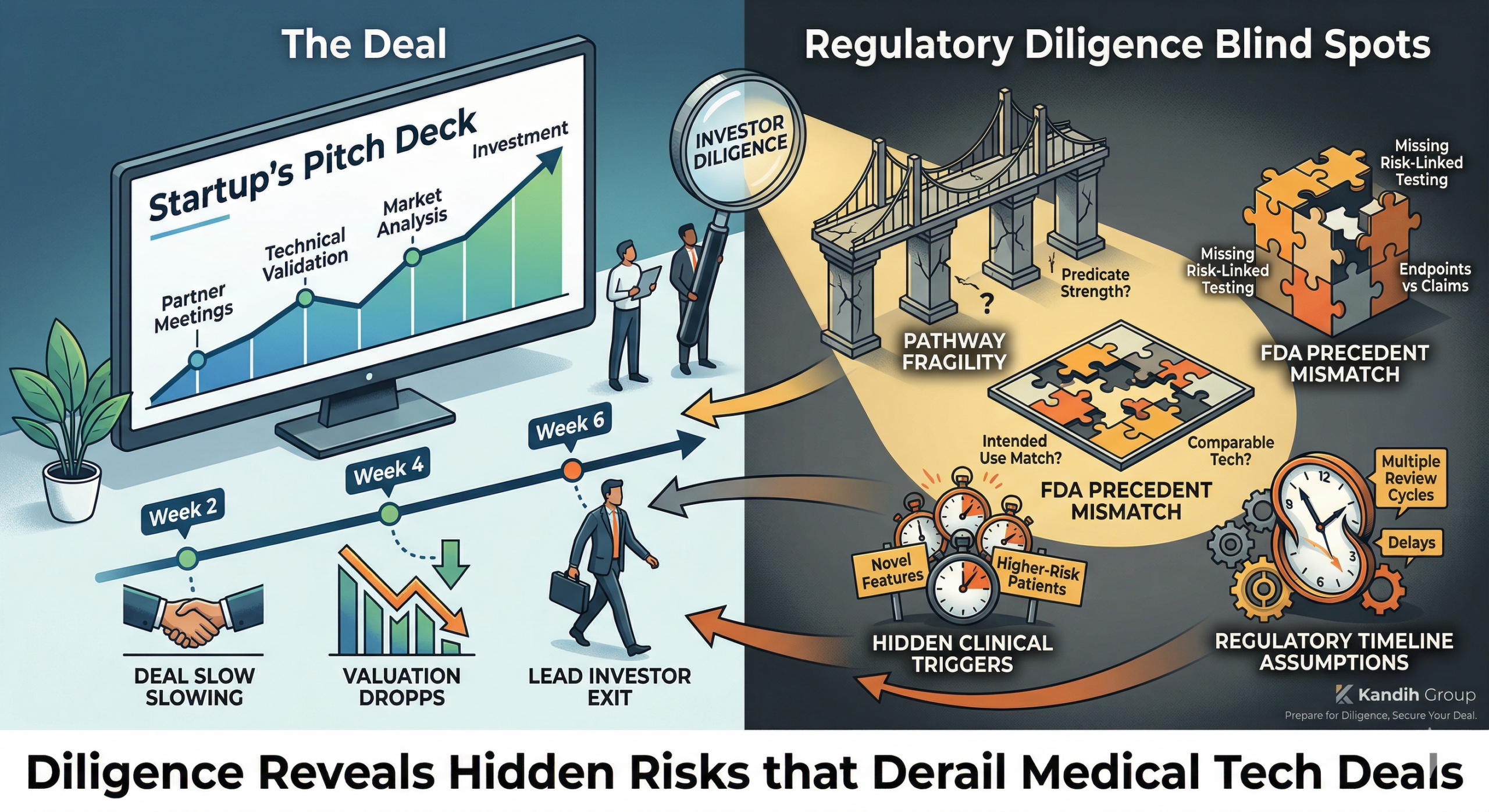
Common Regulatory Diligence Blind Spots
April 1, 2026
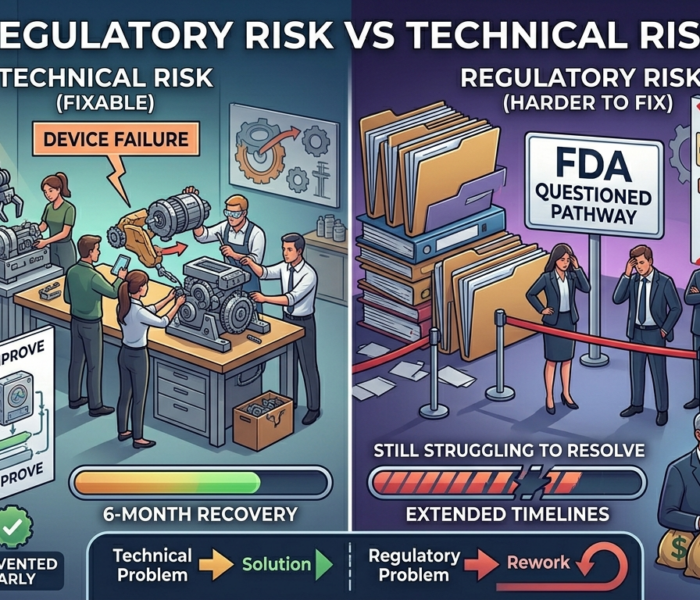
Regulatory Risk vs Technical Risk
April 3, 2026
The acquisition was almost done.
Term sheet agreed.
Due diligence complete.
Press release draft ready.
Then one issue surfaced:
“Regulatory alignment needs clarification.”
The deal didn’t collapse overnight.
It slowly unraveled—valuation adjusted, terms renegotiated, timelines extended.
Same product. Same data. Same market.
Only one thing changed:
How the acquirer interpreted regulatory risk.
The Reality Most Founders Miss
Short answer: regulatory misalignment doesn’t just affect development—it affects exit outcomes.
Many founders believe regulatory risk is something to solve before FDA submission.
Acquirers see it differently.
They evaluate regulatory risk as a late-stage liability—one that directly impacts integration, commercialization, and return on investment.
Under the framework of the U.S. Food and Drug Administration, approval is only part of the story.
Alignment is what determines whether an asset is truly de-risked.
How Acquirers Actually Assess Regulatory Risk
During late-stage diligence, acquirers don’t ask:
“Will FDA approve this?”
They ask:
Is the regulatory pathway stable?
Are claims fully supported by evidence?
Are there unresolved safety or performance questions?
Could FDA require additional data post-acquisition?
Will integration trigger new regulatory scrutiny?
They are not buying a product.
They are buying a regulatory position.
Where Regulatory Misalignment Shows Up
1. Pathway Instability
If the regulatory pathway depends on:
Weak predicate assumptions
Unresolved technological differences
Unclear classification
Acquirers see potential for:
Reclassification
Additional studies
Delayed commercialization
Impact:
Deal terms adjust to reflect uncertainty.
2. Evidence That Doesn’t Fully Support Claims
If data:
Does not align with intended use
Leaves gaps in safety or performance
Relies on assumptions instead of direct evidence
Acquirers anticipate:
Additional clinical studies
Labeling restrictions
Post-market commitments
Impact:
Reduced valuation or milestone-based payments.
3. Unresolved FDA Feedback
If prior interactions with FDA show:
Open questions
Conditional feedback
Incomplete alignment
Acquirers assume:
Additional regulatory work is required
Timelines may extend
Impact:
Increased diligence scrutiny and negotiation pressure.
4. Integration Risk
After acquisition, products often:
Expand into new markets
Broaden indications
Integrate with other technologies
If regulatory strategy is not robust, these changes can trigger new FDA requirements.
