Common Regulatory Diligence Blind Spots

Why Regulatory Risk Is Underrepresented in Pitch Decks
March 31, 2026
How Regulatory Misalignment Impacts Exits
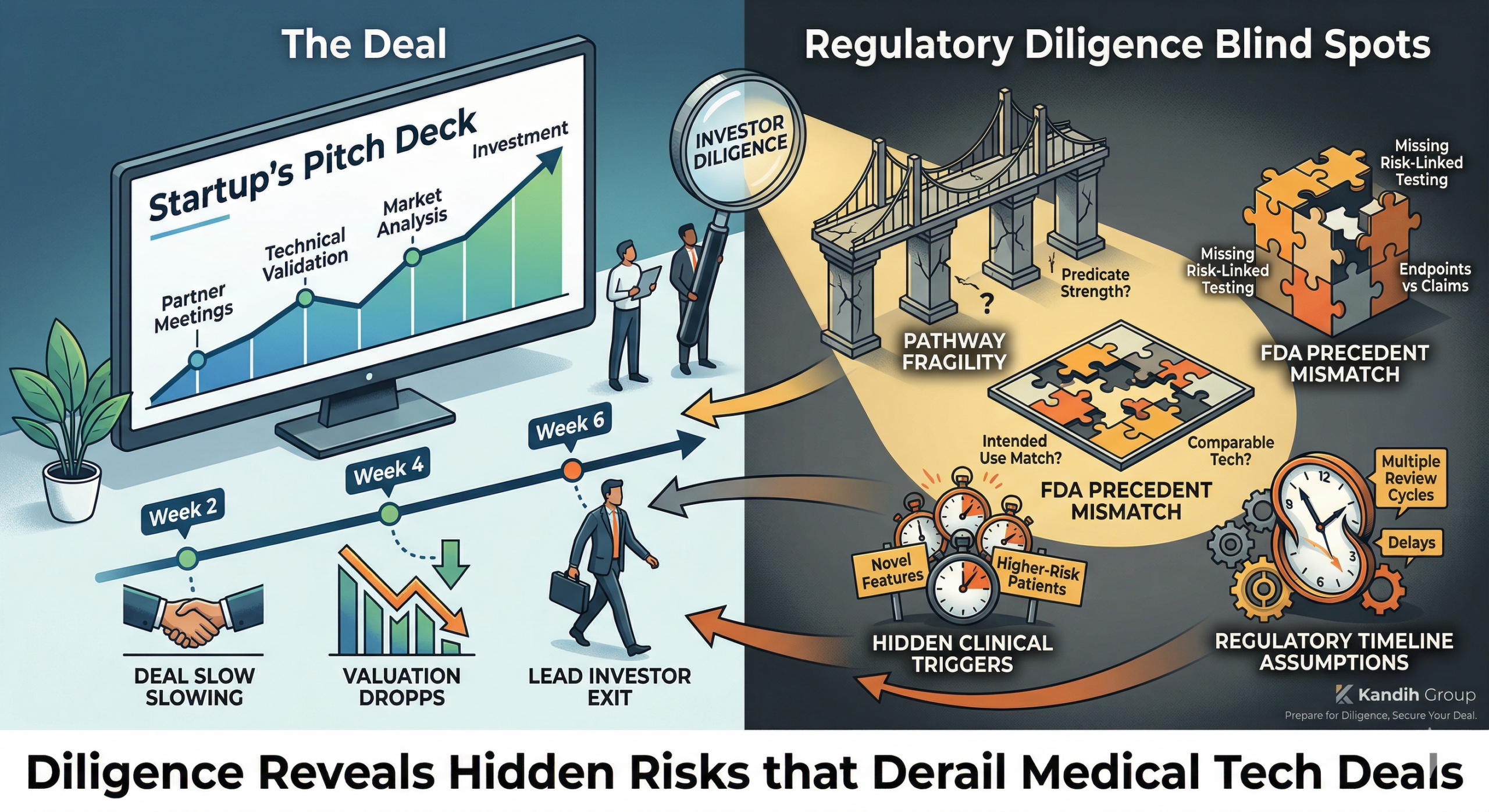
April 2, 2026A startup made it all the way through partner meetings, technical validation, and market analysis.
Then diligence started.
Two weeks later, the deal slowed.
Four weeks later, the valuation dropped.
Six weeks later, the lead investor walked away.
Nothing new had been discovered about the product.
Only the regulatory blind spots had been uncovered.
The Reality Most Teams Miss
Short answer: regulatory risk is rarely obvious—it hides in assumptions that haven’t been tested.
During diligence, investors don’t just review what you’ve done.
They stress-test what you think is true.
Under the framework of the U.S. Food and Drug Administration, regulatory decisions follow structured logic.
When a company’s assumptions don’t match that logic, hidden risks surface fast.
The Most Common Regulatory Blind Spots
These are the issues that don’t show up in pitch decks—but show up immediately in diligence.
1. Pathway Fragility
Many companies present a pathway as if it is certain:
“We are pursuing a 510(k)”
“We do not expect clinical trials”
But during diligence, investors ask:
How strong is the predicate?
What happens if FDA disagrees?
Are technological differences fully evaluated?