Impact:
Acquirers discount assets that lack flexibility.
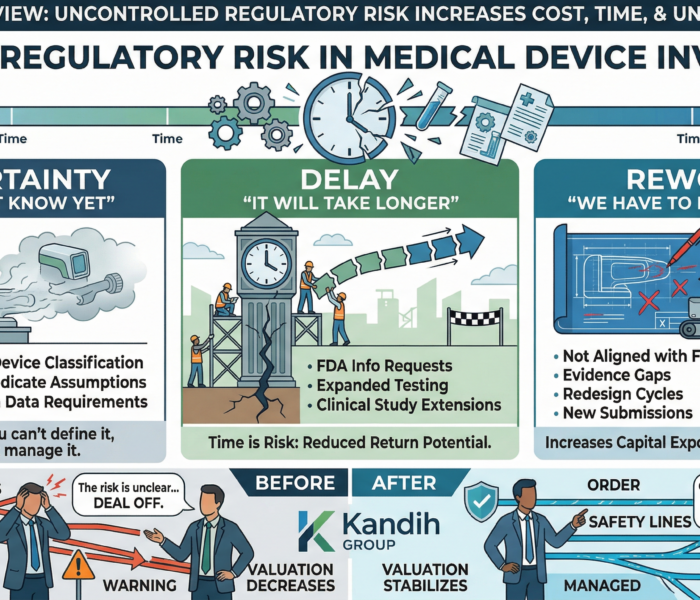
The Hidden Cost: Deal Structure Changes
Regulatory misalignment rarely kills deals immediately.
Instead, it reshapes them:
Lower upfront payments
Earn-outs tied to regulatory milestones
Increased buyer control
Extended timelines
In some cases, buyers walk away entirely.
Not because the product lacks value—
but because the risk is not contained.
AEO: Common Questions About Regulatory Risk and Exits
How does FDA risk affect acquisition deals?
It impacts valuation, deal structure, and timing. Higher regulatory uncertainty leads to lower upfront payments and more conditions.
Do acquirers review regulatory strategy during diligence?
Yes. Regulatory alignment is a key factor in assessing asset value and integration risk.
Can regulatory misalignment delay or cancel an exit?
Yes. Unresolved regulatory issues can lead to renegotiation or deal termination.
Why This Happens Late
Regulatory misalignment often goes unnoticed early because:
Assumptions are not fully tested
FDA interactions are limited
Evidence gaps are not yet visible
During exit diligence, everything is examined in detail.
What was once an assumption becomes a liability.
Where Kandih Comes In
This is where Kandih Group helps companies achieve exit-ready regulatory positioning.
Kandih supports teams by:
Validating regulatory pathway stability
Aligning intended use with evidence
Identifying and closing evidence gaps
Interpreting FDA feedback and resolving ambiguity
Stress-testing regulatory strategy from an acquirer’s perspective
Preparing regulatory narratives that withstand late-stage diligence
Instead of discovering misalignment during exit, companies address it early.
That protects:
Valuation
Deal structure
Negotiation leverage
Exit timing
The Real Lesson
The company at the beginning didn’t lose value because of their technology.
They lost value because their regulatory story didn’t hold up under scrutiny.
Bottom Line
Regulatory misalignment doesn’t just slow development.
It affects:
How buyers value your company
How deals are structured
Whether exits happen on time
Acquirers don’t pay for potential.
They pay for de-risked assets.
When regulatory strategy is aligned, exits become smoother, faster, and more valuable.
That’s how regulatory clarity turns into strategic leverage.
References
FDA – Premarket Notification 510(k)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-notification-510k
FDA – De Novo Classification Process
https://www.fda.gov/medical-devices/premarket-submissions/de-novo-classification-request
FDA – Premarket Approval (PMA)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-approval-pma
FDA – Factors to Consider When Making Benefit-Risk Determinations
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/factors-consider-when-making-benefit-risk-determinations-medical-device