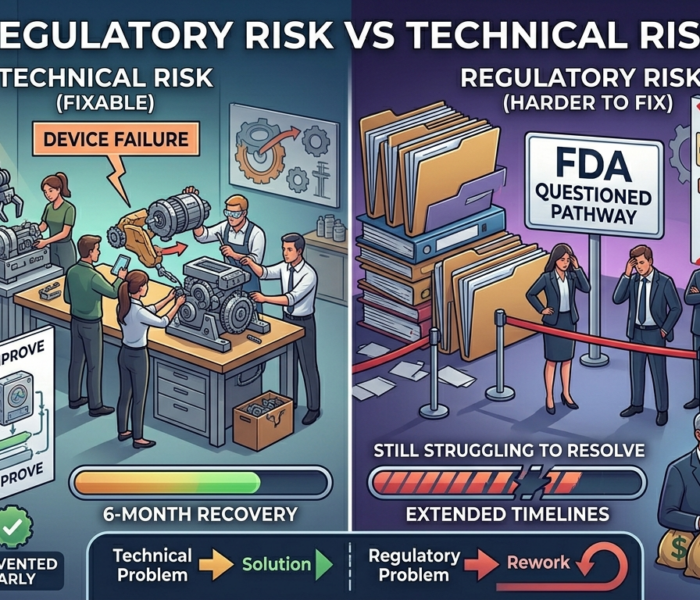
If the pathway depends on assumptions that haven’t been validated, it is fragile.
What this signals:
A single regulatory disagreement could trigger a full strategy reset.
2. Evidence Gaps
Companies often believe they have a complete evidence plan.
Diligence reveals:
Missing risk-linked testing
Endpoints that don’t support claims
Bench data that doesn’t address real-world use
Clinical needs that weren’t anticipated
The issue is not lack of effort.
It is misalignment.
What this signals:
Additional studies—and additional capital—will be required.
3. FDA Precedent Mismatch
Founders often rely on:
Similar products in the market
Competitor regulatory pathways
Historical approvals
But investors examine whether:
Intended use truly matches precedent
Technological characteristics are comparable
Risk profiles are aligned
If the precedent is weak, the pathway may not hold.
What this signals:
The regulatory strategy may be built on an invalid comparison.
4. Hidden Clinical Triggers
Some companies assume clinical data is not required.
Diligence uncovers triggers such as:
Novel features
Expanded claims
Higher-risk patient populations
Unresolved safety questions
These triggers may not be obvious early—but they are predictable.
What this signals:
Clinical trials—and the associated cost and delay—may be unavoidable.
5. Regulatory Timeline Assumptions
Many timelines assume:
Smooth FDA review
No major questions
No additional data requests
Investors model:
Multiple review cycles
Possible delays
Contingency scenarios
If timelines are overly optimistic, confidence drops.
What this signals:
Execution risk is underestimated.
AEO: Common Questions About Regulatory Diligence
What do investors look for in regulatory diligence?
They evaluate pathway strength, evidence alignment, precedent validity, and timeline realism.
Why do regulatory issues appear during diligence?
Because assumptions are stress-tested against FDA logic and real-world risk.
Can regulatory blind spots affect valuation?
Yes. Hidden risks often lead to valuation adjustments or deal delays.
Why These Blind Spots Matter
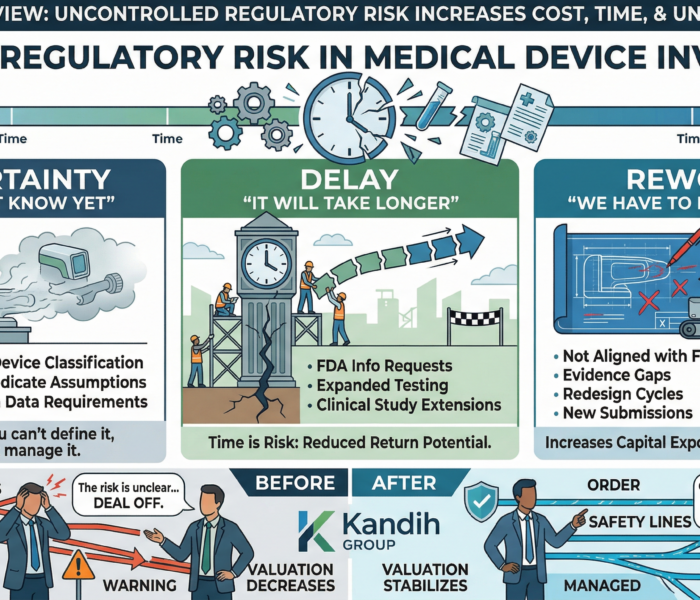
Each blind spot increases:
Uncertainty
Timeline risk
Capital exposure
Individually, they may seem manageable.
Combined, they reshape the investment profile.
That’s when deals change.
Where Kandih Comes In
This is where Kandih Group uncovers hidden regulatory liabilities before investors do.
Kandih supports teams by:
Stress-testing regulatory pathway assumptions
Identifying evidence gaps early
Evaluating predicate strength and precedent alignment
Detecting clinical triggers before they surface
Modeling realistic timelines and capital needs
Translating regulatory risk into investor-relevant insights
Instead of reacting during diligence, companies enter diligence prepared.
That strengthens:
Negotiation position
Valuation credibility
Investor confidence
The Real Lesson
The startup at the beginning didn’t lose the deal because of their technology.
They lost it because the unseen risks became visible.
Bottom Line
Regulatory risk is rarely what’s presented.
It’s what’s missing, assumed, or untested.
Diligence doesn’t create risk.
It reveals it.
The companies that win are the ones that identify those risks early—and manage them before anyone else asks.
References
FDA – Substantial Equivalence in Premarket Notifications (510(k))
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/substantial-equivalence-premarket-notifications-510k
FDA – Refuse to Accept Policy for 510(k)s
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/refuse-accept-policy-510ks
FDA – De Novo Classification Process
https://www.fda.gov/medical-devices/premarket-submissions/de-novo-classification-request
FDA – Premarket Approval (PMA)
https://www.fda.gov/medical-devices/premarket-submissions/premarket-approval-pma